The landscape of breast cancer screening is poised for a significant transformation, as a groundbreaking new study, the WISDOM study, suggests a departure from the long-standing practice of routine annual mammograms for all women. Instead, the research advocates for a tailored approach, matching screening frequency and modalities to a woman’s individual risk profile. This personalized strategy, detailed in findings based on data from 46,000 women across the United States, has demonstrated the potential to enhance both the safety and effectiveness of screening protocols, notably reducing the incidence of advanced cancer diagnoses while ensuring appropriate care levels for all individuals. The study’s initial phase points towards a monumental shift away from generalized, age-based recommendations towards a sophisticated system underpinned by comprehensive risk evaluation, with the University of California, San Francisco (UCSF) serving as the coordinating hub for this pivotal research.
The Evolving Understanding of Breast Cancer Risk
For decades, the prevailing paradigm in breast cancer screening revolved around a "one-size-fits-all" approach, primarily dictated by a woman’s age. This framework, largely influenced by guidelines from organizations such as the American Cancer Society (ACS) and the U.S. Preventive Services Task Force (USPSTF), recommended regular mammography starting at specific ages, often 40 or 50, and continuing at annual or biennial intervals. While these guidelines have undoubtedly saved countless lives by promoting early detection, they were established at a time when understanding of individual risk factors was less advanced. The inherent limitation of this age-centric model is its failure to account for the vast heterogeneity in breast cancer risk among women. Research has consistently shown that a woman’s susceptibility to breast cancer is a complex interplay of genetic predispositions, biological markers, lifestyle choices, and environmental exposures, factors that vary dramatically from person to person.
Traditional guidelines often faced criticism for their broad strokes. For instance, some medical bodies recommend annual mammograms starting at age 40 for women of average risk, while others suggest biennial screening from age 50. This divergence itself highlighted the lack of a universally agreed-upon optimal strategy. Furthermore, the universal approach leads to potential pitfalls: overscreening for women at genuinely low risk, leading to unnecessary anxiety from false positives, biopsies, and cumulative radiation exposure; and conversely, underscreening for women at exceptionally high risk who might benefit from earlier, more frequent, or more advanced screening modalities like MRI. False positives, which can occur in up to 10% of mammograms, often necessitate additional imaging and sometimes biopsies, causing significant psychological distress and financial burden. Overdiagnosis, where a cancer is detected that would never have caused symptoms or harm during a woman’s lifetime, is another concern, potentially leading to overtreatment. The WISDOM study directly addresses these inefficiencies by proposing a more nuanced and precise method of risk stratification.
The Genesis and Methodology of the WISDOM Study
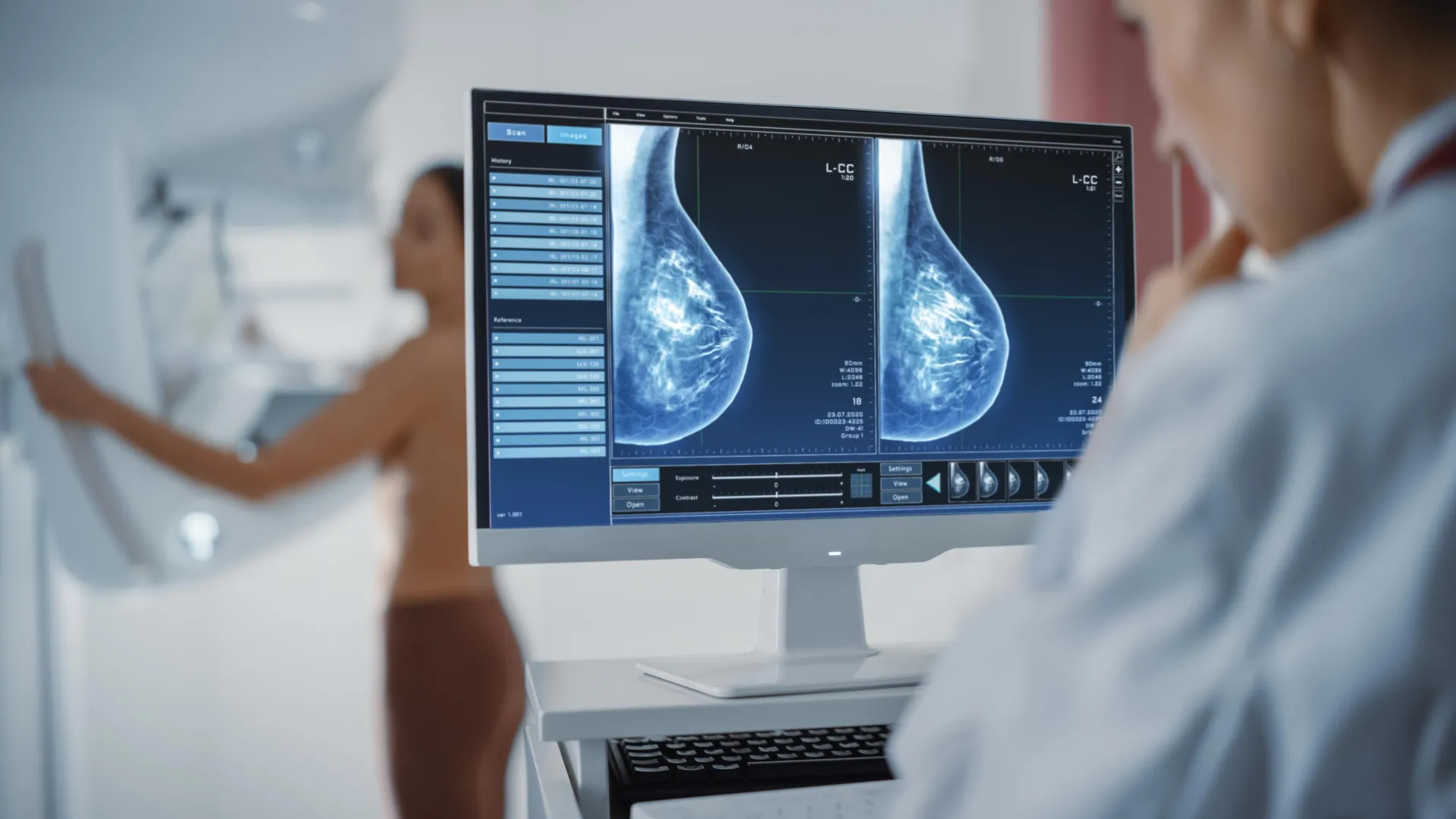
The WISDOM (Women Informed to Screen Depending On Measures of Risk) study, launched in 2016, embarked on a mission to rigorously compare the efficacy of traditional annual mammography against a personalized, risk-based screening strategy. Coordinated by UCSF, the study’s design was ambitious and innovative, enrolling participants from across the United States. In its initial phase, data from 46,000 women formed the bedrock of the findings published in JAMA on December 12, and concurrently presented at the prestigious San Antonio Breast Cancer Symposium. The study’s expansion, now encompassing over 80,000 women, including those as young as 30, underscores its comprehensive scope and long-term vision.
Central to the WISDOM study’s methodology was the deployment of well-validated risk models. These sophisticated algorithms moved beyond simple age assessment, integrating a comprehensive array of data points for each participant. These included demographic information such as age, detailed personal and family health history (e.g., age at first menstruation, age at first live birth, number of biopsies), lifestyle factors (e.g., diet, physical activity, alcohol consumption, body mass index, use of hormone replacement therapy), breast density (a known independent risk factor that can obscure cancers on mammograms), and crucially, genetic information. By synthesizing these diverse elements, which often incorporated established models like the Tyrer-Cuzick model or adaptations, researchers were able to group participants into four distinct risk categories, each warranting a tailored screening regimen. This comprehensive risk assessment aimed to provide a much clearer, individualized picture of a woman’s likelihood of developing breast cancer.
Tailored Screening Recommendations Across Risk Strata
The detailed risk stratification yielded specific recommendations for each group, illustrating the practical application of the personalized approach:
- Lowest Risk Group (26% of participants): Women identified as having the lowest risk were advised to delay routine screening until age 50 or until their risk profile, as determined by the algorithm, escalated to that of a typical 50-year-old woman. This recommendation acknowledges that a significant portion of the population may not benefit from early, frequent screening, thereby reducing unnecessary medical interventions and associated anxieties, while also minimizing cumulative radiation exposure over a lifetime.
- Average Risk Group (62% of participants): The majority of participants fell into this category. For them, biennial screening (every two years) was recommended. This frequency balances the need for detection with minimizing the potential harms of overscreening, aligning with some existing guidelines for this demographic but arrived at through a more individualized assessment, potentially sparing millions of women from annual, often anxiety-inducing, mammograms.
- Elevated Risk Group (8% of participants): For women with an elevated risk profile, annual mammograms were advised. This increased frequency ensures closer surveillance for those with a moderately higher likelihood of developing breast cancer, allowing for earlier detection if the disease were to manifest. This group might include women with a strong family history, certain benign breast conditions, or specific lifestyle factors that collectively elevate their risk.
- Highest Risk Group (2% of participants): This smallest but most vulnerable group received the most intensive screening protocol. Regardless of age, these women were advised to undergo screening twice a year, alternating between mammography and magnetic resonance imaging (MRI). MRI is known for its superior sensitivity in detecting cancers in high-risk women, particularly those with dense breasts or genetic predispositions, providing an enhanced layer of surveillance. This strategy ensures maximal vigilance for those at the greatest genetic or biological predisposition, potentially identifying aggressive cancers at their earliest, most treatable stages.
Personalized Prevention and Patient Empowerment
Beyond merely recommending screening frequencies, the WISDOM study extended its personalized approach to include proactive prevention strategies for higher-risk women. Participants categorized as having elevated or highest risk were not only monitored more closely but also empowered with customized guidance aimed at lowering their overall chances of developing breast cancer. This support system included access to an intuitive online decision-making tool specifically designed to provide information on breast health and risk reduction. Crucially, these women also benefited from direct contact with a breast health specialist, offering personalized advice and fostering a supportive environment for informed decision-making. This direct engagement helped translate complex risk data into understandable, actionable steps.
The recommendations provided encompassed a range of evidence-based interventions. Lifestyle modifications were a key focus, with tailored advice on improving dietary habits (e.g., adopting a plant-rich diet, limiting processed foods) and increasing physical activity levels (e.g., regular moderate-to-vigorous exercise), both of which are known to influence cancer risk by modulating hormones and inflammation. Furthermore, discussions were initiated regarding the potential use of chemoprevention medications, such as tamoxifen or raloxifene, which have been shown to reduce breast cancer risk in high-risk individuals by blocking estrogen’s effects on breast tissue. This holistic approach underscores the study’s commitment not just to early detection, but to comprehensive health management and risk mitigation.
A pivotal outcome reinforcing the efficacy and safety of this personalized model was the finding that it did not lead to a higher rate of late-stage cancer diagnoses. This critical metric demonstrates that by carefully stratifying risk, the study was able to identify cancers at stages no more advanced than those detected by traditional methods, while potentially reducing unnecessary interventions for others. This directly addresses concerns that less frequent screening for low-risk groups might lead to delayed diagnoses. Furthermore, the study included an observational group for women who chose not to be randomized. A remarkable 89% of these women voluntarily opted for the risk-based screening strategy, a strong indicator of patient acceptance and confidence in the personalized approach, suggesting that women are willing to embrace a more nuanced screening schedule when provided with clear, individualized risk information.
Dr. Jeffrey A. Tice, a UCSF professor of Medicine and co-author specializing in breast cancer risk assessment tools, emphasized the efficiency of this strategy. "Shifting resources from lower-risk women to higher-risk women is an efficient, effective approach to screening for and preventing breast cancer," he stated. This highlights the potential for the personalized model to optimize healthcare resource allocation, ensuring that intensive interventions are directed where they can yield the greatest benefit, both in terms of health outcomes and cost-effectiveness. By avoiding unnecessary screenings for low-risk women, resources can be redirected to support more frequent and advanced screenings for high-risk individuals, improving overall public health outcomes without necessarily increasing total healthcare expenditures.
Expanding the Horizon of Genetic Testing
One of the most profound and potentially transformative aspects of the WISDOM study lies in its innovative approach to genetic testing. Since its inception in 2016, the study has expanded its enrollment to over 80,000 women, progressively including participants as young as 30. This deliberate expansion aims to identify younger women who may be at an increased risk of aggressive early-onset cancers due to inherited genetic variants, often before the conventional age for routine screening.
A particularly striking finding from the study was the revelation that 30% of women who tested positive for a genetic variant linked to a higher breast cancer risk reported absolutely no family history of the disease. This statistic is profoundly significant because, under current clinical guidelines from many organizations, a family history of breast or ovarian cancer is often a prerequisite for qualifying for genetic testing. Consequently, a substantial proportion of women carrying high-risk genetic mutations, such as BRCA1 and BRCA2, or other genes like PALB2, CHEK2, and ATM, would traditionally be missed by the healthcare system until a cancer diagnosis occurs, potentially at a later stage. The WISDOM study’s proactive, universal genetic screening within its protocol thus represents a critical step towards identifying these hidden high-risk individuals, enabling earlier preventive measures and targeted surveillance that could dramatically improve their prognoses.
Beyond well-known pathogenic variants like BRCA1 and BRCA2, the study also pioneered the evaluation of smaller, more common DNA changes that, when combined, form a "polygenic risk score" (PRS). A PRS aggregates the effects of numerous common genetic variants, each individually having a small impact on risk, but collectively contributing significantly to an individual’s overall susceptibility. For example, a woman might carry hundreds or thousands of these common variants that, in aggregate, place her at a higher risk, even without a single, high-penetrance gene mutation. This sophisticated approach markedly improved the precision of risk predictions. The integration of PRS resulted in 12% to 14% of participants being reassigned to a different risk category, demonstrating the power of this advanced genetic analysis to refine individual risk assessment beyond what traditional genetic testing or family history alone could achieve. This granular level of genetic insight allows for a more nuanced understanding of individual risk, moving beyond binary "high-risk" or "low-risk" classifications.
Allison S. Fiscalini, MPH, director of the Athena Breast Health Network and the WISDOM study at UCSF, underscored the importance of this pioneering genetic component. "This is one of the first studies to offer genetic testing to all women, regardless of family history," Fiscalini noted. "When used as part of a comprehensive risk assessment, these results could have a real impact on improving the safety and effectiveness of screening and prevention." Her statement highlights the potential for this approach to fundamentally alter who receives genetic testing and, subsequently, who benefits from early, targeted interventions.
Implications for Clinical Guidelines and Public Health
The comprehensive findings of the WISDOM study carry profound implications for the future of breast cancer screening guidelines and clinical practice globally. Dr. Laura J. Esserman, MD, MBA, director of the UCSF Breast Care Center and the study’s first author, articulated this vision clearly: "These findings should transform clinical guidelines for breast cancer screening and alter clinical practice. The personalized approach begins with risk assessment, incorporating genetic, biological, and lifestyle factors, which can then guide effective prevention strategies." Her call to action suggests that the evidence is now compelling enough to warrant a paradigm shift from rigid, age-based protocols to dynamic, individualized plans.
For decades, the recommendations issued by influential bodies like the American Cancer Society (ACS), the U.S. Preventive Services Task Force (USPSTF), and the National Comprehensive Cancer Network (NCCN) have shaped breast cancer screening practices. While these organizations continually review and update their guidelines based on new evidence, the WISDOM study presents a compelling argument for a fundamental re-evaluation of their core tenets. The data suggest that adhering strictly to age-based recommendations may lead to both over-screening for some and under-screening for others, ultimately impacting public health outcomes and resource allocation. The integration of comprehensive risk assessment, including genetic and polygenic risk scores, could lead to more stratified guidelines that acknowledge the diverse risk profiles within the female population. This shift would likely involve recommendations for earlier and more intensive screening for high-risk individuals, while potentially reducing the frequency of screening for those at genuinely low risk, thereby mitigating the harms of overdiagnosis and unnecessary procedures.
The broader impact extends to healthcare economics. Unnecessary mammograms for low-risk women represent a significant cost burden on healthcare systems, estimated to be hundreds of millions of dollars annually in the U.S. alone, when considering the costs of the mammograms themselves, follow-up imaging, and biopsies. Simultaneously, late-stage cancer diagnoses, often a consequence of underscreening high-risk individuals, incur substantially higher treatment costs (e.g., advanced chemotherapy, radiation, surgery, targeted therapies) and yield poorer prognoses. By optimizing screening frequency and modality based on risk, the personalized approach could lead to more efficient allocation of resources, potentially reducing overall healthcare expenditures related to breast cancer management while improving patient outcomes. This shift also holds the promise of reducing patient anxiety and the burden of unnecessary medical procedures.
The Road Ahead: WISDOM 2.0 and Future Directions
The WISDOM study is not a static endpoint but an ongoing journey of scientific discovery and refinement. Researchers are actively continuing to enhance and fine-tune risk assessment methodologies through the WISDOM 2.0 study, which is currently enrolling participants. The overarching objective of this next phase is to further improve the precision with which women at a higher likelihood of developing aggressive breast cancers are identified. By continually refining the risk models and integrating new biological insights, WISDOM 2.0 aims to ensure that screening and prevention strategies are even more precisely tailored to the long-term health needs of each individual. This iterative process of research and development is crucial for translating groundbreaking scientific findings into actionable clinical guidelines and widespread public health benefits.
The successful implementation of risk-based screening on a national scale will require overcoming several challenges. These include educating both healthcare providers and the public about the benefits and nuances of personalized screening, developing standardized and accessible risk assessment tools that can be easily integrated into routine clinical practice, ensuring equitable access to genetic testing and advanced imaging (like MRI) across diverse socioeconomic and