A kidney cancer diagnosis, historically a harbinger of profound uncertainty and fear, is increasingly met with a landscape of evolving treatment options and a burgeoning sense of hope. For individuals and their loved ones grappling with this formidable disease, the advancements in therapeutic strategies, particularly in the realm of immunotherapy, are transforming prognoses and empowering patients to make informed, life-altering decisions. This article delves into the intricacies of kidney cancer, the revolutionary impact of immunotherapy, ongoing research endeavors, and the essential resources available to navigate this challenging journey.
Understanding Kidney Cancer: A Growing Public Health Concern
Kidney cancer originates when cells within the kidneys undergo uncontrolled growth, ultimately forming a tumor. The kidneys, a pair of bean-shaped organs nestled on either side of the spine, are roughly the size of a fist and perform a myriad of vital functions essential for maintaining overall health. These include filtering waste products from the blood, regulating blood pressure, producing urine, and stimulating red blood cell production. Dysfunction in these critical organs can have systemic repercussions, underscoring the severity of kidney-related pathologies.
Kidney cancer encompasses several subtypes, with Renal Cell Carcinoma (RCC) being the most prevalent, accounting for approximately 90% of all kidney cancer diagnoses. Within RCC, clear cell RCC (ccRCC) is the most common histological subtype, followed by papillary RCC and chromophobe RCC. Each subtype exhibits distinct molecular characteristics and may respond differently to various treatments.
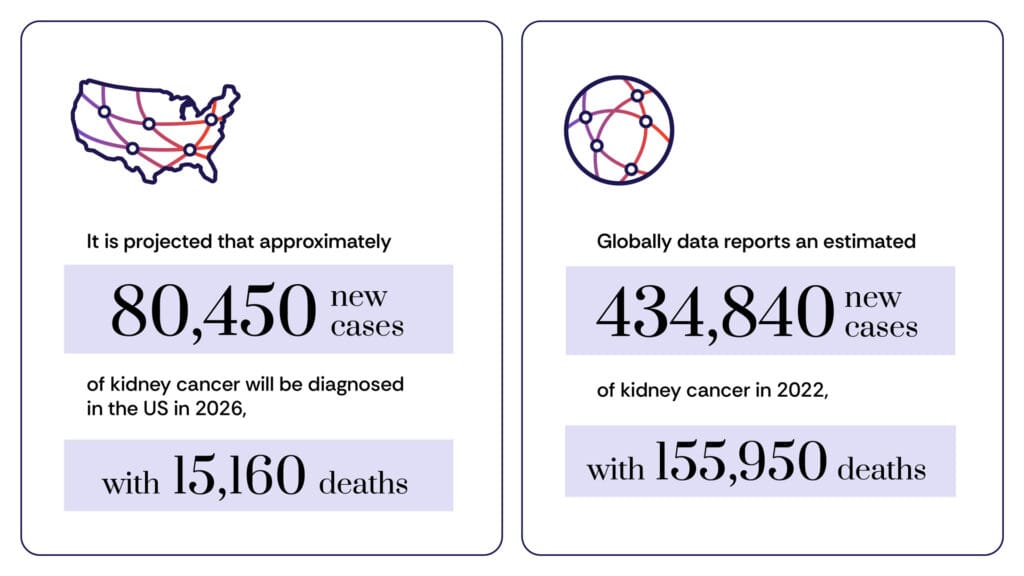
Epidemiologically, kidney cancer ranks among the top ten most common cancers in the United States, representing an estimated 3% to 5% of all cancer diagnoses. The American Cancer Society projected over 81,000 new cases of kidney and renal pelvis cancers and approximately 14,000 deaths from these cancers in the U.S. in 2024. The disease is more frequently diagnosed in older individuals, with the average age at diagnosis being 65, and shows a notable gender disparity, being twice as common in men as in women. Globally, the incidence of kidney cancer has been steadily rising over the past few decades, though encouragingly, death rates have begun to decline in many regions, a testament to advancements in early detection and treatment.
Several risk factors are associated with an increased likelihood of developing kidney cancer. These include smoking, obesity, high blood pressure (hypertension), certain genetic conditions (such as von Hippel-Lindau disease), and long-term dialysis. Occupational exposure to certain substances, such as cadmium or asbestos, has also been implicated. Early symptoms of kidney cancer can be subtle and non-specific, often leading to diagnoses at more advanced stages. Common signs may include blood in the urine (hematuria), persistent back or side pain, a palpable mass in the abdomen, unexplained weight loss, and chronic fatigue. Given the often-asymptomatic nature in early stages, many kidney cancers are incidentally discovered during imaging tests for other conditions. The staging of kidney cancer, which assesses tumor size, lymph node involvement, and distant metastasis, is crucial for guiding treatment decisions and predicting prognosis.

The Evolution of Cancer Therapy: Paving the Way for Immunotherapy
The history of cancer treatment has seen a continuous quest for more effective and less toxic interventions. For decades, the primary pillars of cancer care were surgery, radiation therapy, and chemotherapy. Surgery aimed to physically remove tumors, radiation therapy utilized high-energy rays to destroy cancer cells, and chemotherapy employed potent drugs to kill rapidly dividing cells throughout the body. While these modalities have saved countless lives, they often came with significant side effects due to their non-specific nature, impacting healthy cells alongside cancerous ones.
The late 20th and early 21st centuries witnessed the emergence of targeted therapies, a significant leap forward. These drugs were designed to specifically interfere with molecular pathways crucial for cancer growth and survival, often with fewer systemic side effects than traditional chemotherapy. For kidney cancer, tyrosine kinase inhibitors (TKIs) like sunitinib and pazopanib became standard, targeting pathways involved in angiogenesis (blood vessel formation) and tumor proliferation. While targeted therapies improved outcomes, many patients eventually developed resistance, necessitating further innovation.
The true paradigm shift in oncology came with the advent of immunotherapy. This revolutionary approach harnesses the body’s own immune system, a sophisticated network of cells and organs designed to protect against disease, to recognize and destroy cancer cells. Unlike previous treatments that directly attacked the tumor, immunotherapy empowers the immune system to become the primary weapon, offering the potential for durable responses and, in some cases, long-term remission.
Immunotherapy’s Mechanism: A Deeper Dive into Immune Checkpoint Inhibition
The immune system’s ability to distinguish between "self" and "non-self" is fundamental to its protective function. However, cancer cells are adept at developing sophisticated mechanisms to evade immune surveillance. They can mimic healthy cells, suppress immune cell activity, or even express molecules that act as "brakes" on the immune response, preventing T-cells from launching an attack.
Immunotherapy works by "unmasking" these cancer cells and "unleashing" the immune system. In kidney cancer, the most successful form of immunotherapy currently in use involves immune checkpoint inhibitors (ICIs). These therapies target specific proteins, or "checkpoints," on immune cells or cancer cells that normally serve to regulate the immune response and prevent autoimmunity. By blocking these inhibitory signals, ICIs effectively remove the "brakes" on the immune system, allowing T-cells to recognize and vigorously attack cancer cells.

Two key checkpoint pathways targeted in kidney cancer are the Programmed Death-1 (PD-1)/Programmed Death-Ligand 1 (PD-L1) pathway and the Cytotoxic T-Lymphocyte Antigen 4 (CTLA-4) pathway.
- PD-1/PD-L1 Inhibitors: PD-1 is a protein on the surface of T-cells that, when bound by PD-L1 (expressed on cancer cells or other immune cells), signals the T-cell to stand down. PD-1 or PD-L1 inhibitors block this interaction, allowing T-cells to remain active and kill cancer cells. Examples approved for kidney cancer include Nivolumab (Opdivo), Pembrolizumab (Keytruda), Avelumab (Bavencio), and Cemiplimab (Libtayo).
- CTLA-4 Inhibitors: CTLA-4 is another checkpoint protein found on T-cells that acts early in the immune response to downregulate T-cell activation. Ipilimumab (Yervoy) is a CTLA-4 inhibitor that, by blocking CTLA-4, enhances the initial priming and activation of T-cells, thereby boosting the anti-tumor immune response.
It is crucial for patients and their families to engage in thorough discussions with their healthcare providers to determine if immunotherapy is a suitable treatment option, given the individual characteristics of their cancer and overall health profile.
Transforming Kidney Cancer Treatment: Immunotherapy’s Clinical Impact
The introduction of ICIs has profoundly altered the treatment landscape for advanced kidney cancer. Initially approved for metastatic RCC that had progressed on prior targeted therapies, immunotherapy has since moved into first-line treatment and even adjuvant settings (after surgery to prevent recurrence).
A significant breakthrough has been the demonstration of superior efficacy of combination immunotherapies, or immunotherapy combined with targeted therapies, over older standard treatments.
- ICI-ICI Combinations: The combination of a PD-1 inhibitor (Nivolumab) and a CTLA-4 inhibitor (Ipilimumab) was a landmark development, showing improved overall survival compared to sunitinib in intermediate- and poor-risk metastatic ccRCC patients in the CheckMate 214 trial. This combination leverages different points in the immune activation pathway, leading to a more robust anti-tumor response.
- ICI-TKI Combinations: Combining a PD-1 inhibitor with a TKI has also proven highly effective. For instance, the KEYNOTE-426 trial demonstrated that Pembrolizumab combined with axitinib (a TKI) significantly improved overall survival and progression-free survival compared to sunitinib alone in previously untreated advanced RCC. Similarly, the CLEAR trial showed that Pembrolizumab combined with lenvatinib (another TKI) significantly improved PFS and OS compared to sunitinib. These combinations exploit the complementary mechanisms of action, where TKIs can reduce tumor burden and create a more immune-permissive tumor microenvironment, while ICIs unleash the immune system.
These clinical trial results have translated into real-world benefits, leading to improved progression-free survival and, critically, extended overall survival for many patients with advanced kidney cancer. The "Did you know?" fact from the original content bears repeating here: while the annual rate of new kidney cancer diagnoses has been rising for years, death rates have been falling. This encouraging trend directly reflects the advances in treatment and care, with immunotherapy playing a pivotal role in helping more people live longer and with better quality of life.
However, immunotherapy is not without its challenges. While generally better tolerated than chemotherapy, ICIs can cause unique side effects known as immune-related adverse events (irAEs). These occur when the activated immune system mistakenly attacks healthy tissues, leading to inflammation in various organs such as the skin (rashes), colon (colitis), endocrine glands (thyroiditis, hypophysitis), liver (hepatitis), and lungs (pneumonitis). Effective management of irAEs requires prompt recognition and intervention, often involving corticosteroids or other immunosuppressants, highlighting the importance of specialized oncology care teams experienced in immunotherapy.
The Promise of Tomorrow: Frontiers in Kidney Cancer Research
The rapid success of immunotherapy has fueled an intense period of research, with scientists and clinicians relentlessly pursuing even more effective and personalized strategies for kidney cancer. Ongoing investigations are focused on several key areas:
- Improving Survival in Metastatic Cases: While current immunotherapies have significantly improved outcomes, many patients still do not respond or eventually develop resistance. Research aims to understand these mechanisms of resistance and develop strategies to overcome them, potentially through novel combinations or new classes of drugs.
- Reducing Recurrence After Surgery (Adjuvant Therapy): Expanding the use of immunotherapy in the adjuvant setting after surgical removal of the primary tumor is a critical goal. Clinical trials are evaluating whether immunotherapy can reduce the risk of cancer returning in patients at high risk of recurrence, thereby improving long-term, disease-free survival.
- Personalized Vaccines and Neoantigen Approaches: A promising area involves developing personalized vaccines that target specific "neoantigens" – unique mutations found on a patient’s tumor cells. By identifying these neoantigens, researchers can design vaccines to educate the immune system to specifically recognize and attack the patient’s cancer cells, offering a highly tailored approach.
- Advancements in CAR T-Cell Therapy for Solid Tumors: While CAR T-cell therapy has achieved remarkable success in certain blood cancers, its application in solid tumors like kidney cancer presents unique challenges, including difficulty in trafficking to the tumor site and navigating the immunosuppressive tumor microenvironment. However, ongoing research is exploring novel CAR T-cell designs, alternative targets, and combination strategies to make this powerful therapy effective for kidney cancer.
- Exploring Novel Immune Modulators and Bispecific Antibodies: Beyond current ICIs, researchers are investigating other pathways and molecules that can modulate the immune response. This includes agonists for co-stimulatory receptors (e.g., OX40, CD137), antagonists for other inhibitory receptors, and bispecific antibodies that can simultaneously bind to cancer cells and T-cells, bringing them into close proximity to facilitate an attack.
- The Crucial Search for Predictive Biomarkers: A significant challenge in immunotherapy is identifying reliable biomarkers that can predict which patients are most likely to respond to specific treatments. Such biomarkers would enable clinicians to personalize therapy, avoiding ineffective treatments and unnecessary toxicities for non-responders. Research is actively exploring genetic mutations, gene expression profiles, tumor microenvironment characteristics, and circulating tumor DNA as potential predictive markers.
- The Role of Clinical Trials in Accelerating Progress: Clinical trials remain the bedrock of medical advancement. They are essential for testing new therapies, combinations, and treatment sequences, ensuring that the most promising innovations reach patients safely and effectively. Participation in clinical trials offers patients access to cutting-edge treatments that may not yet be widely available.
Continued dedication to research in these areas is not only expanding the arsenal of available therapies but is also increasingly driven by patient voices, shaping research priorities and ensuring that future treatments are grounded in real-world needs. The ultimate goal is to offer every patient more hope, more time, and a better quality of life.
Navigating a Diagnosis: Empowerment Through Knowledge and Support
Receiving a kidney cancer diagnosis can be an emotionally overwhelming experience, whether it is for oneself or a loved one. In this challenging time, empowerment stems from education, advocacy, and a robust support system.
- Be Your Own Best Advocate: Patients are encouraged to actively participate in their care, asking questions, seeking second opinions, and understanding all available options.
- Educate Yourself: Learn about your specific cancer type, its stage, the nuances of different treatment modalities (including immunotherapy), and the latest research. Reputable sources like the Cancer Research Institute, National Cancer Institute, and the Kidney Cancer Association offer invaluable information.
- Build a Strong Care Team: A multidisciplinary approach is paramount. This team typically includes a urologist, medical oncologist, radiation oncologist, pathologist, radiologist, oncology nurses, nurse navigators, dietitians, social workers, and palliative care specialists. Each plays a vital role in providing comprehensive care.
- Seek Support: Do not face cancer alone. Reach out to family, friends, and consider joining patient support groups. Connecting with others who understand the journey can provide immense emotional and practical support. Organizations like the Kidney Cancer Association offer dedicated resources and communities for patients and caregivers.
When discussing immunotherapy with your care team, several key questions can help clarify the path forward:
- "What type of kidney cancer do I have, and what is its stage?"
- "Is immunotherapy a suitable option for my specific situation?"
- "What are the potential benefits and risks of immunotherapy in my case?"
- "How does immunotherapy compare to other treatment options available to me?"
- "What specific immunotherapy drugs or combinations are being considered, and why?"
- "What are the common immune-related side effects I might experience, and how will they be managed?"
- "What is the expected duration of treatment, and how will my response be monitored?"
- "Are there any clinical trials relevant to my diagnosis that I should consider?"
- "What resources are available to help me manage the financial aspects of treatment?"
- "How will this treatment impact my daily life, and what support services are available?"
Broader Implications and Future Outlook

The advancements in kidney cancer treatment, particularly with immunotherapy, carry significant broader implications. The improved survival rates are shifting the paradigm for many patients from a focus on acute, life-threatening illness to one of managing a chronic condition. For some, durable responses mean living longer with cancer, potentially requiring long-term monitoring and adaptive treatment strategies.
However, the cost of advanced therapies like immunotherapy presents a substantial challenge to healthcare systems globally. Issues of accessibility and affordability are critical, ensuring that these life-saving treatments are available to all who can benefit, regardless of socioeconomic status or geographic location. This necessitates ongoing discussions among policymakers, pharmaceutical companies, and patient advocates to find sustainable solutions.
Looking ahead, the future of kidney cancer treatment is characterized by an unwavering commitment to precision medicine. The integration of genomics, proteomics, and advanced imaging will enable highly personalized treatment plans, tailored to the unique molecular fingerprint of each patient’s tumor. The ongoing work is not merely expanding the list of treatment options; it is profoundly reshaping the narrative around kidney cancer, replacing despair with a robust and evolving sense of hope. With continued research, collaboration, and patient-centered care, the goal of giving every patient more time and a better life moves closer to becoming a reality.

