Northwestern University scientists have achieved a significant breakthrough in spinal cord injury (SCI) research, developing the most sophisticated lab-grown model to date capable of mimicking the complex biological aftermath of human SCI. This advanced model, utilizing human spinal cord organoids, has not only replicated key pathological consequences of injury but has also demonstrated remarkable efficacy for a novel regenerative treatment, offering renewed hope for millions affected by paralysis and loss of function.
The groundbreaking research, published on February 11th in the esteemed journal Nature Biomedical Engineering, marks a pivotal moment in the quest for effective SCI therapies. For the first time, researchers have successfully employed human spinal cord organoids – miniature, self-organizing structures derived from stem cells – to accurately recapitulate the multifaceted biological responses to traumatic injury. This includes critical elements such as widespread cell death, robust inflammatory cascades, and the formation of glial scars, a dense barrier of scar tissue that notoriously impedes nerve regeneration.
A New Era of In Vitro Research: The Human Spinal Cord Organoid
Organoids, derived from induced pluripotent stem cells (iPSCs), represent a paradigm shift in biological research. These lab-grown entities, while simplified versions of full organs, exhibit remarkable fidelity in mirroring the structural complexity, cellular diversity, and functional characteristics of their in vivo counterparts. This makes them invaluable tools for studying disease pathogenesis, testing therapeutic interventions, and unraveling developmental processes, all while offering a more rapid and cost-effective alternative to animal models and human clinical trials.
While previous efforts have yielded spinal cord organoids for fundamental biological studies, the Northwestern team’s model represents a substantial leap forward for injury research. These organoids, measuring several millimeters in diameter, have achieved a level of maturity that allows them to sustain and accurately model traumatic damage. The research team meticulously guided stem cells over several months to develop intricate spinal cord tissue. Crucially, they were the first to successfully incorporate microglia, the resident immune cells of the central nervous system, into these organoids. This inclusion is vital for faithfully replicating the acute inflammatory response that is a hallmark of SCI.
“It’s akin to a pseudo-organ,” explained Samuel I. Stupp, the study’s senior author and a distinguished Board of Trustees Professor at Northwestern University. Stupp, a leading figure in regenerative materials science and director of the Center for Regenerative Nanomedicine (CRN), highlighted the significance of this advancement. “We were the first to introduce microglia into a human spinal cord organoid, which was a huge accomplishment. This means our organoid possesses all the signaling molecules that the resident immune system produces in response to an injury. This makes it a more realistic and accurate model of spinal cord injury.”
Replicating the Devastation of Spinal Cord Injury
To rigorously test their model and therapeutic approach, the Northwestern researchers simulated two prevalent forms of spinal cord trauma within the organoids. One injury model involved a precise scalpel cut, mimicking lacerations that can occur during surgery or severe physical trauma. The second model involved a compressive contusion, designed to replicate the blunt force impact associated with events like serious car accidents or falls.
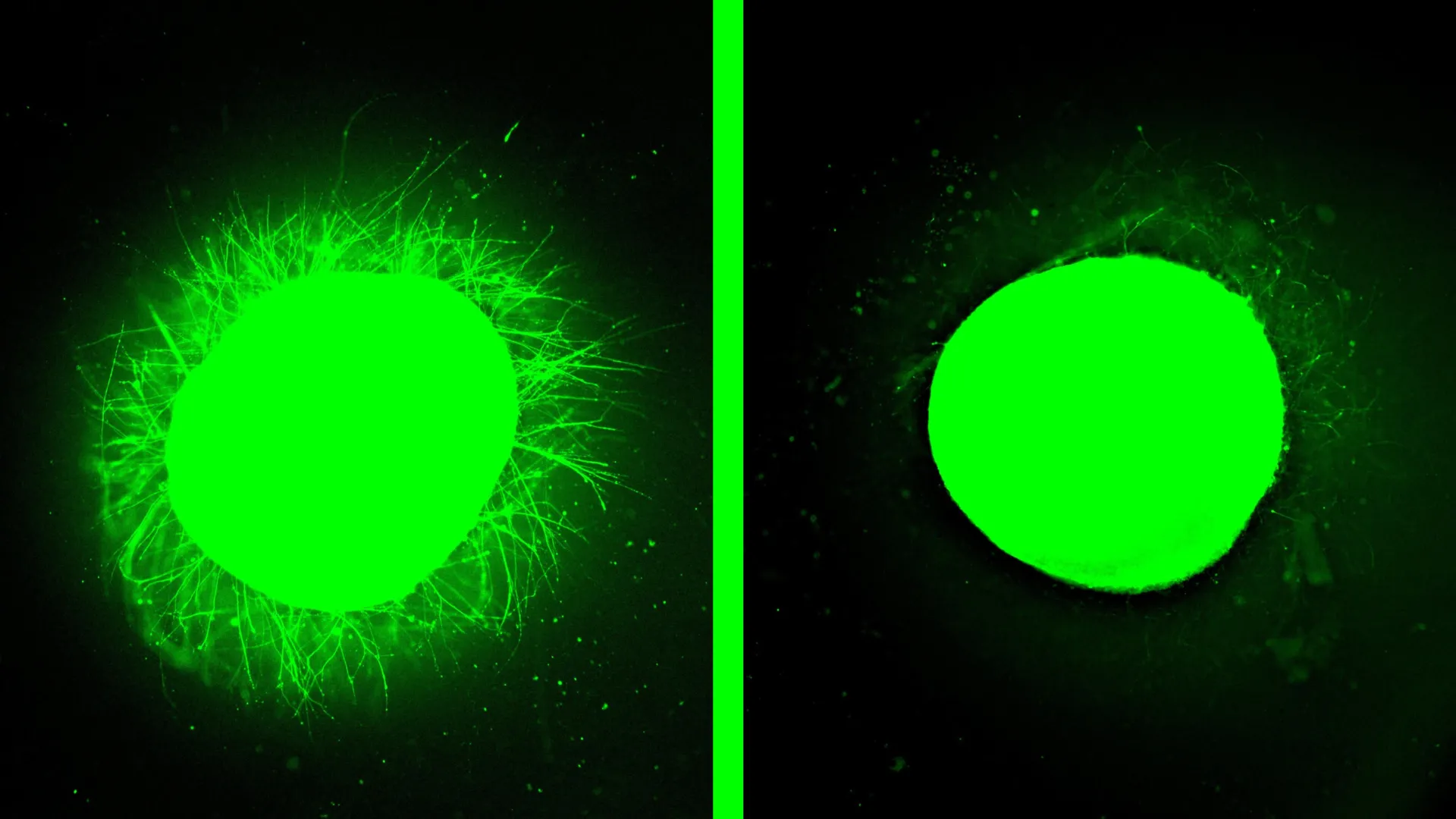
In both injury scenarios, the organoids exhibited characteristic pathological features observed in human SCIs. Extensive cell death was documented, alongside the pronounced formation of glial scars. These scars, a critical impediment to neurological recovery, are composed of reactive astrocytes and other cells that proliferate and deposit extracellular matrix proteins, creating a physical and biochemical barrier that prevents damaged neurons from regrowing their axons.
“We were able to clearly differentiate between the astrocytes that constitute normal tissue and those within the glial scar, which are significantly enlarged and densely packed,” Stupp elaborated. “Furthermore, we detected the production of chondroitin sulfate proteoglycans, molecules within the nervous system that are known to play a crucial role in responding to injury and disease.” The presence of these specific biological markers further validates the organoid model’s capacity to accurately reflect the complex cellular and molecular changes that occur following SCI.
The Promise of "Dancing Molecules" in Tissue Regeneration
The true game-changer in this research lies in the application of a novel regenerative therapy developed by Stupp’s lab, dubbed "dancing molecules." This innovative treatment, which leverages the dynamic motion of therapeutic molecules to promote healing, has shown dramatic results in the injured organoids. The therapy, a type of supramolecular therapeutic peptide (STP), was first introduced in 2021. It works by utilizing controlled molecular motion to repair tissue and potentially reverse paralysis following traumatic SCI.
STPs function by forming large assemblies of over 100,000 molecules that interact with cell receptors, thereby activating the body’s intrinsic repair mechanisms. The concept of supramolecular therapies, which harness the collective behavior of molecules, is also present in contemporary GLP-1 drugs used for weight loss and diabetes, an area Stupp’s lab explored nearly 15 years ago.
In the current study, when the damaged spinal cord organoids were treated with these "dancing molecules," the outcomes were striking. The injured tissue displayed substantial neurite outgrowth, a critical indicator of nerve repair. Neurites are the slender projections of nerve cells, including axons, which are essential for transmitting signals between neurons. The therapy also led to a significant reduction in scar-like tissue, making it barely detectable.
“One of the most exciting aspects of organoids is their utility in testing new therapies directly in human tissue,” Stupp stated. “Short of conducting a clinical trial, this is the only objective way to achieve such an assessment. We designed two distinct injury models within a human spinal cord organoid and applied our therapy to ascertain if the results mirrored those previously observed in animal studies. Following the application of our therapy, the glial scar substantially faded, becoming nearly imperceptible, and we observed significant neurite growth, mirroring the axon regeneration seen in our animal models. This strongly suggests that our therapy has a high probability of efficacy in humans.”
Mechanism of Action: The Power of Molecular Dynamics
The effectiveness of the "dancing molecules" therapy is attributed to its supramolecular motion. This refers to the molecules’ ability to move rapidly and even temporarily detach from their supportive nanofiber network. This dynamic behavior is crucial for their interaction with cell receptors, which are themselves in constant motion.
“Given that cells and their receptors are in perpetual motion, one can infer that molecules exhibiting faster movement would encounter these receptors more frequently,” Stupp explained in a previous context. “If molecules are sluggish and less ‘social,’ they may never establish contact with the cells.”
The therapy is administered as a liquid injection that swiftly self-assembles into a nanofiber scaffold, mimicking the extracellular matrix of the spinal cord. By fine-tuning the molecular dynamics within this structure, researchers have enhanced the therapy’s ability to engage with the continuously shifting cell receptors.
In prior animal experiments, a single injection of this therapy, administered 24 hours post-injury, enabled mice to regain mobility within four weeks. Formulations with more rapid molecular motion consistently outperformed slower versions, underscoring the principle that increased molecular dynamism enhances bioactivity and cellular signaling.
The research team further reinforced the importance of molecular motion through experiments on healthy organoids. “Even before we developed the injury model, we tested the therapy on a healthy organoid,” Stupp recounted. “The dancing molecules generated extensive long neurites on the organoid’s surface. In stark contrast, when we used molecules with minimal or no motion, we observed no such outgrowth. The difference was remarkably vivid.”
Broader Implications and Future Directions
The promising results obtained from this study have significant implications for the future of spinal cord injury treatment. The U.S. Food and Drug Administration (FDA) has already granted Orphan Drug Designation to this "dancing molecules" therapy, a status that provides incentives for developing drugs for rare diseases and conditions, including SCI. This designation signifies the FDA’s recognition of the therapy’s potential and its commitment to facilitating its development.
The findings provide robust validation for the therapeutic potential of "dancing molecules" in human tissue, moving the scientific community closer to realizing effective treatments for SCI. The ability to accurately model SCI in a human organoid system also opens doors for accelerated drug discovery and development. Researchers can now screen and test a multitude of potential therapeutic agents with unprecedented speed and accuracy, significantly reducing the time and cost associated with traditional research methodologies.
Looking ahead, the Northwestern team plans to engineer even more advanced organoid models. Their future endeavors include developing organoids that can replicate chronic SCI, a condition characterized by thicker and more persistent scar tissue, which presents a greater therapeutic challenge. Stupp also envisions a future where these miniature spinal cords could play a role in personalized medicine. By generating implantable tissue from a patient’s own stem cells, the risk of immune rejection could be substantially minimized, paving the way for highly individualized regenerative therapies.
The study, titled "Injury and therapy in a human spinal cord organoid," received crucial support from the Center for Regenerative Nanomedicine at Northwestern University and a generous gift from the John Potocsnak Family specifically designated for spinal cord injury research. This collaborative effort underscores the importance of sustained investment and interdisciplinary approaches in tackling complex medical challenges.
The development of this sophisticated human spinal cord organoid model, coupled with the demonstrated efficacy of the "dancing molecules" therapy, represents a monumental stride forward. It offers a tangible pathway toward restoring function and improving the quality of life for individuals living with the devastating consequences of spinal cord injury. The scientific community will be closely watching as this research progresses toward potential clinical applications.