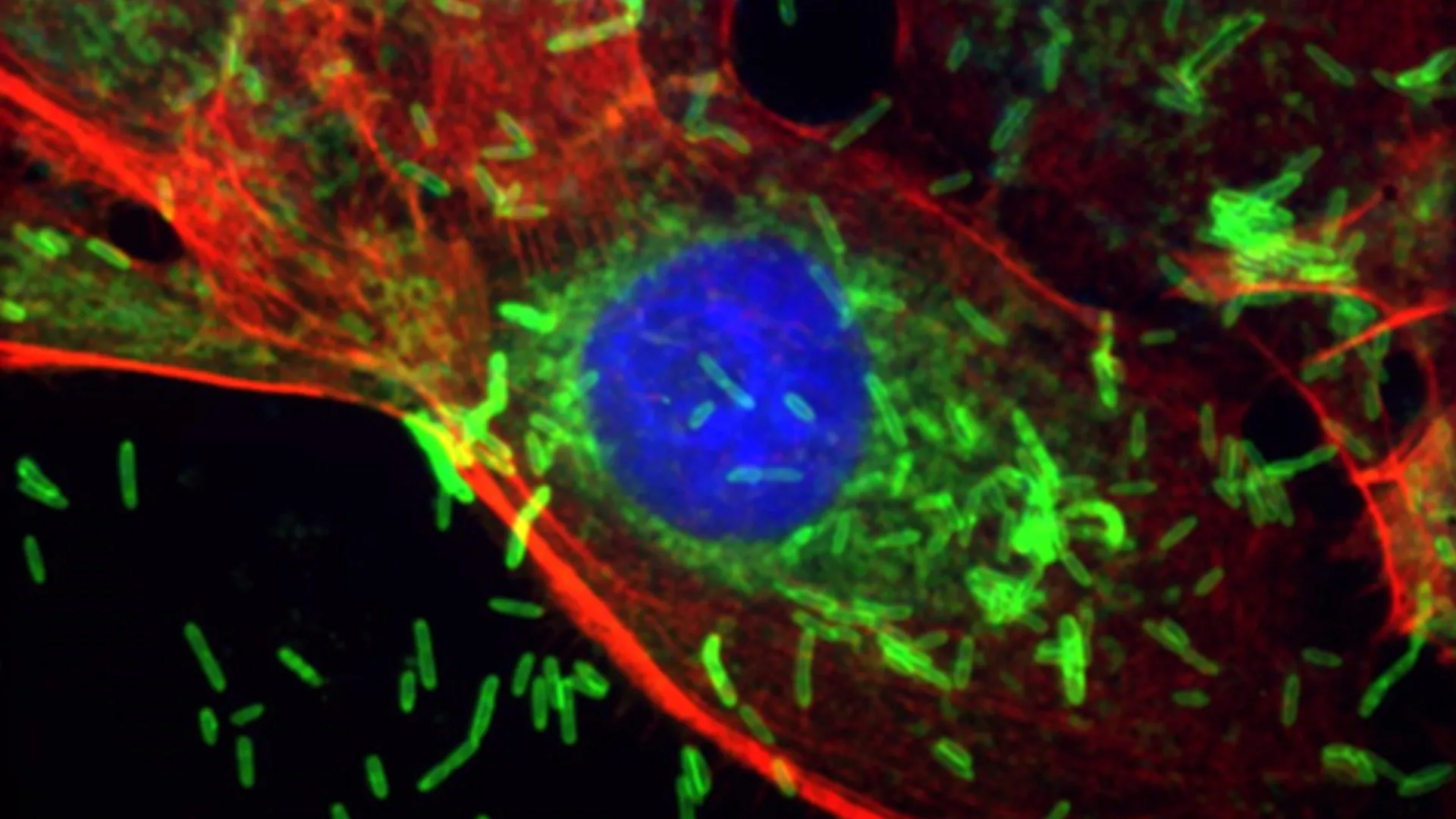
Unveiling the Microbial Culprit: Fusobacterium nucleatum
The study primarily focuses on Fusobacterium nucleatum, a microbe that has garnered increasing attention in oncology research over the past decade. Historically recognized as a significant pathogen in oral infections, particularly periodontitis or gum disease, F. nucleatum has more recently been implicated in a range of systemic health issues, including inflammatory bowel disease and adverse pregnancy outcomes. Critically, prior to this Johns Hopkins investigation, F. nucleatum had already been definitively linked to colorectal cancer, where its presence in tumor tissue is associated with poorer prognoses and resistance to chemotherapy. This established notoriety provided a compelling impetus for the Johns Hopkins team to explore its potential role in other malignancies, especially given the growing body of observational evidence linking oral health to breast cancer.
The research meticulously details how this ubiquitous oral bacterium transcends its natural habitat. The scientists demonstrated that F. nucleatum possesses the remarkable ability to enter the bloodstream, likely facilitated by inflamed or bleeding gums characteristic of periodontal disease. Once in circulation, it can travel through the body and specifically settle within breast tissue, an environment previously considered relatively sterile from such microbial invaders. Upon colonization, the bacterium initiates a cascade of detrimental biological processes. It triggers localized inflammation, a well-established precursor to cancerous changes, and induces early cellular alterations within the breast tissue itself. These changes are not benign; in animal models designed to mimic human breast cancer, the presence of F. nucleatum significantly accelerated the growth rate of existing tumors and dramatically increased the metastatic spread of cancer cells from the primary breast tumor to distant sites, most notably the lungs. This direct demonstration of its capacity to influence both primary tumor progression and metastasis underscores its potential as a formidable oncogenic cofactor.
"The key takeaway from our extensive investigation is the unequivocal demonstration that this oral microbe can not only reside within breast tissue but that there is a profound, mechanistic connection between this pathogen and the initiation and progression of breast cancer," explained Dr. Sharma, a senior author of the study. Dr. Sharma elaborated that the team’s initial curiosity and subsequent deep dive were inspired by a multitude of smaller-scale epidemiological studies. Over recent years, a growing body of research, encompassing thousands of patients across various demographics, has consistently identified a statistical correlation between periodontal disease and an elevated risk of breast cancer. These observational links, while significant, lacked the biological mechanistic explanations that the Johns Hopkins team sought to uncover. "We felt it was imperative to dig deeper beyond mere correlation and see if we could uncover the underlying, intricate biological connections that bridge oral health to such a devastating systemic disease," added Dr. Sheetal Parida, the first author of the study and a dedicated research associate working alongside Dr. Sharma.
The Molecular Blueprint of Damage: DNA Alterations and Cellular Reprogramming
To unravel the precise mechanisms through which F. nucleatum exerts its oncogenic influence, the researchers employed a sophisticated array of experimental approaches, utilizing both advanced mouse models and human breast cancer cell lines. These experiments provided crucial insights into how the bacterium physically affects breast tissue at a cellular and molecular level.
In a particularly revealing set of experiments, F. nucleatum was directly introduced into the breast ducts of experimental models. This localized exposure resulted in the rapid development of metaplastic and hyperplastic lesions. These are noncancerous, yet concerning, cellular changes characterized by either an excessive proliferation of cells (hyperplasia) or a transformation of cells into a different type (metaplasia). Importantly, these morphological changes were consistently accompanied by a pronounced inflammatory response within the breast tissue, direct evidence of DNA damage at the cellular level, and a significant increase in cell proliferation rates—all hallmarks of early-stage oncogenesis. When the bacterium was introduced systemically, entering the bloodstream, it exhibited an even more aggressive profile, significantly boosting the growth and metastatic spread of pre-existing tumors, confirming its role in accelerating disease progression.
The team also meticulously uncovered a critical biological process underpinning these observed effects. Exposure to F. nucleatum was found to directly inflict damage upon cellular DNA, a common pathway to cancer. However, more alarmingly, it also activated cellular repair systems that are inherently prone to errors. One such system, known as nonhomologous end joining (NHEJ), is a rapid but often imprecise mechanism for reconnecting broken DNA strands. While essential for cell survival, NHEJ can introduce mutations during its repair process, thereby increasing genetic instability—a fundamental driver of cancer. Furthermore, even a brief exposure to F. nucleatum led to a marked increase in the levels of a specific protein called PKcs. This protein was found to be intricately linked to several highly aggressive characteristics in cancer cells: enhanced cell movement, increased invasive potential, acquisition of stem-like traits (which contribute to tumor recurrence and resistance), and, critically, heightened resistance to conventional chemotherapy agents. This molecular insight provides a potential therapeutic target and explains how F. nucleatum could contribute to more aggressive and difficult-to-treat forms of breast cancer.
Genetic Vulnerability: The Critical Role of BRCA1 Mutations
A particularly striking finding of the study was the identification of specific cellular populations that appeared to be disproportionately susceptible to the detrimental effects of F. nucleatum. Epithelial cells, which form the lining of the breast ducts where most breast cancers originate, showed heightened vulnerability. Even more significantly, breast cancer cells carrying mutations in the BRCA1 gene exhibited an amplified susceptibility.
Individuals with inherited BRCA1 mutations are known to have a substantially increased lifetime risk of developing breast and ovarian cancers. The Johns Hopkins team discovered a crucial molecular explanation for their enhanced vulnerability to F. nucleatum. These BRCA1-mutant cells were found to express elevated levels of a specific surface sugar, Gal-GalNAc (galactose-N-acetylgalactosamine). This particular sugar acts as a molecular "handshake," facilitating the attachment and subsequent entry of F. nucleatum into the cells. As a direct consequence, BRCA1-mutant cells absorbed significantly greater quantities of F. nucleatum and, disturbingly, retained the bacteria over extended periods, even across multiple cell generations. This prolonged intracellular presence of the bacterium intensified the DNA damage and amplified the array of cancer-promoting effects, suggesting a synergistic interaction between genetic predisposition and environmental microbial factors.
"Our findings represent a pivotal step, revealing a direct, mechanistic link between common oral microbes and both breast cancer risk and its progression, especially in genetically susceptible individuals such as those with BRCA1 mutations," Dr. Sharma emphasized. She further elaborated on the complex interplay of risk factors, stating, "Nothing in biology happens in isolation. The results strongly suggest that multiple risk factors converge, with F. nucleatum acting as a potent environmental factor that may cooperate with inherited BRCA1 mutations to not only promote breast cancer but also to drive increased tumor aggressiveness." This insight profoundly reshapes our understanding of breast cancer etiology, moving beyond a purely genetic or purely environmental viewpoint to embrace a multifactorial model.
A Broader Scientific Landscape: Connecting Oral Health and Systemic Disease
The Johns Hopkins study is not an isolated finding but rather a significant piece in a larger, evolving scientific puzzle that connects the human microbiome to systemic health and disease. For decades, the oral cavity was largely considered separate from the rest of the body in terms of its microbial impact, beyond acute infections. However, this perspective has dramatically shifted. The "many small studies" mentioned by Dr. Sharma, which inspired this deeper investigation, have consistently highlighted associations between chronic periodontal disease—a common inflammatory condition affecting up to half of adults worldwide—and an increased risk for various systemic diseases, including cardiovascular disease, diabetes, rheumatoid arthritis, and certain cancers.
The concept of the "oncomicrobiome"—the idea that specific microbial communities or individual pathogens can directly contribute to cancer development—has gained substantial traction. Beyond F. nucleatum‘s role in colorectal cancer and now breast cancer, other examples include Helicobacter pylori and gastric cancer, and the human papillomavirus (HPV) and cervical cancer. These cases demonstrate that microbes can instigate chronic inflammation, produce genotoxic metabolites, or directly manipulate host cell processes to drive malignant transformation. The Johns Hopkins research adds breast cancer to this growing list, solidifying the importance of considering microbial factors in oncology. This chronological progression of understanding, from simple observation to detailed molecular mechanism, underscores a paradigm shift in medical research, recognizing the intricate interplay between our resident microbes and our health.
Leading cancer research institutions and public health organizations are increasingly acknowledging the profound impact of the microbiome. While direct clinical recommendations are still under development, the scientific community widely agrees that maintaining a healthy microbiome, including oral hygiene, is a crucial component of overall well-being and disease prevention. This study provides powerful, actionable evidence in that direction, moving the conversation from general wellness to specific cancer risk reduction.
Profound Implications for Breast Cancer Prevention and Treatment
The findings from the Johns Hopkins Kimmel Cancer Center carry profound implications across the spectrum of breast cancer management, from primary prevention to advanced therapeutic strategies.
Redefining Risk Factors and Proactive Strategies: The most immediate and tangible implication relates to breast cancer prevention. If F. nucleatum is indeed a causal or co-causal agent, then maintaining excellent oral hygiene and effectively treating periodontal disease could emerge as a surprisingly potent, yet accessible, strategy for reducing breast cancer risk. This is particularly relevant for individuals already at higher risk due to genetic predispositions, such as BRCA1 mutations. Public health campaigns could evolve to include oral health as a critical component of cancer prevention, alongside established recommendations like diet, exercise, and avoiding smoking. This could empower individuals with a new, actionable method to proactively manage their cancer risk, transforming a routine aspect of daily life into a potentially life-saving intervention. Further research will be crucial to establish the precise extent to which improving oral health impacts breast cancer incidence in large human cohorts.
Paving the Way for Novel Therapeutic Avenues: Beyond prevention, the study opens doors for innovative diagnostic and therapeutic approaches. If F. nucleatum is a driver of tumor aggressiveness and chemoresistance, especially through the activation of proteins like PKcs, then targeting this bacterium or its associated pathways could become a novel therapeutic strategy. This could involve:
- Antibiotic interventions: Specific antibiotics targeting F. nucleatum could be explored as an adjunct therapy for breast cancer patients, particularly those with aggressive tumors or BRCA1 mutations.
- Microbiome modulation: Strategies to alter the overall oral or gut microbiome to reduce the prevalence or impact of F. nucleatum could be investigated.
- Targeted drug development: The identified molecular pathways, such as the activity of PKcs, could become targets for new drug development aimed at disrupting the bacterium’s ability to promote DNA damage, accelerate growth, or induce chemoresistance. This could lead to more personalized and effective treatments for patients.
- Enhanced screening: The presence of F. nucleatum in breast tissue or specific systemic markers could potentially serve as novel biomarkers for early detection or for identifying patients at higher risk for aggressive disease.
The researchers emphasize that while the findings are compelling, more extensive work is needed to fully understand how these discoveries translate directly into routine patient care. Future studies will be pivotal in exploring whether interventions aimed at maintaining good oral health can demonstrably play a significant role in reducing breast cancer incidence and improving outcomes in human populations. These could include large-scale clinical trials investigating the impact of periodontal treatment on breast cancer recurrence or development.
The collaborative spirit of this research is evident in its multidisciplinary team. In addition to Dr. Sharma, the research team included Sheetal Parida, Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson, and Cynthia Sears. This extensive roster underscores the complexity and rigor required for such a pioneering study, drawing on expertise across microbiology, oncology, genetics, and pathology. The study received crucial financial backing from prestigious organizations including the Breast Cancer Research Foundation, Congressionally Directed Medical Research Programs Department of Defense Breast Cancer Research Program grants (BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention, and the Bloomberg~Kimmel Institute for Cancer Immunotherapy. Such robust support highlights the recognized potential impact of this research on public health and the ongoing fight against breast cancer. The findings not only offer a fresh perspective on breast cancer etiology but also pave the way for a future where oral hygiene might be as integral to cancer prevention as lifestyle choices.