Ovarian cancer remains the most lethal of all gynecological malignancies, a reality driven primarily by its propensity for late-stage discovery and its aggressive, unconventional mode of spread. For decades, the medical community has grappled with the "silent" nature of the disease, which often remains asymptomatic until it has metastasized extensively throughout the peritoneal cavity. While the speed of this progression has been well-documented in clinical settings, the underlying biological mechanisms facilitating such rapid dissemination have remained largely enigmatic. However, a groundbreaking study led by researchers at Nagoya University in Japan has finally identified a sophisticated "hijacking" mechanism that explains how these cancer cells navigate the abdominal environment with such lethal efficiency.
The research, recently published in the prestigious journal Science Advances, demonstrates that ovarian cancer cells do not operate in isolation. Instead, they actively subvert the body’s own defensive structures to facilitate their journey. By recruiting mesothelial cells—the protective lining of the internal organs and abdominal wall—the cancer cells create hybrid "super-clusters" that act as invasive vanguard units. These partnerships not only accelerate the physical invasion of healthy tissue but also provide a biological shield that renders the cancer significantly more resistant to standard chemotherapy protocols.
The Peritoneal Cavity: A Unique Landscape for Metastasis
To understand the significance of the Nagoya University findings, one must first understand the unique environment in which ovarian cancer thrives. Unlike many other common cancers, such as breast, lung, or prostate cancer, which typically spread through the lymphatic system or the bloodstream (hematogenous metastasis), ovarian cancer utilizes a process known as transcoelomic spread.
In the early stages of the disease, malignant cells detach from the primary tumor on the ovary or the fallopian tube. These cells then enter the peritoneal fluid—the lubricating liquid found within the abdominal cavity. Under normal physiological conditions, this fluid is in constant motion, driven by the rhythmic movements of the diaphragm during breathing and the peristaltic contractions of the digestive tract. This constant circulation acts as a conveyor belt, carrying detached cancer cells to distant sites within the abdomen, including the liver, the omentum, and the intestines.
Historically, scientists believed these floating cancer cells were passive travelers, eventually landing on an organ surface and beginning the arduous process of invasion. The new data, however, suggests a much more active and collaborative process occurring within the fluid itself.
The Discovery of Hybrid Cell Clusters
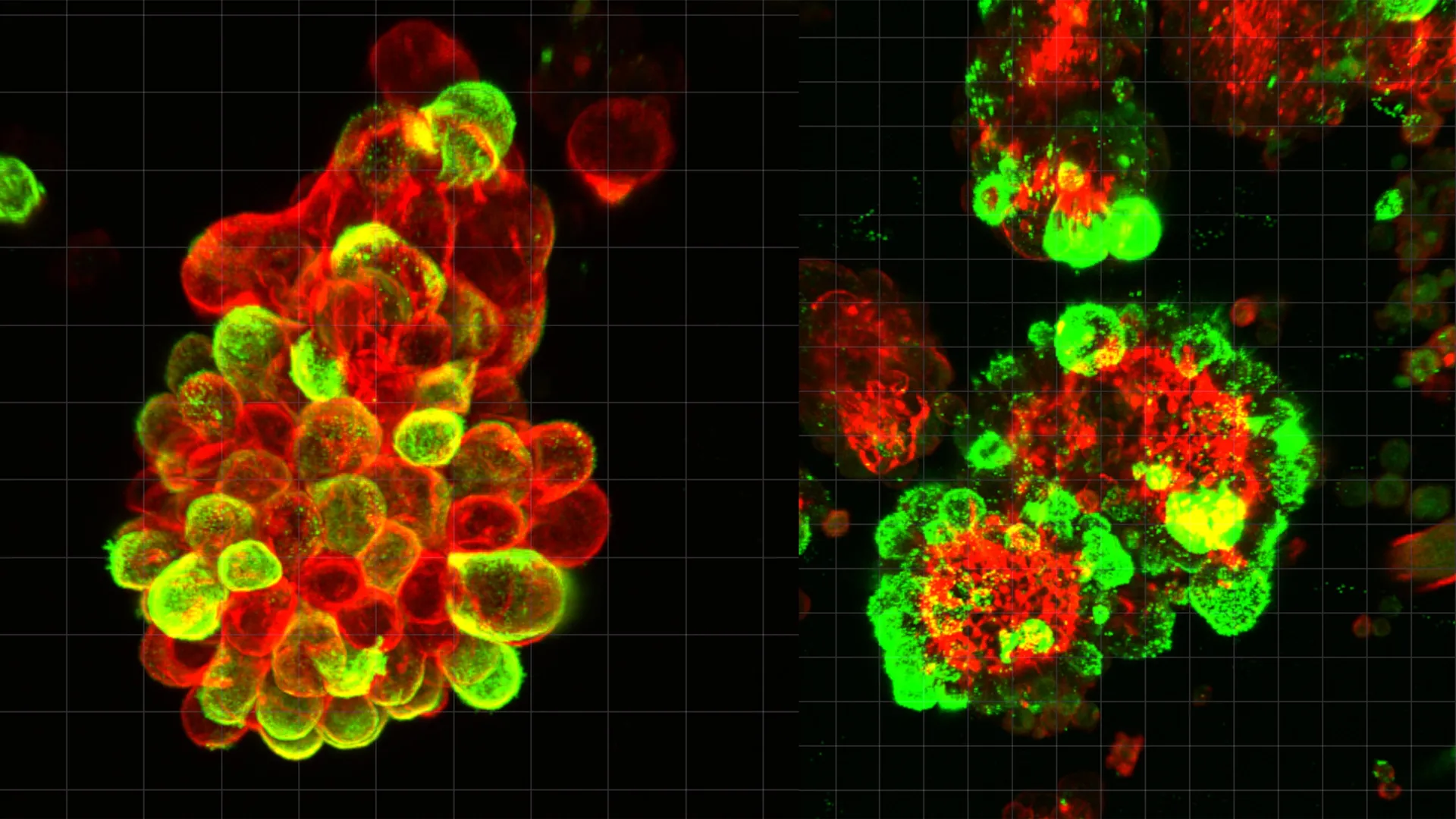
The research team, led by Dr. Kaname Uno of the Nagoya University Graduate School of Medicine, began their investigation by analyzing abdominal fluid (ascites) collected from patients diagnosed with advanced ovarian cancer. Using high-resolution microscopy and single-cell analysis, they discovered that the cancer cells were rarely drifting as solitary units. Instead, they were found in compact, spherical clusters composed of both malignant cells and "hijacked" mesothelial cells.
The study estimated that approximately 60% of the cancer cell spheres observed in the abdominal fluid were these hybrid structures. This finding challenged the long-standing assumption that mesothelial cells were merely passive victims of cancer. Under normal circumstances, the mesothelium acts as a frictionless, protective barrier that prevents organs from adhering to one another and provides a first line of defense against pathogens. The Nagoya University study reveals that ovarian cancer cells effectively turn these defenders into accomplices.
The mechanism of this transformation is rooted in molecular signaling. The researchers identified that ovarian cancer cells secrete a specific signaling protein known as Transforming Growth Factor-beta 1 (TGF-β1). When mesothelial cells, which have naturally shed into the abdominal fluid, come into contact with this protein, they undergo a fundamental change in their biological properties. This transformation causes the mesothelial cells to develop "invadopodia"—sharp, spike-like cellular protrusions designed to penetrate the extracellular matrix of healthy tissue.
Invadopodia: The Biological Drills of Metastasis
The formation of invadopodia is the critical turning point in the metastatic process. In the hybrid clusters, the mesothelial cells position themselves at the leading edge of the sphere. As the cluster encounters the surface of an abdominal organ, the mesothelial cells use their invadopodia to "drill" through the protective lining of the organ.
"The cancer cells themselves remain relatively unchanged in their invasive capacity," explained Dr. Uno. "Instead, they manipulate the mesothelial cells to do the heavy lifting of tissue invasion. The cancer cells undergo minimal genetic or molecular changes during this phase; they simply follow the pathways and openings that the mesothelial cells create for them."
This "outsourcing" of the invasion process allows the cancer to spread with remarkable speed. Because the cancer cells do not need to undergo the complex genetic mutations typically required for independent invasion (a process known as the epithelial-to-mesenchymal transition, or EMT), they can begin colonizing new sites almost immediately upon arrival. Furthermore, the researchers found that these hybrid spheres are significantly more resilient. When exposed to common chemotherapy agents, the hybrid clusters showed a much higher survival rate than clusters consisting solely of cancer cells, suggesting that the mesothelial component provides a protective microenvironment for the malignancy.
A Physician’s Motivation: The Chronology of Research
The impetus for this research was deeply personal for the lead author. Before transitioning into full-time molecular research, Dr. Kaname Uno spent eight years practicing as a gynecologist. During his clinical tenure, he encountered a patient whose case would redefine his career trajectory.
The patient had undergone a routine gynecological screening and received a completely clear set of results. However, only three months later, she returned to the clinic with severe abdominal symptoms. Upon investigation, doctors found that she had progressed to advanced-stage ovarian cancer that had already spread throughout her abdominal cavity. Despite the best available treatments, the disease was too far advanced to be contained.
"The fact that our existing diagnostic tools and understanding of the disease failed to catch the cancer in such a short window was devastating," Dr. Uno recalled. This experience drove him to leave clinical practice and join the Graduate School of Medicine at Nagoya University, where he sought to uncover the biological reasons for such rapid, undetectable progression.
The study followed a rigorous multi-year chronology:
- Clinical Observation: Analysis of ascites from human patients to identify the presence of hybrid clusters.
- Molecular Mapping: Utilizing single-cell RNA sequencing to identify the TGF-β1 signaling pathway.
- In Vitro Modeling: Recreating the hybrid spheres in a laboratory setting to observe the formation of invadopodia.
- In Vivo Validation: Using mouse models to confirm that hybrid clusters lead to faster and more widespread metastasis compared to pure cancer cell clusters.
- Microscopic Real-Time Tracking: Using advanced imaging to watch the "drilling" process as it occurred.
Implications for Future Treatment and Diagnostics
The discovery of the mesothelial-cancer cell partnership opens several new frontiers for the treatment of ovarian cancer, which has seen relatively few major breakthroughs in survival rates over the last two decades.
Currently, the standard of care involves cytoreductive surgery followed by platinum-based chemotherapy. While this is often effective at shrinking the primary tumor and killing many circulating cancer cells, it does not specifically target the signaling mechanisms that allow hybrid clusters to form. By identifying TGF-β1 as the primary catalyst for this partnership, the Nagoya University study points toward the potential use of TGF-β inhibitors as a supplemental therapy. If the signaling can be blocked, the cancer cells would be unable to recruit their "traveling companions," significantly slowing the rate of spread and making the cells more vulnerable to traditional chemotherapy.
Furthermore, the study suggests a shift in how the disease might be monitored. "Liquid biopsies"—tests that look for cancer markers in bodily fluids—have traditionally focused on blood. However, since ovarian cancer cells primarily travel through the peritoneal fluid and avoid the bloodstream, blood-based tests like the CA-125 marker are often unreliable for early detection. The researchers propose that monitoring the presence and concentration of these hybrid cell clusters in the abdominal fluid could provide a much more accurate prediction of disease progression and a patient’s likely response to treatment.
Broader Impact on Oncology
The findings from Nagoya University contribute to a growing body of evidence suggesting that the "microenvironment" of a tumor—the non-cancerous cells and fluids surrounding it—is just as important as the cancer cells themselves. This study highlights a form of "collective invasion" that may be present in other types of cancer that spread through body cavities, such as mesothelioma or certain types of gastric and pancreatic cancers.
As the global medical community shifts toward personalized and targeted medicine, understanding these specific cellular interactions is vital. If clinicians can disrupt the "social network" of a tumor, they may be able to turn a rapidly progressing, terminal disease into a manageable condition.
The research conducted by Dr. Uno and his colleagues at Nagoya University serves as a critical bridge between clinical observation and molecular discovery. By answering the question of why ovarian cancer moves so quickly, they have provided the foundation for a new generation of therapies designed not just to kill cancer cells, but to dismantle the very infrastructure of their survival. For the thousands of women diagnosed with ovarian cancer each year, this insight into the "silent killer’s" tactics offers a newfound sense of hope for earlier intervention and more effective long-term treatment.