The quest for more effective cancer therapies is perpetually hampered by a fundamental challenge: the unpredictable response of patients to the same treatment. A groundbreaking study, published in the prestigious journal Nature Communications, has illuminated a critical factor underlying this variability, particularly concerning PARP inhibitors—a vital class of targeted drugs. Led by Dr. Louise Fets at the MRC Laboratory of Medical Sciences (LMS), the research meticulously tracked the journey of these potent drugs within ovarian tumor samples, employing sophisticated imaging techniques to unveil a hidden mechanism of drug accumulation. The findings reveal that lysosomes, the cell’s internal recycling centers, act as unexpected reservoirs, trapping and subsequently releasing PARP inhibitors, profoundly influencing their therapeutic impact.
The Uneven Landscape of Cancer Drug Distribution
In recent years, the landscape of cancer treatment has undergone a dramatic transformation, with targeted therapies offering renewed hope and improved prognoses for countless individuals. PARP inhibitors, in particular, have revolutionized the management of ovarian cancer, demonstrating remarkable efficacy in a significant subset of patients. However, the persistent reality of non-response and the eventual development of drug resistance remain significant clinical hurdles. For these targeted agents to exert their life-saving effects, they must achieve a critical concentration within cancer cells, initiating the cascade of events that leads to tumor cell death. Despite this fundamental requirement, the precise mechanisms governing drug distribution within the complex architecture of tumors, and crucially, within individual cancer cells, have remained largely elusive.
This pioneering research challenges the simplistic notion that drug effectiveness is solely contingent upon reaching the tumor. Instead, it underscores the vital importance of how these molecules disseminate within the tumor microenvironment and, more granularly, inside individual malignant cells. To probe this intricate process, the research team ingeniously utilized patient-derived ovarian tumor samples, carefully preserved and maintained in a viable state ex vivo. These meticulously prepared "explants" served as living laboratories, allowing scientists to directly administer PARP inhibitors and observe, in real-time, the drugs’ intricate movements through authentic human tumor tissue.
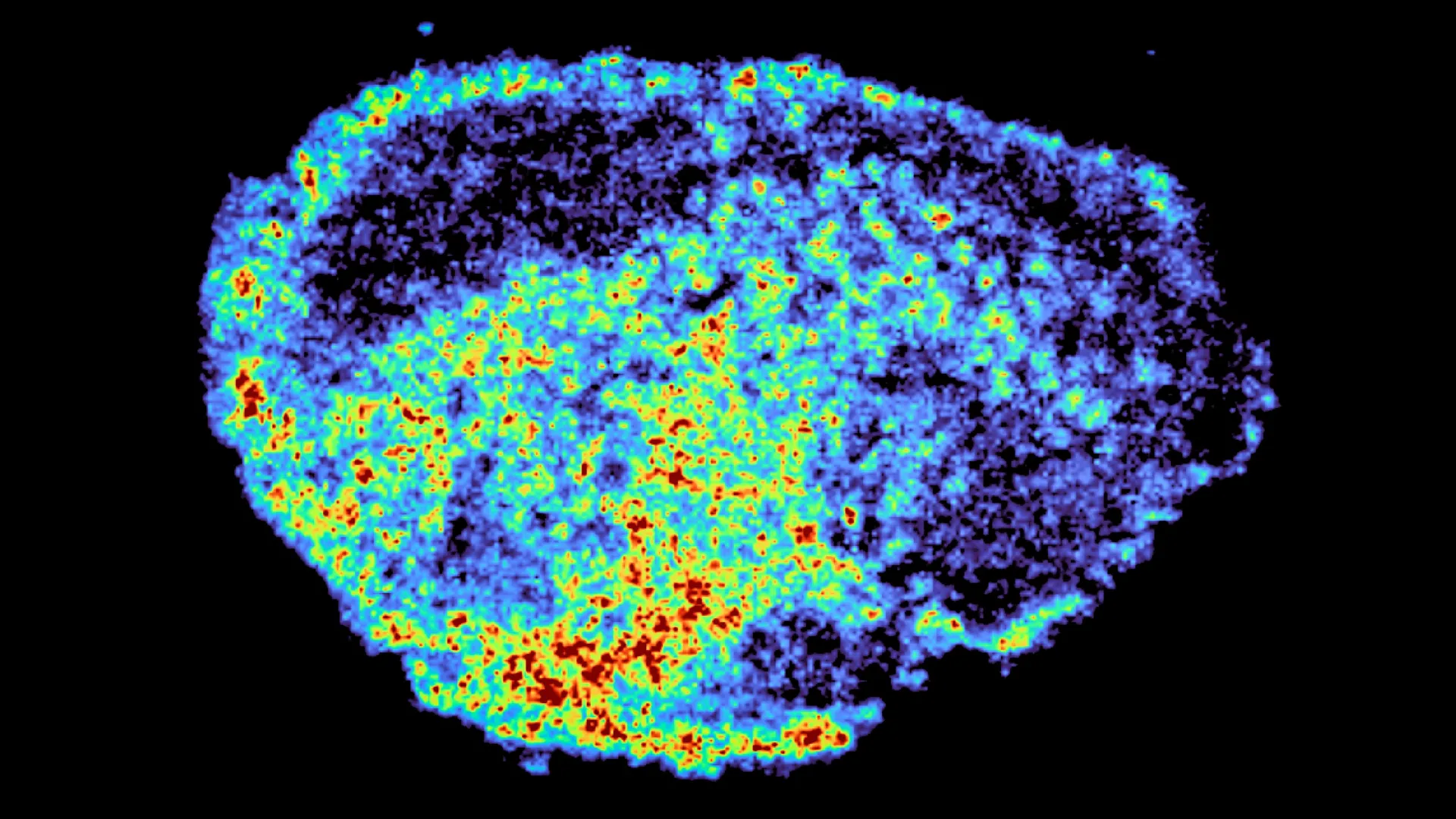
The study’s methodological sophistication was paramount. By integrating mass spectrometry imaging, the researchers were able to generate highly detailed, spatially resolved maps that pinpointed precisely where the PARP inhibitors accumulated within the tumor samples. This powerful imaging technique was then synergistically combined with spatial transcriptomics, a revolutionary technology that enables the examination of gene activity within specific anatomical locations on the same tissue slice. This dual approach allowed scientists to correlate drug concentration with cellular function, revealing striking disparities in drug distribution. These variations were not only evident across different tumors from distinct patients but also manifested significantly within individual tumor samples, even when the same drug dosage was applied.
"A novel aspect of this study was the use of mass spectrometry imaging to directly measure and visualize drug uptake in patient tumour tissue," stated Dr. Zoe Hall, a senior author of the study and Associate Professor at Imperial College London’s Department of Metabolism, Digestion and Reproduction. "Through the spatial mapping of drug molecules, we could pinpoint regions of high and low drug and compare gene expression, from the same tissue slice, using spatial transcriptomics." This integrated approach provided an unprecedented granular view of drug behavior within the complex tumor milieu.
Lysosomes: Unveiling the Hidden Drug Reservoirs
The central revelation of the study lies in the pivotal role played by lysosomes in orchestrating this uneven drug distribution. The research demonstrates that certain PARP inhibitors exhibit a pronounced affinity for these intracellular organelles, leading to their sequestration within lysosomes rather than their even dispersal throughout the cell. This phenomenon effectively creates internal pockets of drug concentration, akin to hidden storage depots.
These lysosomes, therefore, function as dynamic, slow-release reservoirs. By retaining the drug and gradually releasing it, they create localized areas of heightened drug exposure for some cancer cells, while leaving other cells within the same tumor exposed to significantly lower concentrations. Crucially, the study elucidated that this lysosomal sequestration is not a universal behavior among all PARP inhibitors. The findings indicate that specific drugs, such as rucaparib and niraparib, are demonstrably affected by this lysosomal trapping mechanism. In contrast, other PARP inhibitors, exemplified by olaparib, appear to bypass this process, suggesting a distinct intracellular fate.
"We were surprised to see large variability in drug accumulation at the single-cell level," commented Dr. Carmen Ramirez Moncayo, the study’s first author and a Postdoctoral Researcher at the LMS. "This variability was driven by the build-up of a drug in lysosomes, which are acting as reservoirs, increasing the exposure of cancer cells to drugs, by storing and releasing the drug when needed." This observation underscores the intricate cellular choreography that dictates drug efficacy and highlights the potential for therapeutic intervention at the lysosomal level.
Implications for the Future of Personalized Cancer Therapy
The implications of these findings for the future of cancer treatment are profound. PARP inhibitors are currently a cornerstone of therapy for a range of cancers, including ovarian, breast, and prostate malignancies, and their therapeutic potential is actively being explored in numerous other cancer types. A deeper understanding of how these critical drugs are internalized, stored, and subsequently released within cancer cells has the potential to usher in an era of truly personalized treatment strategies. By tailoring therapeutic approaches based on an individual patient’s cellular drug handling mechanisms, clinicians may be able to significantly enhance treatment effectiveness, mitigate the development of drug resistance, and ultimately reduce the incidence of cancer relapse.
"By understanding how drugs are taken up into cells, we can understand whether this influences why cancer drugs work for some people and not for others," explained Dr. Louise Fets, a senior author and Head of the LMS’ Drug Transport and Tumour Metabolism Group. "Eventually, we hope to be able study the molecular signature of a patient’s tumor to help to tailor therapeutic approaches in a more personalized way." This vision of personalized medicine, informed by detailed cellular-level insights, represents a significant leap forward in the fight against cancer.
It is important to acknowledge the context of the current study. The research was conducted using tumor tissue maintained outside the body, a powerful experimental model that allows for precise control and observation. In the clinical setting, however, drug delivery occurs via the bloodstream, and the inherent disorganization of tumor vasculature in many cancers can introduce further complexities and contribute to uneven drug distribution. Future research endeavors will undoubtedly build upon these foundational discoveries by employing animal models and larger patient cohorts. These subsequent studies will aim to comprehensively unravel the intricate interplay between drug delivery dynamics, tumor structural heterogeneity, and lysosomal sequestration mechanisms within the complex and dynamic milieu of living patients, including those with relapsed cancers.
The research was generously supported by funding from the Medical Research Council, Cancer Research UK, a PhD studentship from the Integrative Toxicology Training Partnership administered by the MRC Toxicology Unit, and a Victoria’s Secret Global Fund for Women’s Cancers Career Development Award, in partnership with Pelotonia and AACR. This collaborative effort underscores the multidisciplinary and multi-institutional commitment required to tackle the complex challenges in cancer research.
A Timeline of Discovery and Future Directions
The journey leading to this significant discovery can be broadly contextualized within the ongoing evolution of cancer therapy.
- Early 2000s: The concept of targeting DNA repair pathways in cancer gains traction, paving the way for the development of PARP inhibitors.
- Mid-2000s: Initial clinical trials demonstrate promising efficacy for PARP inhibitors, particularly in ovarian cancers with BRCA mutations.
- 2010s: PARP inhibitors receive regulatory approval for specific cancer types, marking a significant advancement in targeted therapy. Clinical observations of differential patient responses begin to fuel the need for deeper mechanistic understanding.
- Late 2010s – Early 2020s: Advancements in imaging technologies, such as mass spectrometry imaging, and spatial transcriptomics become more accessible and sophisticated, enabling researchers to investigate drug distribution at an unprecedented level of detail.
- Present Study (Published in Nature Communications): Dr. Fets’ team at the LMS leverages these advanced technologies to investigate PARP inhibitor uptake in patient-derived tumor explants, identifying lysosomes as key determinants of drug distribution and efficacy.
Looking ahead, the implications of this research are far-reaching. The ability to predict which patients are likely to benefit most from specific PARP inhibitors, based on their tumor’s lysosomal handling of the drugs, could transform clinical decision-making. Furthermore, this knowledge could spur the development of novel drug formulations or combination therapies designed to overcome lysosomal sequestration and enhance drug delivery to all cancer cells within a tumor. The ultimate goal is to move beyond broad therapeutic strategies and embrace a future where cancer treatment is precisely tailored to the unique biological characteristics of each patient’s disease, maximizing efficacy while minimizing toxicity and the specter of recurrence. This study represents a critical step in that ongoing and vital endeavor.