Ovarian cancer has long held a reputation as the "silent killer" of the gynecological world, characterized by its ability to spread extensively throughout the abdominal cavity before the onset of noticeable symptoms. For decades, the oncology community has grappled with the biological mechanics behind this rapid dissemination, which often renders the disease incurable by the time of diagnosis. However, a landmark study led by a research team at Nagoya University in Japan has finally decoded the cellular conspiracy driving this process. Published in the prestigious journal Science Advances, the research reveals that ovarian cancer cells do not act as solitary invaders. Instead, they hijack healthy mesothelial cells to form aggressive hybrid clusters that serve as specialized "vessels" for metastasis and shields against chemotherapy.
The discovery shifts the fundamental understanding of how ovarian cancer moves through the human body. Unlike many common malignancies that utilize the bloodstream or lymphatic system as a highway to distant organs, ovarian cancer predominantly employs transcoelomic spread—a process where cells detach from the primary tumor and drift through the peritoneal fluid. The Nagoya University study demonstrates that during this floating phase, cancer cells actively recruit mesothelial cells, which normally form a protective, friction-reducing lining for the internal organs. Once joined, these two cell types create a synergistic partnership that is significantly more lethal than either cell type alone.
The Mechanics of Cellular Hijacking and the Role of TGF-β1
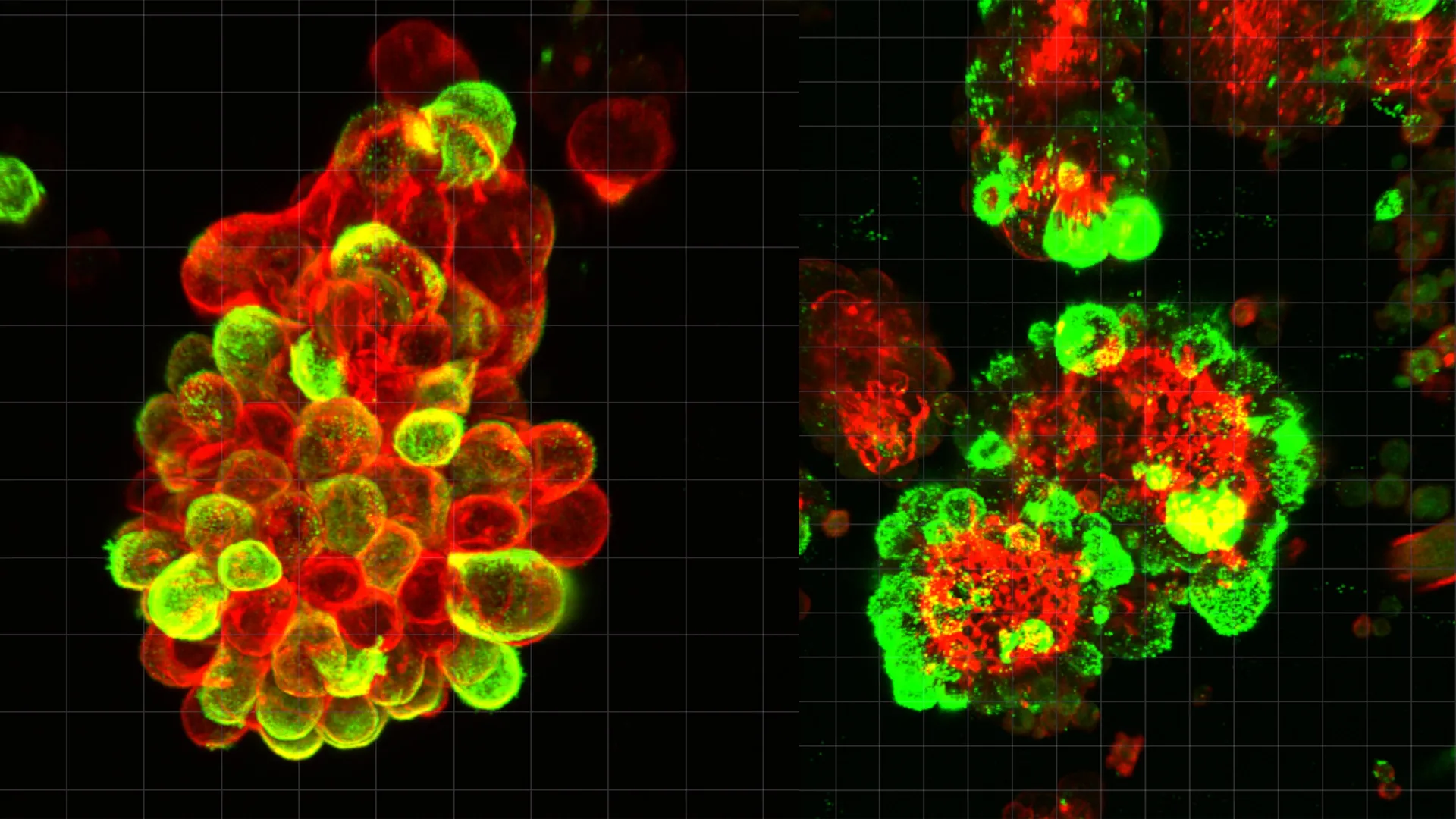
The research team, led by Dr. Kaname Uno of the Nagoya University Graduate School of Medicine, focused their investigation on the "floating stage" of cancer progression. By analyzing abdominal fluid—clinically referred to as ascites—from human patients, the researchers observed that cancer cells were rarely isolated. Instead, they were encased within compact, spherical structures alongside mesothelial cells. The study estimated that approximately 60% of all malignant clusters found in the abdominal fluid were these hybrid formations.
The catalyst for this partnership is a signaling molecule known as Transforming Growth Factor-beta 1 (TGF-β1). The study found that ovarian cancer cells secrete high concentrations of TGF-β1, which acts as a molecular "reprogramming" agent. When mesothelial cells, which have naturally shed from the abdominal lining, come into contact with this protein, they undergo a dramatic transformation. They lose their protective function and instead develop invadopodia—sharp, spike-like protrusions composed of actin filaments.
These invadopodia act as biological drill bits. When a hybrid cluster settles on the surface of an organ, such as the liver, diaphragm, or omentum, it is the recruited mesothelial cells, not the cancer cells themselves, that lead the invasion. The mesothelial spikes puncture the basement membrane of the target tissue, creating physical openings. The cancer cells then follow through these newly created pathways, colonizing the organ with remarkable efficiency.
A Chronology of Discovery: From the Clinic to the Laboratory
The impetus for this research was rooted in clinical frustration. Before entering the field of molecular research, Dr. Kaname Uno served as a gynecologist for eight years. During his tenure, he encountered a case that would define his scientific career. A female patient under his care had undergone routine gynecological screenings that returned entirely normal results. However, only three months later, she presented with advanced, Stage IV ovarian cancer that had already colonized her entire abdominal cavity.
"The speed of the progression was devastating," Dr. Uno recalled during the presentation of his findings. "Our existing diagnostic tools and our understanding of the disease’s timeline were insufficient to save her. I needed to understand why this specific cancer could bypass our defenses so quickly."
This clinical observation led Dr. Uno to pursue a PhD at Nagoya University, where he utilized advanced microscopy and single-cell RNA sequencing to track the behavior of cells within the peritoneal environment. The research team employed mouse models to validate their findings, injecting human-derived cancer cells into the abdominal cavities of the subjects. They observed that the presence of mesothelial cells within the clusters directly correlated with the speed of organ invasion and the overall tumor burden.
Supporting Data: Survival Rates and the Failure of Traditional Chemotherapy
The implications of this study are underscored by the sobering statistics surrounding ovarian cancer. According to the American Cancer Society, the five-year survival rate for ovarian cancer is significantly lower than that of breast cancer, largely due to late-stage detection. When the disease is caught in Stage I, the survival rate exceeds 90%. However, only about 15% of cases are diagnosed at this stage. For the majority of patients diagnosed at Stage III or IV, the survival rate drops to approximately 30%.
One of the most critical findings of the Nagoya University study concerns chemotherapy resistance. Standard treatment for ovarian cancer typically involves a combination of cytoreductive surgery and platinum-based chemotherapy, such as carboplatin and paclitaxel. However, the researchers found that the hybrid clusters were significantly more resilient to these drugs than solitary cancer cells.
The mesothelial cells within the cluster appear to provide a protective niche, potentially altering the microenvironment to neutralize the effects of cytotoxic drugs. This explains why many patients initially respond well to chemotherapy—which kills off the vulnerable, solitary cancer cells—only to experience a rapid recurrence driven by the survival of these hardy hybrid spheres. By "outsourcing" the hard work of tissue invasion and environmental protection to mesothelial cells, the cancer cells remain genetically stable and shielded from the stressors that would otherwise trigger cell death.
Comparative Analysis: Transcoelomic vs. Hematogenous Spread
To appreciate the significance of this discovery, one must contrast ovarian cancer with other major malignancies. In breast, lung, or prostate cancer, metastasis typically occurs through the blood (hematogenous spread). Cancer cells must undergo a complex process called the Epithelial-Mesenchymal Transition (EMT) to survive the high-pressure environment of the bloodstream and eventually exit through vessel walls to reach new organs.
In contrast, ovarian cancer cells bypass the circulatory system. The peritoneal cavity is a fluid-filled space where organs are suspended. The fluid is in a constant state of flux, driven by the rhythmic movement of the diaphragm during breathing and the peristaltic motion of the intestines. This movement acts as a natural dispersal system, carrying the hybrid clusters to every corner of the abdomen. Because this fluid does not follow the fixed, predictable pathways of blood vessels, tracking the spread of the disease through conventional blood tests (liquid biopsies) has historically been difficult. The Nagoya University study suggests that the "floating phase" is not a passive period of drifting but a highly active stage of recruitment and preparation.
Future Implications for Treatment and Diagnostic Monitoring
The identification of the TGF-β1 signaling pathway and the role of invadopodia provides the medical community with concrete new targets for therapeutic intervention. Current treatments are almost exclusively focused on the cancer cells themselves. This research suggests that a dual-target approach may be necessary.
Potential future therapies could involve:
- TGF-β1 Inhibitors: By blocking the signal that cancer cells send to mesothelial cells, doctors might prevent the formation of the hybrid clusters entirely, keeping the cancer cells in a less invasive, solitary state.
- Invadopodia Disruption: Drugs that interfere with the formation of the actin-based spikes in mesothelial cells could prevent the clusters from "drilling" into healthy organs.
- Ascites Monitoring: Instead of relying solely on blood-based biomarkers like CA-125, which can be unreliable, doctors could monitor the abdominal fluid for the presence of these hybrid spheres. The concentration and composition of these clusters could serve as a real-time indicator of how a patient is responding to treatment or how aggressively the disease is progressing.
Conclusion and Expert Reaction
The oncology community has reacted to the Nagoya University study with cautious optimism. Independent researchers note that while the study was conducted using a combination of patient samples and animal models, the biological mechanism is robust and explains many of the clinical "blind spots" associated with ovarian cancer.
The study concludes that the "intelligence" of ovarian cancer lies in its ability to manipulate its environment rather than relying solely on its own genetic mutations. By turning the body’s protective lining into a weapon of invasion, the cancer achieves a level of efficiency that has long baffled physicians. As Dr. Uno and his team continue their work, the focus shifts toward translating these laboratory insights into clinical trials. For the thousands of women diagnosed with ovarian cancer each year, this research offers the first clear roadmap for stopping the "silent killer" before it can complete its deadly journey through the abdomen.