The biological landscape of gynecological oncology has long been haunted by the aggressive and elusive nature of ovarian cancer. Frequently referred to as the "silent killer," this malignancy remains the deadliest of all cancers affecting the female reproductive system. The primary reason for its high mortality rate is the speed and stealth with which it colonizes the abdominal cavity, often remaining undetected until it has reached an advanced stage. For decades, the scientific community has struggled to understand why ovarian cancer moves with such devastating efficiency. However, a groundbreaking study led by researchers at Nagoya University, recently published in the journal Science Advances, has finally provided a definitive explanation for this rapid progression.
The research reveals that ovarian cancer cells do not act as solitary invaders. Instead, they employ a sophisticated strategy of cellular recruitment, hijacking healthy mesothelial cells—the protective lining of the internal organs and abdominal cavity—to serve as "scouts" and "engineers." These hijacked cells lead the charge, physically carving paths through healthy tissue that the cancer cells then exploit. This symbiotic relationship results in the formation of hybrid cell clusters that are not only highly mobile but also significantly more resistant to conventional chemotherapy than independent cancer cells.
The Discovery of Cellular Collaboration in the Peritoneal Cavity
To uncover the mechanics of this spread, the research team, led by Dr. Kaname Uno, meticulously analyzed abdominal fluid (ascites) collected from patients diagnosed with ovarian cancer. Historically, it was believed that cancer cells drifted through this fluid as individual entities or small, homogenous clumps. The Nagoya University study challenged this assumption by utilizing high-resolution microscopy and single-cell genetic sequencing.
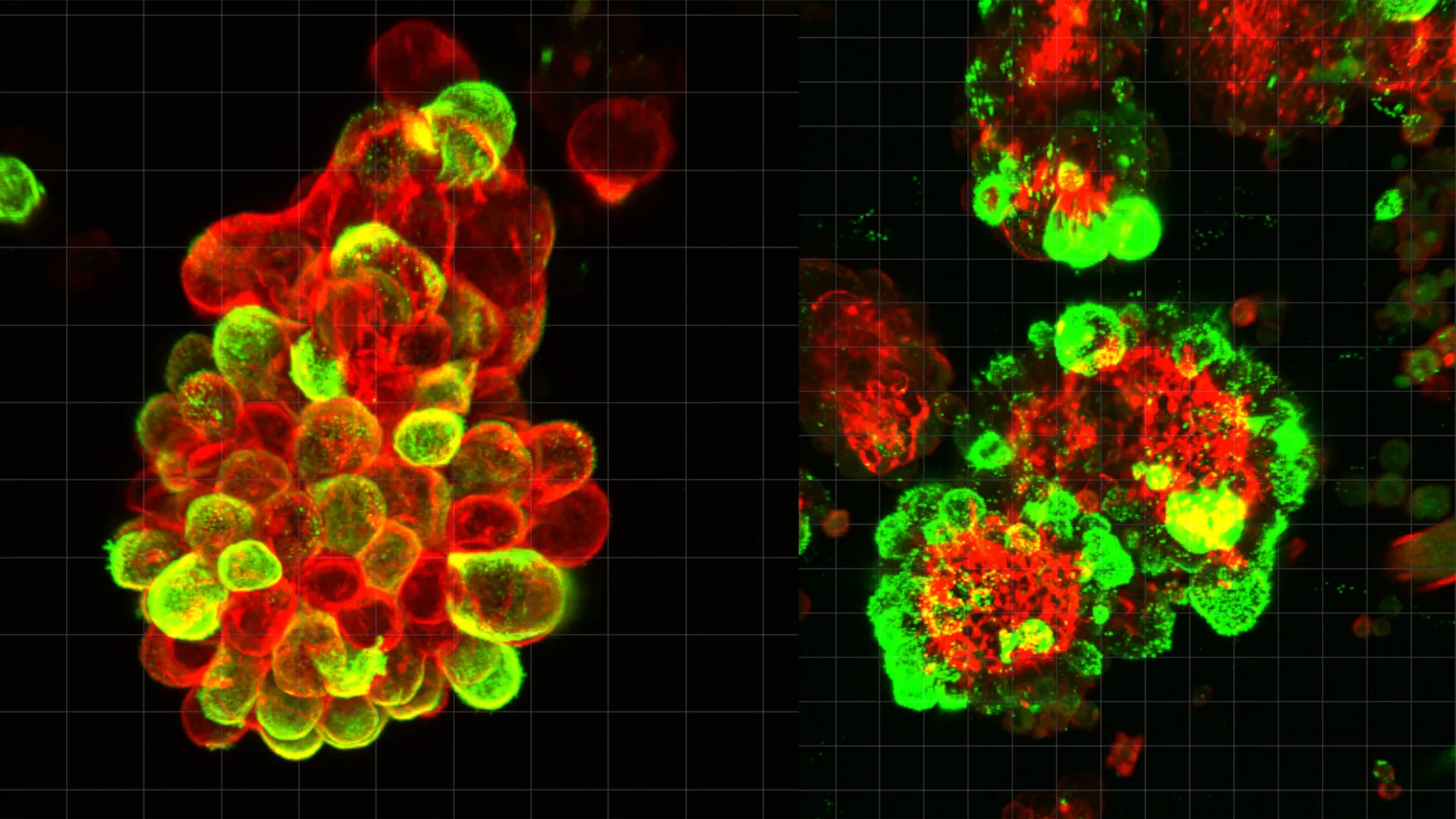
The team discovered that the cancer cells were frequently attached to mesothelial cells, forming compact, spherical hybrid clusters. Statistical analysis indicated that approximately 60% of the cancer cell spheres observed in the abdominal fluid were these mixed-cell hybrids. The presence of these mesothelial cells was not accidental; the cancer cells actively manipulated them through chemical signaling. Specifically, the cancer cells secrete a potent signaling protein known as Transforming Growth Factor-beta 1 (TGF-β1).
When mesothelial cells—which are naturally designed to provide a smooth, frictionless surface for internal organs—come into contact with TGF-β1, they undergo a radical transformation. They lose their protective characteristics and develop "invadopodia." These are sharp, spike-like protrusions rich in actin filaments that function like biological drill bits. These structures allow the hybrid clusters to penetrate the basement membranes of abdominal organs with far greater force than a lone cancer cell could generate.
A Departure from Traditional Metastatic Models
The spread of ovarian cancer represents a unique challenge in oncology because it deviates significantly from the metastatic pathways of more common cancers, such as breast or lung cancer. In those diseases, the primary tumor typically sheds cells into the lymphatic system or the bloodstream. This hematogenous spread follows the vascular "highways" of the body, which, while dangerous, provides doctors with a somewhat predictable map for monitoring and a medium—the blood—that can be tested for biomarkers.
Ovarian cancer, by contrast, utilizes the fluid-filled space of the peritoneal cavity. Because the abdominal fluid is in constant motion—driven by the diaphragm during respiration, the beating of the heart, and the peristaltic movements of the digestive tract—it acts as a chaotic transport system. Cancer cells that detach from the primary tumor are swept away to various "landing zones" throughout the abdomen, including the liver, intestines, and diaphragm.
Until the publication of this study, the "floating phase" of these cells was poorly understood. Scientists were unsure how these drifting cells maintained their viability and how they managed to adhere to and invade new organs so quickly once they landed. The discovery of the hybrid spheres explains this efficiency: the cancer cells bring their own "invasion machinery" with them in the form of recruited mesothelial cells.
The Role of Invadopodia and Chemotherapy Resistance
The formation of these hybrid clusters does more than just facilitate movement; it provides a survival advantage. The study found that when cancer cells are bundled with mesothelial cells, they exhibit a marked increase in resistance to standard chemotherapy drugs, such as carboplatin and paclitaxel.
The mesothelial cells appear to create a protective microenvironment within the sphere, shielding the inner cancer cells from the full impact of cytotoxic agents. Furthermore, the invadopodia produced by the mesothelial cells ensure that the moment a cluster makes contact with an organ, the invasion process begins almost instantaneously. This "outsourcing" of labor allows the cancer cells to remain in a relatively stable state, conserving energy and maintaining their genetic integrity while the mesothelial cells do the heavy lifting of tissue penetration.
Dr. Kaname Uno, a Visiting Researcher at Nagoya University’s Graduate School of Medicine and the study’s lead author, noted that the cancer cells themselves undergo minimal genetic changes during this process. "They manipulate mesothelial cells to do the tissue invasion work," Dr. Uno explained. "The cancer cells just migrate through the openings that the mesothelial cells create, allowing the malignancy to spread without needing to evolve complex new invasive traits on their own."
Methodology: From Clinical Experience to Molecular Analysis
The origins of this research are deeply rooted in clinical practice. Before transitioning into full-time research, Dr. Uno spent eight years as a practicing gynecologist. His decision to investigate the rapid spread of ovarian cancer was fueled by a specific clinical tragedy. He treated a patient who had received a completely normal gynecological screening, only to be diagnosed with advanced, stage IV ovarian cancer just three months later.
The speed with which the disease had progressed—moving from undetectable to terminal in 90 days—exposed the limitations of current diagnostic and therapeutic frameworks. This experience motivated Dr. Uno to pursue a PhD and utilize advanced technology to look at the disease from a new perspective.
To validate their findings, the Nagoya University team employed a multi-faceted approach:
- Patient Fluid Analysis: Direct observation of hybrid clusters in samples from human patients.
- Advanced Microscopy: Real-time imaging of the formation of invadopodia and the subsequent tissue drilling process.
- Mouse Models: In vivo experiments to track how these hybrid spheres behave in a living biological system compared to single-cell suspensions.
- Single-Cell RNA Sequencing: Analyzing the gene expression of individual cells within the clusters to identify the exact pathways (like TGF-β1) being activated.
Implications for Future Therapeutics and Diagnostics
The identification of the TGF-β1 signaling pathway and the role of mesothelial cells opens several new doors for the treatment of ovarian cancer. Current treatment protocols are almost entirely "cancer-centric," focusing on the destruction of the malignant cells themselves. However, the Nagoya study suggests that a "microenvironment-centric" approach may be necessary.
Potential future therapies could include:
- TGF-β1 Inhibitors: By blocking the signal that cancer cells send to mesothelial cells, doctors might be able to prevent the formation of hybrid clusters and the development of invadopodia, effectively "disarming" the cancer’s ability to invade.
- Targeting Invadopodia: Developing drugs that inhibit the structural proteins required for mesothelial cells to form their drilling protrusions.
- Liquid Biopsies of Abdominal Fluid: Instead of relying solely on blood tests (which are often ineffective for early-stage ovarian cancer), doctors could analyze small samples of abdominal fluid for the presence of these hybrid spheres to predict disease progression or assess how well a patient is responding to treatment.
Statistical Context and the Global Burden
Ovarian cancer remains a significant global health challenge. According to the World Cancer Research Fund, there are nearly 314,000 new cases of ovarian cancer diagnosed annually worldwide. Because symptoms—such as bloating, pelvic pain, and difficulty eating—are often vague and mimic less serious conditions, approximately 70% of patients are diagnosed at Stage III or IV. At these stages, the five-year survival rate drops significantly, often falling below 30%.
The findings from Nagoya University provide a biological explanation for these grim statistics. If the cancer can "recruit" help to spread across the abdomen in a matter of weeks, the window for early detection is much narrower than previously thought.
The oncology community has reacted to the study with cautious optimism. While the transition from laboratory discovery to clinical treatment can take years, the clarity provided by this research offers a concrete target for drug development. By understanding that ovarian cancer is a collaborative effort between different cell types, researchers can move away from the "lone wolf" model of metastasis and toward a more holistic understanding of how tumors manipulate the human body to ensure their own survival.
As research continues, the hope is that by breaking the partnership between cancer and mesothelial cells, the "silent killer" can finally be silenced, providing better outcomes for the thousands of women diagnosed with this devastating disease each year.