The field of immunotherapy has long operated under the premise that the effectiveness of a vaccine is primarily determined by its chemical composition—the specific antigens and adjuvants selected to trigger an immune response. However, a decade of research at Northwestern University has culminated in a paradigm-shifting discovery: the physical architecture of these components at the nanoscale is just as critical as the ingredients themselves. In a study published February 11 in the journal Science Advances, researchers demonstrated that by simply altering the orientation and position of a single cancer-targeting peptide within a vaccine, they could dramatically amplify the immune system’s ability to identify and destroy tumors.
This breakthrough marks a significant milestone in the emerging discipline of "structural nanomedicine." Led by nanotechnology pioneer Chad A. Mirkin and clinical oncologist Dr. Jochen Lorch, the research team utilized Spherical Nucleic Acids (SNAs) to treat cancers driven by the human papillomavirus (HPV). Their findings suggest that the future of oncology may lie not just in the discovery of new drugs, but in the precise structural engineering of existing ones.
The Evolution of Structural Nanomedicine
Structural nanomedicine is a concept that challenges the traditional "blender approach" to vaccine manufacturing. For decades, the standard protocol for developing vaccines, including the recent mRNA vaccines for COVID-19, involved mixing various components into a formulation where the relative positions of molecules were largely stochastic or uncontrolled. While effective for many infectious diseases, this lack of structural precision has proven to be a bottleneck in the development of therapeutic cancer vaccines, which require a much more potent and targeted immune activation.
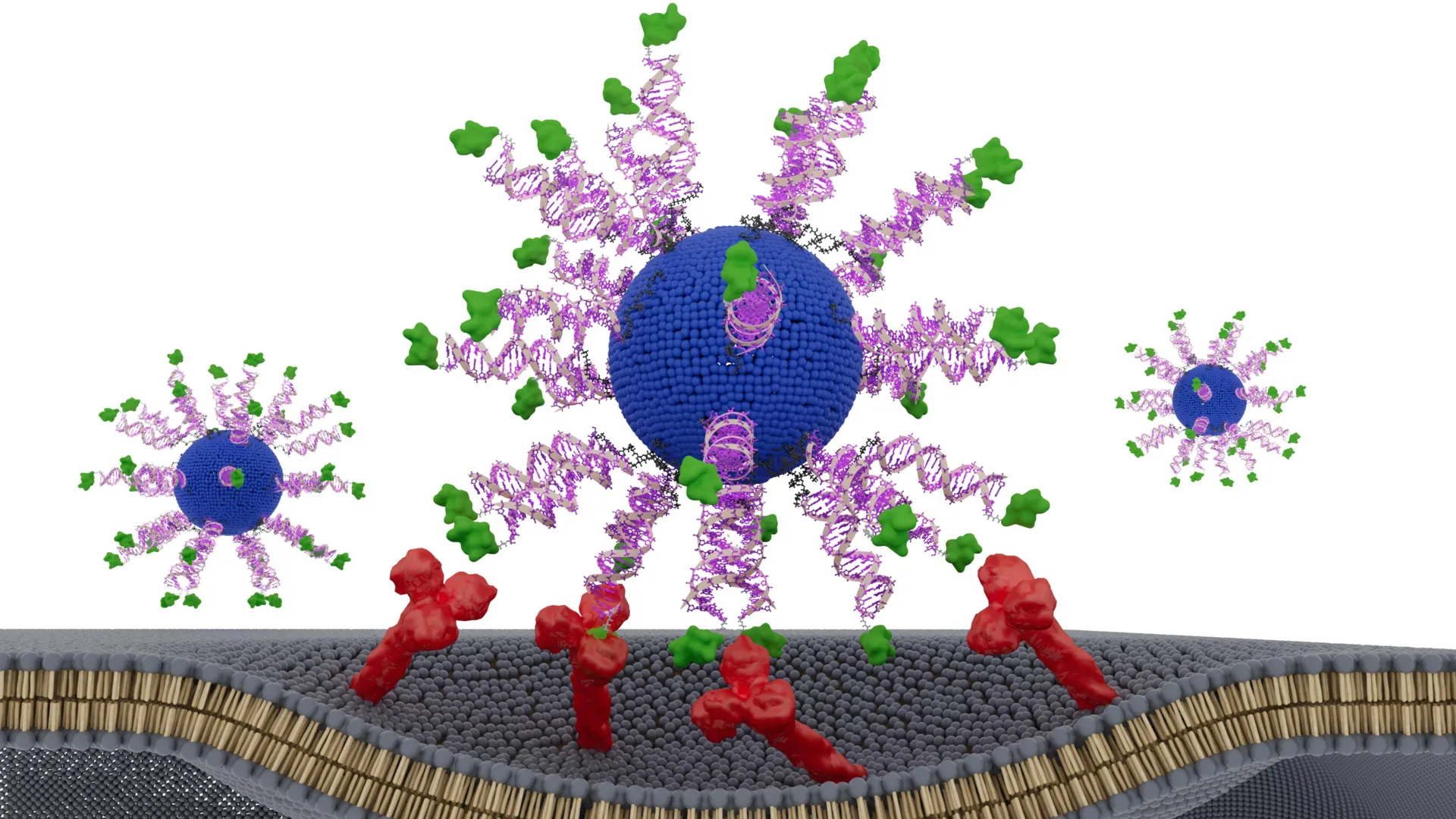
Chad A. Mirkin, the George B. Rathmann Professor of Chemistry at Northwestern’s Weinberg College of Arts and Sciences and director of the International Institute for Nanotechnology, has spent years advocating for a "bottom-up" approach to medicine. Mirkin, who invented SNAs, describes these structures as globular forms of DNA or RNA arranged around a nanoparticle core. Unlike linear DNA, which is often repelled by cell membranes, the spherical geometry of SNAs allows them to enter immune cells naturally and efficiently, where they can then release their therapeutic cargo.
The latest study provides the most compelling evidence to date that the spatial arrangement of these components—down to the orientation of a single peptide—can be the deciding factor between a vaccine that fails and one that successfully eradicates a tumor.
Deciphering the HPV Challenge
The research focused specifically on HPV-driven cancers, a growing public health concern. While preventive vaccines like Gardasil have been highly successful in reducing the incidence of new HPV infections, they are ineffective for individuals who have already developed cancer. HPV is the primary cause of nearly all cervical cancers and is increasingly linked to a surge in head and neck cancers, particularly oropharyngeal squamous cell carcinoma.
To address this, the Northwestern team sought to create a therapeutic vaccine. Unlike preventive vaccines, which generate antibodies to block a virus from entering cells, therapeutic vaccines must activate CD8+ "killer" T cells. these cells are the specialized infantry of the immune system, capable of recognizing specific proteins (antigens) displayed on the surface of cancerous cells and mounting a direct attack.
The challenge in cancer immunotherapy has always been the "strength" of this signal. Tumors are notoriously good at hiding from the immune system or suppressing T cell activity. The Northwestern study aimed to determine if the physical layout of an SNA vaccine could overcome this suppression.
Methodology: The Precision of N-Terminus vs. C-Terminus
The research team engineered a series of SNA vaccines, all containing the exact same chemical ingredients: a lipid core, immune-stimulating DNA (the adjuvant), and a specific fragment of an HPV protein known as the E7 peptide (the antigen). The only variable across the different vaccine versions was the placement and orientation of the E7 peptide.
The scientists tested three primary configurations:
- Encapsulated: The peptide was hidden inside the lipid core of the nanoparticle.
- Surface-Linked (C-terminus): The peptide was attached to the outer surface of the SNA via its C-terminus (the "tail" end of the protein chain).
- Surface-Linked (N-terminus): The peptide was attached to the outer surface via its N-terminus (the "head" end of the protein chain).
The results were stark. Despite having identical chemical concentrations, the vaccine that presented the antigen on the surface via its N-terminus outperformed the others by a massive margin.
Supporting Data: Quantitative Gains in Immune Potency
In laboratory evaluations and humanized animal models, the N-terminus surface-linked vaccine demonstrated a level of efficacy that surprised the researchers. Key data points from the study include:
- Interferon-Gamma Production: The N-terminus configuration triggered an eightfold increase in the production of interferon-gamma, a critical signaling protein that coordinates the immune system’s attack on cancer cells.
- T Cell Activation: This specific arrangement generated a significantly higher population of highly active, tumor-infiltrating CD8+ T cells.
- Tumor Growth Inhibition: In animal models of HPV-positive cancer, the optimized vaccine led to a marked reduction in tumor volume and a significant extension of survival rates compared to the other configurations.
- Human Patient Samples: Perhaps most importantly, the researchers tested the vaccine designs on tumor samples taken from human patients with head and neck cancer. The optimized SNA design increased the cancer-killing efficacy of the patients’ own immune cells by twofold to threefold.
Dr. Jochen Lorch, a professor of medicine at Northwestern University Feinberg School of Medicine and a co-leader of the study, emphasized that these improvements were achieved without increasing the dosage or adding new chemicals. "The immune system is sensitive to the geometry of molecules," Lorch noted. By optimizing the attachment point, the researchers ensured that the antigen was processed more efficiently by dendritic cells, which then "trained" the T cells more effectively.
The Role of SNAs in Modern Pharmacology
The success of this study adds to the growing body of evidence supporting the use of Spherical Nucleic Acids in a wide range of medical applications. To date, Mirkin’s laboratory has utilized structural nanomedicine to develop vaccine candidates for melanoma, triple-negative breast cancer, colon cancer, and prostate cancer.
Currently, seven different SNA-based drugs are in various stages of human clinical trials. Furthermore, the technology has already been integrated into over 1,000 commercial products, ranging from diagnostic tools to skincare. The versatility of the SNA platform stems from its modularity; scientists can swap out different antigens and adjuvants while maintaining the structural integrity of the sphere.
This modularity is what Mirkin believes will allow for a "bottom-up" revolution in medicine. By identifying the ideal structural configurations for different diseases, researchers can create a library of "blueprints" for vaccines that are optimized for maximum efficacy and minimum toxicity.
Implications for the Pharmaceutical Industry and AI Integration
The findings have profound implications for the pharmaceutical industry, particularly in how "failed" drugs are perceived. Mirkin suggests that many vaccine candidates that showed promise in the lab but failed in clinical trials may not have been chemically flawed, but rather structurally suboptimal.
"We may have passed up perfectly acceptable vaccine components simply because they were in the wrong configurations," Mirkin said. By revisiting these components through the lens of structural nanomedicine, the industry could potentially salvage years of research and billions of dollars in investment.
Looking forward, the research team anticipates that artificial intelligence (AI) and machine learning will play a pivotal role in the next phase of this work. The number of variables in a complex nanomedicine—including particle size, density, orientation, and placement—creates thousands of possible combinations. AI can analyze these myriad possibilities far faster than traditional trial-and-error methods, identifying the "sweet spot" for structural design before a single nanoparticle is synthesized in the lab.
A New Framework for Cancer Treatment
The study published in Science Advances does more than just offer a new hope for HPV patients; it provides a rigorous framework for the design of all future nanovaccines. As the medical community moves toward more personalized and precise treatments, the ability to control the physical architecture of a drug will become a standard requirement.
The research was a collaborative effort across multiple disciplines, involving the Weinberg College of Arts and Sciences, the McCormick School of Engineering, and the Northwestern University Feinberg School of Medicine. It was supported by the National Cancer Institute, the Lefkofsky Family Foundation, and the Robert H. Lurie Comprehensive Cancer Center.
As the "major train" of structural nanomedicine continues to gain momentum, the Northwestern team is already moving toward the next stage of development. Their goal is to transition these optimized HPV vaccine designs into formal clinical trials, bringing a new level of precision to the fight against cancer. In the words of Chad Mirkin, "Structure matters—consistently and without exception." This realization may very well be the key to unlocking the full potential of the human immune system in the battle against its most formidable foe.