Pancreatic cancer stands as one of the most formidable and lethal malignancies, notoriously difficult to diagnose in its nascent stages and often resistant to conventional therapeutic approaches. This grim reality has galvanized the scientific community, prompting an intensive search for innovative strategies to intercept the disease before it progresses to an untreatable state. While the involvement of nerves in facilitating cancer spread, particularly in advanced stages, has been a recognized phenomenon, the precise role of the nervous system in the very genesis of pancreatic cancer has, until now, remained largely enigmatic.
A pivotal study conducted by researchers at Cold Spring Harbor Laboratory (CSHL) has unveiled compelling evidence that the nervous system is far more than a passive bystander or a mere conduit for metastasis; it actively contributes to the earliest phases of pancreatic cancer development, even before overt tumors manifest. This groundbreaking discovery fundamentally alters the understanding of pancreatic cancer progression and opens promising new avenues for early intervention and treatment.
A Deeper Look into Perineural Invasion and Beyond
For decades, the prevailing understanding of nerve involvement in cancer centered on "perineural invasion" (PNI). As Jeremy Nigri, a postdoctoral researcher in Professor David Tuveson’s laboratory at CSHL, explains, PNI refers to the process where "cancer cells will migrate within the nerve and use the nerve as a way to metastasize." This aggressive characteristic is particularly common in pancreatic cancer, as well as in prostate and certain head and neck cancers, and is often associated with poorer patient prognosis. PNI signifies that nerves serve as a ‘highway’ for established cancer cells to spread to distant sites or infiltrate surrounding tissues.
However, the new research from Nigri and his CSHL colleagues pushes the timeline of nerve involvement significantly earlier. Their findings, recently published in Cancer Discovery, a prestigious journal of the American Association for Cancer Research, demonstrate that the nervous system initiates its detrimental influence not merely as a consequence of tumor formation, but as an active participant in the conditions that foster precancerous growth. This represents a paradigm shift, moving beyond the established view of nerves as post-tumor invasion routes to recognizing them as architects of the tumor microenvironment itself, right from the disease’s inception.
The Silent Killer: Pancreatic Cancer’s Dire Statistics
To fully appreciate the significance of this research, it is crucial to understand the daunting landscape of pancreatic cancer. Globally, pancreatic cancer is the seventh leading cause of cancer-related deaths, and in many developed countries, it ranks even higher, often as the third or fourth leading cause. The 5-year survival rate for pancreatic cancer remains tragically low, hovering around 12% in the United States, a figure that has seen only modest improvements over the past few decades despite advancements in oncology. This dismal prognosis is primarily attributed to the disease’s insidious nature: symptoms often do not appear until the cancer is advanced and has spread, making early detection exceedingly rare. Furthermore, the tumor microenvironment of pancreatic cancer is notoriously complex and fibrotic, creating a formidable barrier that limits drug delivery and efficacy, rendering many standard treatments ineffective.
Current treatment modalities typically involve surgery (if the tumor is resectable, which is only about 15-20% of cases), chemotherapy (such as FOLFIRINOX or gemcitabine/nab-paclitaxel), and sometimes radiation therapy. While immunotherapy has revolutionized the treatment of several other cancers, it has shown limited success in pancreatic cancer, underscoring the urgent need for novel therapeutic targets and strategies. The CSHL study offers a beacon of hope in this challenging environment by identifying a previously unappreciated vulnerability in the disease’s earliest stages.
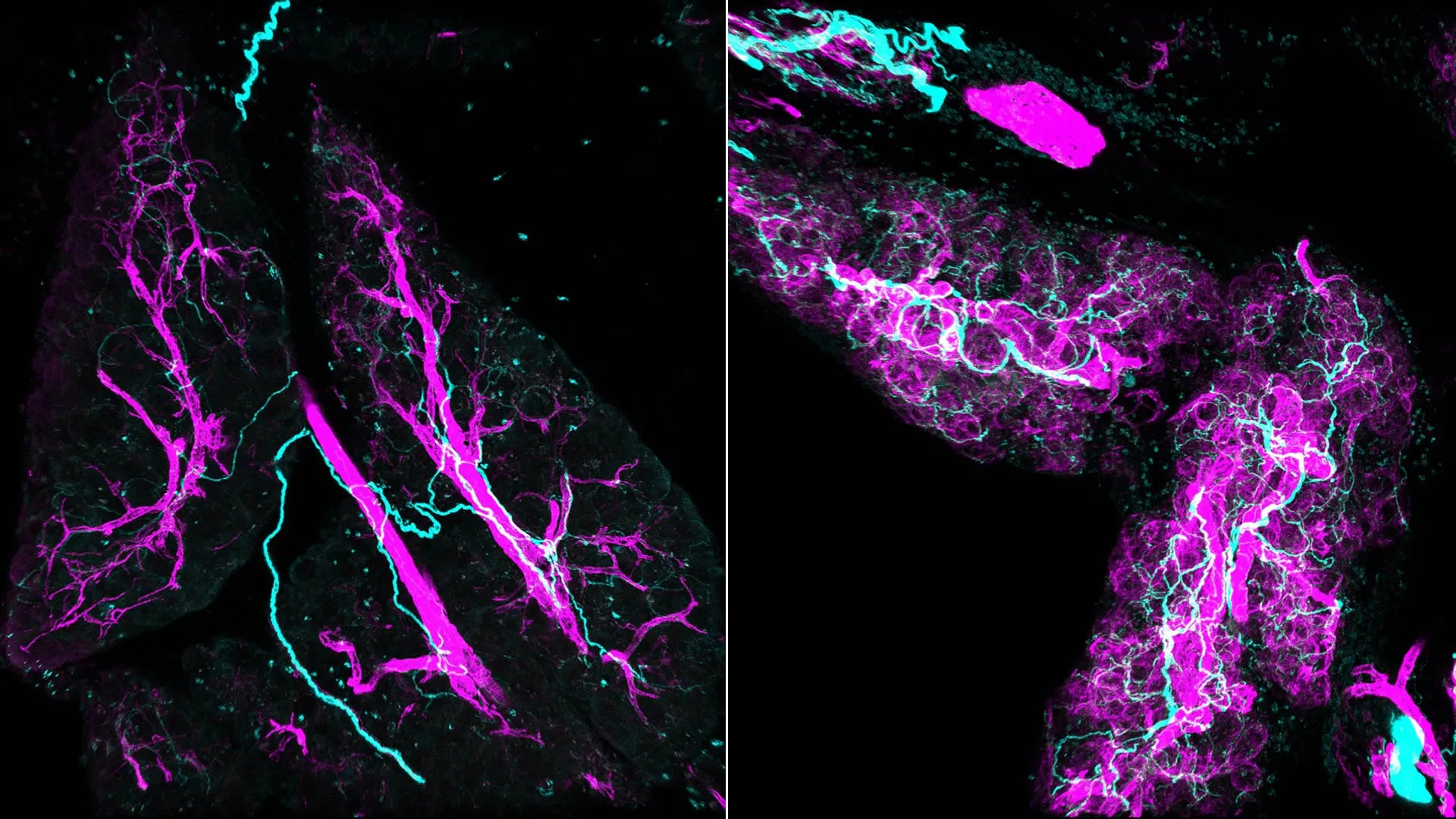
Unveiling the Hidden Network: The Power of 3D Imaging
A critical breakthrough enabling this discovery was the application of advanced 3D imaging techniques. The research team employed a method known as whole-mount immunofluorescence, which allowed them to capture intricate, three-dimensional images of precancerous lesions and their surrounding cellular architecture. This technological leap was instrumental in revealing what traditional 2D imaging, which typically shows nerve fibers as scattered dots, could not.
"When we first saw this picture, I was shocked," Nigri recounted, expressing the profound impact of the visualization. "I couldn’t even imagine the lesion like this. I’d only ever seen it in 2D." The 3D images painted a starkly different picture: a dense, intricate, and interconnected network of nerve fibers intricately woven throughout the early pancreatic lesions, specifically observed to be tightly wrapped around tumor-promoting fibroblasts known as myCAFs (myofibroblast-like cancer-associated fibroblasts). This unprecedented visualization provided the first tangible evidence of the extensive and intimate physical interaction between nerves and precancerous cells at such an early stage.
A Vicious Cycle: The MyCAF-Nerve Feedback Loop
The CSHL team’s investigations, spanning both experimental models in mice and analyses of human cells, meticulously uncovered the molecular mechanisms driving this early nerve-cancer crosstalk. They identified a harmful, self-sustaining feedback loop between myCAFs and nerves, which actively fuels precancerous growth.
The cycle begins with myCAFs, which are known to be key architects of the tumor microenvironment, releasing specific chemical signals. While the exact repertoire of these signals is a subject of ongoing research, they are understood to be potent chemoattractants that lure nearby nerve fibers from the sympathetic nervous system. The sympathetic nervous system is a crucial component of the autonomic nervous system, responsible for regulating the body’s ‘fight or flight’ response, influencing heart rate, blood pressure, and metabolism.
Once attracted to the precancerous lesions, these sympathetic nerve fibers release norepinephrine, a primary neurotransmitter associated with the sympathetic nervous system. Norepinephrine then binds to specific receptors located on the surface of the myCAFs. This binding event triggers a cascade of intracellular events within the myCAFs, most notably a significant surge in intracellular calcium levels. This calcium spike, in turn, further activates the myCAFs, enhancing their pro-tumorigenic functions and encouraging the proliferation of precancerous cells. Simultaneously, the activated myCAFs release even more nerve-attracting signals, thereby strengthening and perpetuating this self-reinforcing feedback loop. This continuous interplay effectively creates a conducive environment for cancer cells to thrive and progress, long before they are considered fully malignant.
Interrupting the Loop: A Glimmer of Hope for Treatment
The discovery of this critical feedback loop naturally led the researchers to investigate whether interrupting this nerve-myCAF interaction could impede cancer progression. In a series of compelling experiments, the team utilized a neurotoxin to selectively disable the sympathetic nervous system in their experimental models. The results were striking: "We show reduced fibroblast activation and a nearly 50% reduction in tumor growth," Nigri stated, highlighting the dramatic impact of disrupting this early nerve activity. This significant reduction in tumor growth provides strong causal evidence that the sympathetic nervous system is not just passively involved but actively drives early pancreatic cancer development.
This finding carries profound implications for therapeutic development. Given that this detrimental interaction between myCAFs and nerves occurs at such an early stage of the disease, targeting it presents a novel opportunity for therapeutic intervention, potentially even as a preventative measure for high-risk individuals or to slow progression in early-stage disease.
Potential New Therapeutic Strategies: Repurposing Existing Drugs
One of the most exciting aspects of this research is the potential for repurposing existing medications. The findings suggest that drugs designed to modulate sympathetic nervous system activity could be effective. Doxazosin, for instance, is an alpha-adrenergic blocker commonly prescribed to treat high blood pressure and benign prostatic hyperplasia. By blocking alpha-adrenergic receptors, doxazosin could potentially interfere with norepinephrine’s ability to bind to myCAFs, thereby disrupting the harmful feedback loop.
"The idea that an existing medication, already approved for other conditions, could be leveraged to tackle such an aggressive cancer is incredibly promising," remarks Dr. Eleanor Vance, an independent oncologist not involved in the study. "It could significantly shorten the path to clinical trials, offering hope to patients much sooner than developing entirely new compounds."
The researchers envision these new strategies not as standalone cures, but as complementary approaches to established treatments like chemotherapy or immunotherapy. By targeting the fundamental drivers of the tumor microenvironment from an early stage, these interventions could potentially enhance the efficacy of existing therapies, reduce tumor burden, and ultimately improve patient outcomes.
Looking Ahead: From Bench to Bedside
The CSHL team is keenly aware that this discovery is an important first step. "The next step will be to study this more in detail and try to find a way to block the crosstalk between fibroblasts and nerves," Nigri affirms. This involves further elucidating the specific signaling molecules involved, identifying optimal drug targets, and conducting preclinical studies to validate the safety and efficacy of potential therapeutic candidates.
Organizations like the Lustgarten Foundation and the Pancreatic Cancer Action Network, major funders of pancreatic cancer research, have expressed their enthusiasm for these findings. "Breakthroughs like these are precisely why we invest in innovative research," states a representative from the Pancreatic Cancer Action Network. "They offer a fresh perspective on a notoriously tough disease and provide tangible hope for the patients and families we serve. We are committed to supporting scientists like Dr. Nigri in their efforts to translate these discoveries into life-saving treatments."
This research marks a significant turning point in the understanding of pancreatic cancer. By revealing the nervous system’s active and early role in driving the disease, CSHL scientists have not only redefined the pathogenesis of this deadly cancer but also illuminated a novel therapeutic vulnerability. The long-term vision is clear: to leverage this knowledge to develop effective interventions that can halt pancreatic cancer in its tracks, offering a much-needed lifeline to those affected by this devastating illness and ultimately improving the tragically low survival rates that have plagued the medical community for too long. The journey from laboratory discovery to clinical application is often arduous, but the foundational insights provided by this study offer a powerful new direction in the relentless fight against pancreatic cancer.