The landscape of global diagnostics is undergoing a seismic shift as a team of engineers and scientists at Arizona State University (ASU) unveils a new technology capable of detecting lethal pathogens with unprecedented speed, accuracy, and affordability. The device, known as Nanoparticle-Supported Rapid Electronic Detection, or NasRED, represents a fusion of nanotechnology and electronic sensing that could bridge the widening gap between high-end laboratory testing and accessible point-of-care diagnostics. By utilizing a single drop of blood or other bodily fluids, the NasRED platform delivers results in approximately 15 minutes at a cost of roughly $2 per test, offering a potential solution to some of the most pressing challenges in modern public health, from pandemic surveillance to the management of chronic illnesses in remote regions.
The NasRED Innovation: Technical Specifications and Mechanism
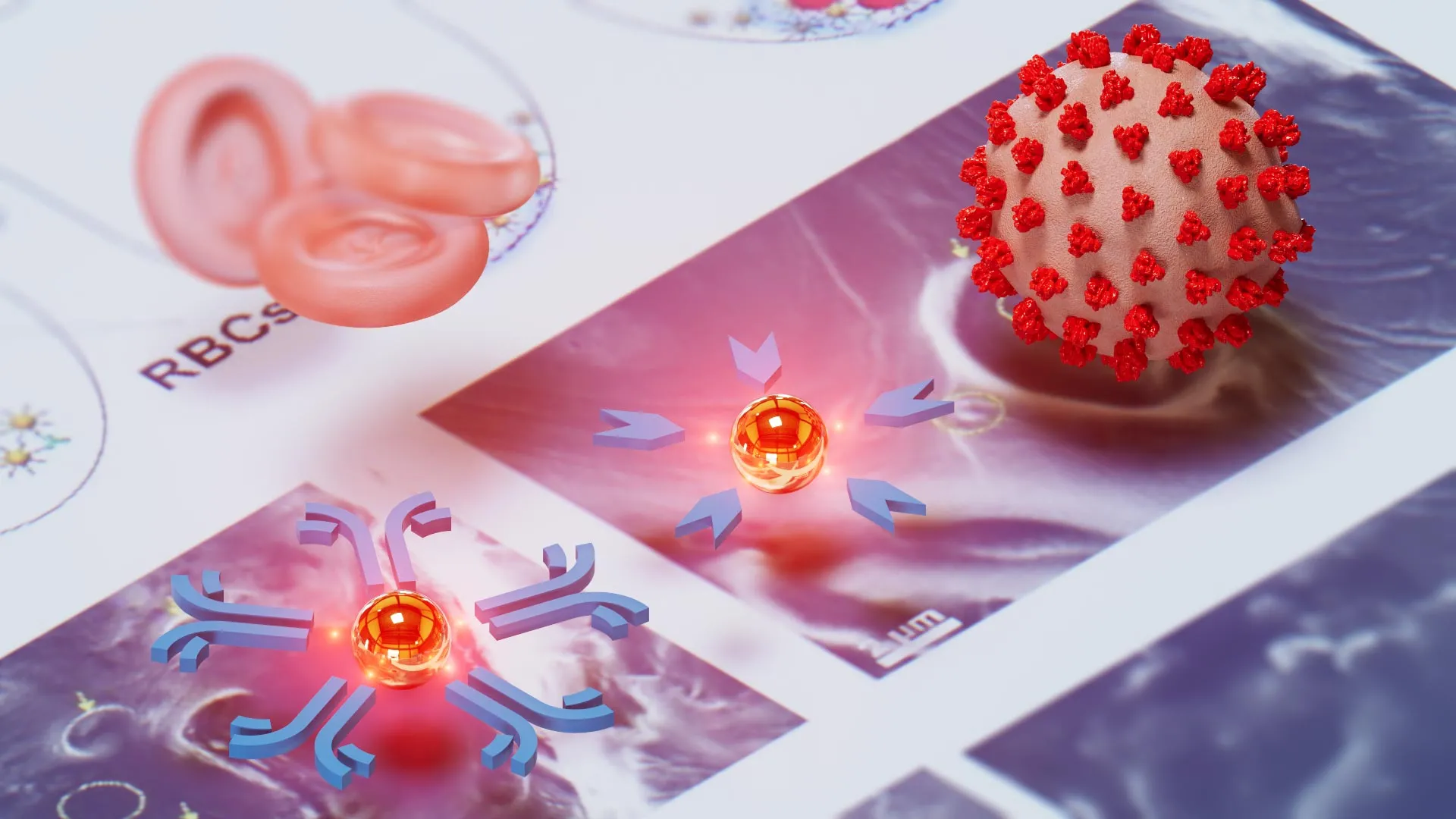
At the heart of the NasRED system is a sophisticated application of gold nanoparticles, engineered at the molecular level to act as highly sensitive biological sensors. These nanoparticles are coated with specific ligands—either antibodies or antigens—depending on the target disease. In the case of viral detection, such as for COVID-19 or Ebola, the nanoparticles are outfitted with antibodies that behave like microscopic magnets, seeking out and binding to viral proteins. Conversely, to detect an immune response, the particles are coated with antigens to attract antibodies produced by the patient’s own body.
The physical mechanism of the test relies on the principle of sedimentation. When the target pathogen or antibody is present in a sample, it causes the functionalized gold nanoparticles to clump together and sink to the bottom of a testing tube. If the sample is negative, the nanoparticles remain suspended, maintaining a cloudy or tinted appearance in the liquid. The NasRED device then utilizes a custom-built electronic detector equipped with a small LED light beam. By measuring the amount of light that passes through the upper portion of the liquid, the device can quantify the presence of a disease with extreme precision. A clearer fluid at the top indicates that the nanoparticles have precipitated, confirming a positive result.
Lead researcher Chao Wang, an associate professor at ASU’s Biodesign Center for Molecular Design and Biomimetics and the School of Electrical, Computer and Energy Engineering, emphasizes that the primary achievement of NasRED is its ability to combine the ease of a rapid antigen test with a sensitivity that exceeds even the most rigorous laboratory standards. The device can detect concentrations in the attomolar range—a level of sensitivity comparable to finding a single drop of ink dispersed across 20 Olympic-sized swimming pools.
Addressing the Global Burden of Infectious Disease
The development of NasRED comes at a critical juncture for global health. Infectious diseases remain a leading cause of mortality worldwide, accounting for more than 10 million deaths annually. The burden is disproportionately felt in low- and middle-income countries, where the lack of infrastructure often precludes the use of gold-standard diagnostic methods like Polymerase Chain Reaction (PCR) or Enzyme-Linked Immunosorbent Assay (ELISA). These traditional methods require expensive, bulky equipment, stable electricity, and highly trained personnel—luxuries that are often unavailable in rural or resource-constrained settings.
Furthermore, diagnostic errors and delays are not confined to the developing world. In the United States, research published in BMJ Quality & Safety indicates that nearly 800,000 Americans are killed or permanently disabled every year due to diagnostic failures. Many of these tragedies involve treatable infections or vascular events that were missed during the initial window of intervention. By providing lab-quality results at the point of care, NasRED aims to eliminate the "turnaround time" that often dictates the difference between recovery and fatality.
For marginalized populations, such as those living in remote areas or individuals in high-risk groups for HIV and Hepatitis C, the portability of NasRED is a transformative feature. Frequent, localized testing can catch infections in their earliest stages, preventing the progression to severe complications like liver disease or cancer and halting the further transmission of the virus within the community.
Comparative Data: NasRED vs. Traditional Diagnostics
The efficacy of the NasRED platform is highlighted by its performance metrics relative to existing diagnostic technologies. In a study recently published in the journal ACS Nano, the ASU research team demonstrated that NasRED is approximately 3,000 times more sensitive than the conventional ELISA test. Despite this increased sensitivity, the device requires 16 times less sample volume and provides results 30 times faster than the laboratory standard.
When compared to molecular tests like the Abbott ID NOW—a widely used rapid molecular platform for COVID-19—NasRED achieved comparable sensitivity without the need for complex sample preparation or the thermal cycling required for genetic amplification. The ability to detect viral loads directly from raw bodily fluids significantly simplifies the diagnostic workflow, making it feasible for use by frontline health workers with minimal training.
The modularity of the system further enhances its utility. Because the gold nanoparticles can be functionalized with a wide variety of proteins, the same electronic hardware can be used to screen for an array of conditions. The ASU team has already successfully demonstrated the platform’s capability in detecting Shiga toxin-producing E. coli, African swine fever, and various cancer biomarkers.
Chronology of Development and Future Roadmap
The journey toward the NasRED device began with earlier iterations of nanoparticle technology designed to combat the Ebola virus. During previous outbreaks, the need for a rapid, field-deployable test became evident as samples often had to be transported long distances to centralized labs, delaying quarantine efforts. The ASU team successfully adapted their initial concepts to create a more robust, electronic-based detection system that could withstand the rigors of field use while improving sensitivity by several orders of magnitude.
In the wake of the COVID-19 pandemic, the researchers pivoted to apply the technology to SARS-CoV-2. The current study in ACS Nano serves as a validation of the platform’s "pinpoint accuracy" in distinguishing the coronavirus from other respiratory infections.
Looking ahead, the research team is focused on two primary objectives: miniaturization and automation. While the current setup requires small benchtop equipment for mixing and spinning samples, the ultimate goal is to create a fully integrated, handheld device. Such a tool would function similarly to a home pregnancy test or a glucose monitor but with the sophisticated analytical power of a high-end medical laboratory. This evolution would pave the way for widespread home testing and real-time public health surveillance.
Expanding the Diagnostic Frontier: Lyme Disease and Chronic Conditions
One of the most promising applications for NasRED lies in the detection of Lyme disease. Currently, diagnosing early-stage Lyme infection is notoriously difficult, often relying on subjective clinical symptoms because existing blood tests frequently return false negatives during the first few weeks of infection. Chao Wang was recently honored with the Bay Area Lyme Foundation Emerging Leader Award, a recognition that includes funding to adapt NasRED for early Lyme detection.
The high sensitivity of the NasRED platform is uniquely suited to identifying the low levels of antibodies or bacterial proteins present in the early stages of a tick-borne illness. If successful, this could revolutionize the treatment of Lyme disease, allowing patients to begin antibiotic therapy before the infection spreads to the nervous system or joints.
Beyond infectious diseases, the ASU team is exploring the use of NasRED for monitoring chronic conditions and neurodegenerative diseases. Preliminary research into Alzheimer’s-related proteins suggests that the device could eventually play a role in the early screening of dementia, providing a non-invasive and affordable alternative to expensive PET scans or cerebrospinal fluid draws.
Implications for Public Health and Economic Stability
The economic implications of a $2, 15-minute diagnostic tool are profound. Rapid and accurate testing is a cornerstone of "test-to-treat" strategies, which aim to reduce the overall cost of healthcare by preventing hospitalizations and long-term disability. By enabling immediate diagnosis, NasRED allows for the swift administration of targeted therapies, reducing the reliance on broad-spectrum antibiotics and helping to combat the global rise of antimicrobial resistance.
In the context of pandemic preparedness, the NasRED platform offers a scalable solution for monitoring emerging outbreaks. Because the test is modular, health authorities could theoretically deploy updated nanoparticle "cartridges" within weeks of identifying a new pathogen, creating a global network of early-warning sensors.
The ASU research team, which includes Yeji Choi, Seyedsina Mirjalili, Ashif Ikbal, and several other contributors from the School of Electrical, Computer and Energy Engineering, views NasRED not just as a piece of medical equipment, but as a public health game-changer. As the technology moves toward commercialization and regulatory approval, its potential to save lives by democratizing access to high-quality diagnostics remains its most significant promise. In a world where the next health crisis may be just around the corner, tools like NasRED provide a vital line of defense, ensuring that geography and socioeconomic status are no longer barriers to life-saving information.