Researchers at Northwestern University have unveiled a transformative discovery in the field of immunotherapy, demonstrating that the physical architecture of a vaccine is as critical to its success as its chemical composition. For over a decade, scientists led by nanotechnology pioneer Chad A. Mirkin have investigated how the spatial arrangement of vaccine components influences the body’s immune response. Their latest study, published on February 11 in the journal Science Advances, applies these principles to therapeutic vaccines for cancers driven by the human papillomavirus (HPV). By making minute adjustments to the orientation of a single targeting peptide, the team achieved a significant increase in the immune system’s ability to identify and destroy malignant tumors, marking a milestone in the emerging field of structural nanomedicine.
The research shifts the focus from what ingredients are included in a vaccine to how those ingredients are organized at the nanoscale. Traditionally, vaccine development has relied on a "blender approach," where antigens (the targets) and adjuvants (the stimulants) are mixed into a formulation with little control over their relative positions. Northwestern’s findings suggest that this lack of structural precision may be the reason many promising vaccine candidates fail in clinical trials. By utilizing Spherical Nucleic Acids (SNAs)—globular DNA structures that naturally penetrate immune cells—the researchers have demonstrated that the "geometry of immunity" can be optimized to turn a weak response into a potent anti-tumor attack.
The Evolution of Structural Nanomedicine
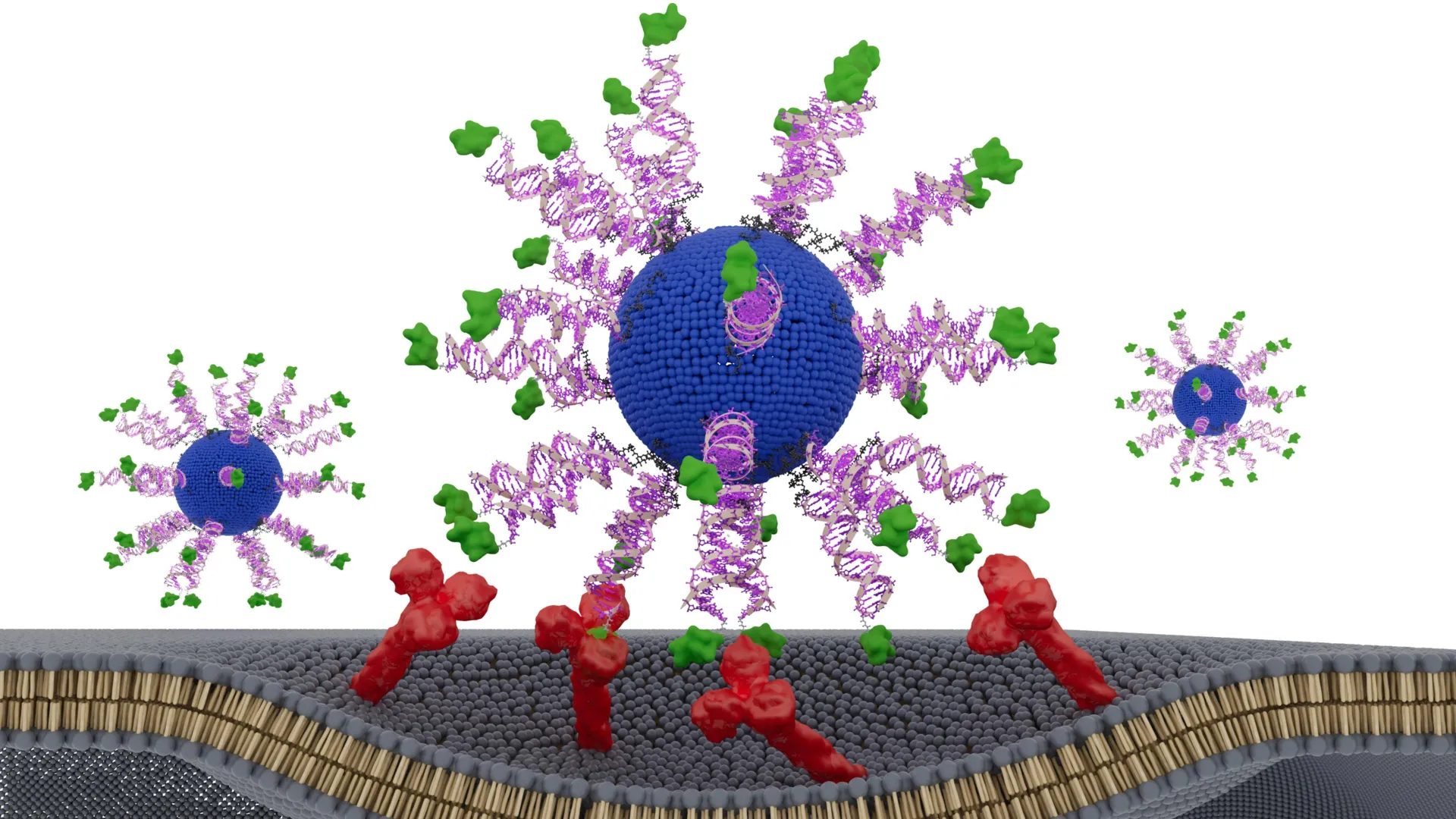
The concept of structural nanomedicine was introduced by Chad A. Mirkin, the George B. Rathmann Professor of Chemistry at Northwestern’s Weinberg College of Arts and Sciences and director of the International Institute of Nanotechnology. Mirkin, who invented SNAs, has spent years advocating for a "bottom-up" approach to drug design. Unlike conventional linear DNA or RNA, SNAs consist of a nanoparticle core densely functionalized with highly oriented oligonucleotides. This radial arrangement grants them unique biological properties, including the ability to enter cells without the need for toxic delivery agents and resistance to enzymatic degradation.
The timeline of this technology spans more than two decades of fundamental research. Early work focused on the basic chemical properties of SNAs, which eventually led to their use in diagnostic tools and later in therapeutics. Over the last ten years, the Northwestern team has systematically applied the SNA platform to various forms of cancer, including melanoma, prostate cancer, and triple-negative breast cancer. To date, seven SNA-based drugs have entered human clinical trials, and the technology has been integrated into over 1,000 commercial products. The latest study represents a culmination of this effort, providing a blueprint for how structural precision can be used to combat HPV-driven malignancies, which include cervical cancer and a growing number of head and neck cancers.
Experimental Methodology and the HPV Challenge
The study specifically addressed the need for therapeutic vaccines—vaccines designed to treat existing disease rather than prevent infection. While preventive HPV vaccines (such as Gardasil) have been highly effective at reducing infection rates, they offer no benefit to patients who have already developed HPV-positive tumors. These tumors are characterized by the expression of specific viral proteins, such as the E7 protein, which the immune system often fails to recognize as a threat.
To overcome this, the Northwestern team designed a series of SNA vaccines containing three identical components: a lipid core, immune-activating DNA sequences, and a peptide fragment derived from the HPV E7 protein. While the ingredients remained constant, the researchers engineered three distinct structural configurations:
- Encapsulated: The peptide was hidden inside the lipid core of the nanoparticle.
- C-terminus Surface Display: The peptide was attached to the surface of the SNA via its C-terminus.
- N-terminus Surface Display: The peptide was attached to the surface via its N-terminus.
These variations were tested in humanized animal models and in ex vivo samples taken from patients with HPV-positive head and neck cancers. The goal was to observe how these subtle changes influenced the activation of CD8+ "killer" T cells, the primary effector cells responsible for hunting and destroying cancer cells.
Data-Driven Results: The Power of Orientation
The results of the study were stark and highlighted the sensitivity of the immune system to molecular geometry. The configuration featuring the peptide attached via its N-terminus on the surface of the SNA outperformed all other designs. This specific arrangement led to a cascade of enhanced immune markers:
- Interferon-Gamma Production: The N-terminus configuration triggered an eightfold increase in the production of interferon-gamma, a critical signaling protein that coordinates the anti-tumor response.
- T Cell Efficacy: Killer T cells generated by this specific vaccine were significantly more aggressive. In patient tumor samples, the rate of cancer cell destruction increased by twofold to threefold compared to other configurations.
- Tumor Suppression: In animal models, the N-terminus vaccine resulted in marked slowing of tumor growth and a substantial increase in overall survival rates.
Dr. Jochen Lorch, a professor of medicine at Northwestern’s Feinberg School of Medicine and co-leader of the study, emphasized that these improvements were achieved without increasing the dosage or adding new chemical agents. "The immune system is sensitive to the geometry of molecules," Lorch noted. By optimizing the attachment point, the researchers ensured that the antigen was processed more efficiently by dendritic cells, which then "trained" the T cells to recognize the tumor more effectively.
Moving Beyond the Blender Approach
A significant portion of the study’s analysis focused on the limitations of current vaccine manufacturing. Mirkin described the current state of the industry as being in a "blender" phase, where complexity has increased but structural control has lagged. He cited the COVID-19 mRNA vaccines as a primary example. While highly effective, these vaccines consist of lipid nanoparticles where the distribution of mRNA and other components varies from one particle to the next.
"No two particles are the same in current formulations," Mirkin stated. While this may suffice for infectious diseases where the immune target is easily accessible, cancer immunotherapy requires a higher degree of precision. Malignant tumors often employ immunosuppressive tactics to hide from the body. To break through this "immune "coldness," a vaccine must deliver a perfectly timed and structured set of signals to immune cells. The Northwestern study provides the first definitive evidence that structural nanomedicine can provide this level of control, potentially leading to vaccines that are both more effective and less toxic.
Implications for Future Cancer Therapy and AI Integration
The implications of this research extend far beyond HPV. The Northwestern team has already demonstrated the versatility of the SNA platform in other "hard-to-treat" cancers. By establishing a framework where structure is a primary variable, researchers can now revisit previously failed vaccine candidates. Many drugs that showed promise in the lab but failed in clinical trials may simply have been delivered in the wrong configuration.
Looking forward, Mirkin and his colleagues anticipate that artificial intelligence (AI) and machine learning will play a pivotal role in the next generation of vaccine design. The number of variables in a structural nanomedicine—including particle size, DNA density, peptide orientation, and core composition—creates thousands of possible combinations. AI algorithms can be trained to analyze these structural permutations, predicting which arrangements will yield the highest efficacy and lowest toxicity. This would shift the discovery process from trial-and-error to a predictive, engineering-based model.
Institutional and Funding Context
The study was a collaborative effort involving several of Northwestern University’s premier research entities, including the International Institute of Nanotechnology, the McCormick School of Engineering, and the Robert H. Lurie Comprehensive Cancer Center. The interdisciplinary nature of the work—combining chemistry, engineering, and clinical oncology—was essential for translating nanoscale observations into potential clinical applications.
The research was supported by significant funding from the National Cancer Institute (NCI) under award numbers R01CA257926 and R01CA275430. Additional support was provided by the Lefkofsky Family Foundation. These investments reflect a broader federal and private interest in nanotechnology as a frontier for solving the complexities of cancer treatment.
As the field of structural nanomedicine gains momentum, the Northwestern study serves as a foundational proof of concept. The "train roaring down the tracks," as Mirkin described it, suggests a future where medicines are built from the bottom up, with every atom and molecule placed with purpose. For patients with HPV-driven cancers and other terminal malignancies, this shift toward structural precision offers a new pathway for durable, effective treatment.