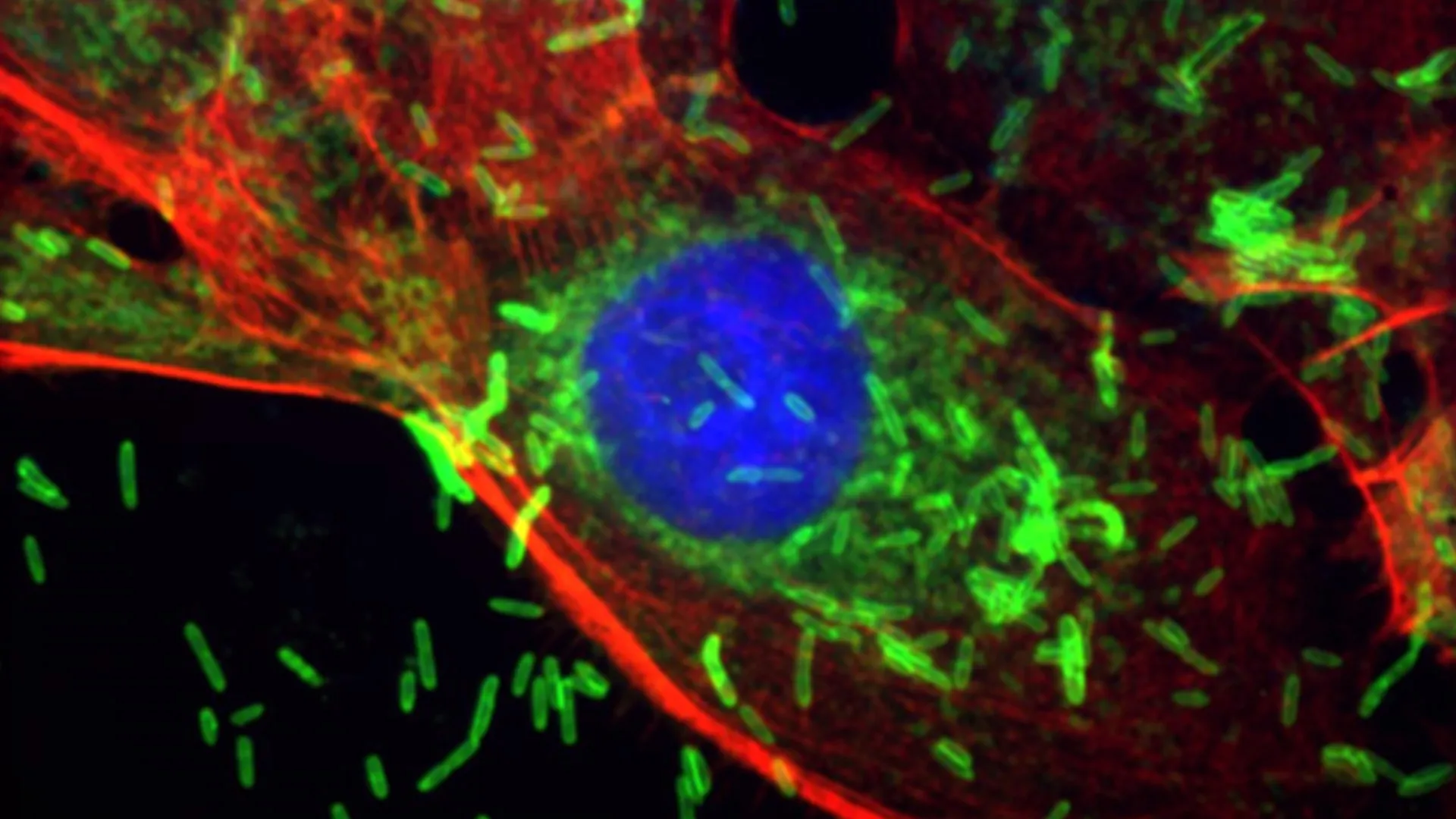
Scientists at the Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy have uncovered a surprising and concerning link between oral health and the insidious progression of breast cancer. Their pioneering research reveals that Fusobacterium nucleatum, a bacterium commonly associated with periodontal disease, may not only help initiate breast cancer but also significantly accelerate tumor growth and promote its metastatic spread by directly damaging cellular DNA and altering the fundamental behavior of cancer cells. This discovery marks a pivotal moment in understanding the complex interplay between the human microbiome and oncogenesis, opening new avenues for prevention, early detection, and targeted therapies.
The comprehensive study, meticulously detailed in the prestigious journal Cell Communication & Signaling, zeroes in on Fusobacterium nucleatum, a microbial entity that has previously garnered attention for its involvement in colorectal cancer and other malignancies. Researchers meticulously traced the bacterium’s journey, demonstrating its capacity to enter the bloodstream from oral cavities and subsequently establish itself within breast tissue. Once ensconced in this new environment, F. nucleatum was observed to trigger a cascade of inflammatory responses and instigate early cellular transformations unequivocally linked to the onset of cancer. Crucially, in sophisticated animal models mirroring human breast cancer, the presence of this bacterium dramatically expedited tumor proliferation and amplified the dissemination of cancer cells from the primary breast site to distant organs, most notably the lungs, a common metastatic target.
"The paramount takeaway from our extensive investigation is the definitive presence of this oral microbe within breast tissue and the undeniable connection between this specific pathogen and breast cancer," stated Dr. Cynthia Sears, a lead researcher from Johns Hopkins, whose team’s work was inspired by an accumulating body of smaller, epidemiological studies. These earlier investigations, collectively examining thousands of patients, had consistently hinted at a correlation between periodontal disease and an elevated risk of breast cancer, laying the groundwork for the current mechanistic exploration. "We were driven to delve deeper, to move beyond correlation and uncover the precise, underlying biological connections that could explain these observed links," added Dr. Sheetal Parida, the first author of the study and a dedicated research associate collaborating with Dr. Sears.
A Deep Dive into the Microbial Culprit and Its Mechanisms
Fusobacterium nucleatum is a gram-negative anaerobic bacterium that is a common inhabitant of the human oral cavity. While often present in healthy mouths in small numbers, its overgrowth is a hallmark of periodontal disease, a chronic inflammatory condition affecting the gums and supporting structures of the teeth. Periodontal disease, which affects nearly half of all adults over 30 in the United States, is characterized by persistent inflammation, tissue destruction, and bleeding gums, providing an entry point for bacteria into the bloodstream. The global prevalence of severe periodontitis is estimated at 10-15% of the adult population, representing a vast reservoir for systemic bacterial dissemination. For decades, the focus on F. nucleatum was primarily confined to oral pathology, but its increasing identification in extra-oral sites, particularly in the context of cancer, has underscored its role as an opportunistic pathogen with far-reaching systemic implications.
The Johns Hopkins study meticulously elucidated how F. nucleatum exerts its oncogenic influence. Experiments utilizing both advanced mouse models and cultures of human breast cancer cells provided a granular view of the bacterium’s tissue-altering capabilities. When F. nucleatum was directly introduced into the delicate breast ducts of the animal models, it provoked significant cellular abnormalities. These included the formation of metaplastic and hyperplastic lesions – noncancerous yet concerning changes where cells either proliferate excessively (hyperplasia) or transform into a different cell type (metaplasia). These cellular aberrations were invariably accompanied by pronounced inflammation, clear evidence of DNA damage within the affected cells, and a marked increase in cellular proliferation rates. Even more alarmingly, when the bacterium was introduced systemically into the bloodstream, mimicking its natural route from the oral cavity, it robustly augmented the growth and metastatic spread of pre-existing tumors.
The research team further peeled back the layers of biological complexity, uncovering a critical molecular pathway through which F. nucleatum orchestrates these detrimental effects. Exposure to the bacterium was found to inflict direct damage to cellular DNA, subsequently triggering the activation of the cell’s DNA repair systems. However, these activated repair mechanisms, particularly one known as nonhomologous end joining (NHEJ), are notoriously error-prone. While NHEJ is crucial for rapidly reconnecting broken DNA strands, its haste often comes at the cost of fidelity, leading to the introduction of mutations that can drive cancerous transformation. Even transient exposure to F. nucleatum was sufficient to elevate the levels of a specific protein, PKcs (Protein Kinase C-zeta). This increase in PKcs was directly correlated with enhanced cancer cell movement, increased cellular invasion into surrounding tissues, the acquisition of stem-like traits (which contribute to tumor recurrence and resistance), and a heightened resistance to conventional chemotherapy agents. This finding suggests a multifaceted mechanism where the bacterium not only initiates damage but also empowers cancer cells with aggressive properties.
The BRCA1 Connection: Amplified Risk in Genetically Susceptible Individuals
A particularly striking revelation from the study was the differential susceptibility observed among various cell types. Epithelial cells, which form the lining of the breast ducts where most breast cancers originate, alongside breast cancer cells harboring mutations in the BRCA1 gene, were found to be exceptionally vulnerable to the effects of F. nucleatum. These BRCA1-mutant cells exhibited significantly elevated levels of a specific surface sugar, Gal-GalNAc, which acts as a molecular "docking station" that facilitates the attachment and subsequent entry of the bacteria into the cells. Consequently, these genetically predisposed cells absorbed substantially more F. nucleatum and, remarkably, retained the bacteria over extended periods, even across multiple cell generations. This persistent intracellular presence of the bacterium intensified the DNA damage and amplified the cancer-promoting effects within these already vulnerable cells.
The BRCA1 gene is a tumor suppressor gene, playing a critical role in DNA repair and maintaining genomic stability. Mutations in BRCA1 are well-established as a major hereditary risk factor for breast and ovarian cancers, accounting for a significant proportion of familial cases. Individuals carrying a BRCA1 mutation face a substantially higher lifetime risk of developing breast cancer, often at an earlier age, and their tumors tend to be more aggressive, frequently exhibiting triple-negative characteristics. The current findings suggest a potent synergy, where an inherited genetic predisposition (BRCA1 mutation) interacts with an environmental microbial factor (F. nucleatum) to dramatically escalate cancer risk and accelerate disease progression.
"Our findings unequivocally establish a direct link between specific oral microbes and both breast cancer risk and its aggressive progression, particularly within individuals who are genetically susceptible," Dr. Sears emphasized. "Nothing in biology happens in isolation. The compelling results of our study strongly suggest that multiple risk factors converge, with F. nucleatum acting as a critical environmental cofactor that may cooperate with inherited BRCA1 mutations to not only promote the development of breast cancer but also to significantly enhance tumor aggressiveness." This insight underscores the emerging paradigm in cancer research that moves beyond single-factor causation to embrace a more holistic understanding of disease etiology.
Broader Impact and Implications for Prevention and Treatment
This groundbreaking research from Johns Hopkins carries profound implications for several facets of public health and oncology.
For Prevention Strategies:
The most immediate implication is the potential re-evaluation of oral hygiene as a critical component of overall cancer prevention, particularly for breast cancer. While the researchers emphasize that further human studies are needed, the mechanistic link established here suggests that diligent oral health practices – including regular brushing, flossing, and professional dental check-ups – could play a tangible role in reducing breast cancer risk by mitigating the systemic spread of F. nucleatum. This could lead to public health campaigns that integrate oral health into broader cancer prevention guidelines, similar to recommendations for diet and exercise. For individuals with known risk factors, such as a family history of breast cancer or identified BRCA1 mutations, maintaining exemplary oral hygiene might become an even more crucial preventive measure.
For Early Detection and Risk Assessment:
The study opens the door for novel screening methods. Future research might explore the feasibility of detecting F. nucleatum in breast tissue biopsies, nipple aspirate fluid, or even through specific biomarkers in blood tests, especially in high-risk populations. The presence of the bacterium or elevated PKcs levels could potentially serve as a prognostic indicator for aggressive disease or as a marker for increased risk in individuals with BRCA1 mutations. This could allow for more personalized risk assessment and earlier intervention strategies.
For Therapeutic Development:
The identified mechanisms provide exciting new targets for therapeutic intervention. Strategies could involve:
- Antibiotic therapy: Specifically targeting F. nucleatum in high-risk individuals or as an adjuvant treatment for existing breast cancer. However, the broader implications of systemic antibiotic use need careful consideration to avoid disrupting beneficial microbiota.
- Anti-inflammatory agents: Given the bacterium’s role in triggering inflammation, anti-inflammatory drugs might be explored to mitigate its effects.
- Targeting PKcs: Developing drugs that inhibit PKcs could potentially reduce cancer cell movement, invasion, stem-like properties, and chemoresistance, offering a new avenue for combating aggressive tumors.
- Targeting bacterial adhesion: Developing agents that interfere with the Gal-GalNAc binding mechanism could prevent F. nucleatum from attaching to and entering breast cells, particularly in BRCA1-mutant individuals.
For Cancer Research:
This study significantly advances the understanding of the tumor microenvironment and the intricate host-microbe interactions that influence cancer development and progression. It highlights the microbiome as a dynamic, influential factor, moving beyond a passive bystander to an active participant in oncogenesis. This will undoubtedly spur further interdisciplinary research, fostering collaborations between microbiologists, oncologists, immunologists, and dentists to unravel the full scope of microbial influence on cancer. It also emphasizes the importance of considering environmental factors in the context of genetic predispositions.
Chronology and Context
The journey towards this discovery has been incremental. For decades, researchers observed epidemiological links between chronic inflammation, including that from periodontal disease, and various systemic diseases, including certain cancers. The early 2000s saw a surge in research linking specific microbes, including F. nucleatum, to colorectal cancer. These findings set a precedent for investigating its role in other cancers. The "many small studies" connecting periodontal disease to breast cancer, as mentioned by Dr. Sears, served as the crucial epidemiological foundation, providing the impetus for the Johns Hopkins team to embark on their mechanistic quest. Their meticulous work, spanning several years, progressed from identifying the bacterium’s presence in breast tissue to dissecting its molecular mechanisms of action and finally demonstrating its impact on tumor initiation and metastasis in preclinical models. This systematic approach exemplifies the rigorous scientific process required to establish causality beyond mere correlation.
Official Responses and Funding Acknowledgements
While direct official statements from external bodies are yet to emerge, the implications are clear. Funding organizations like the Breast Cancer Research Foundation and the Congressionally Directed Medical Research Programs (Department of Defense Breast Cancer Research Program grants BC191572 and BC210668) have played a critical role in supporting this high-impact research. Their investment underscores a recognition of the need for innovative approaches to understand and combat breast cancer, including exploring unconventional risk factors. The Bloomberg~Kimmel Institute for Cancer Immunotherapy’s support further highlights the potential for microbial insights to inform immunotherapeutic strategies, given the bacterium’s role in inflammation and cellular changes.
Future Frontiers
The researchers are clear that while these findings are transformative, they represent a significant step rather than a conclusive end. "More work is unequivocally needed to understand how these findings can be robustly translated into tangible patient care strategies," the team emphasized. Future studies will be critical in:
- Validating in human cohorts: Confirming the presence and impact of F. nucleatum in human breast cancer patients, particularly correlating bacterial load with disease aggressiveness and patient outcomes.
- Intervention studies: Designing clinical trials to explore whether targeted interventions, such as specific oral hygiene regimens or antimicrobial treatments, can genuinely reduce breast cancer risk or improve prognoses.
- Exploring other microbes: Investigating if other oral or gut microbes also play a role in breast cancer development, contributing to a broader understanding of the cancer "microbiome."
- Longitudinal studies: Tracking individuals over time to observe the development of breast cancer in relation to their oral microbiome status.
In addition to Dr. Sears, the dedicated research team comprised Sheetal Parida, Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson, and Cynthia Sears. Their collective efforts have illuminated a previously underappreciated pathway in breast cancer biology, offering a beacon of hope for novel prevention and treatment strategies rooted in the intricate world of microbial ecology. The message is clear: the health of our mouths may have far more profound implications for our overall well-being, including our susceptibility to one of the most prevalent cancers, than previously imagined.