Northwestern University researchers have achieved a significant milestone in the quest to understand and treat human spinal cord injuries (SCIs) with the development of the most sophisticated lab-grown model to date. This innovative platform, utilizing human spinal cord organoids, has not only replicated the complex biological consequences of SCI with unprecedented fidelity but has also demonstrated the remarkable efficacy of a novel regenerative therapy known as "dancing molecules." The findings, published on February 11 in the prestigious journal Nature Biomedical Engineering, represent a pivotal step forward, offering a more accurate and accessible avenue for evaluating potential treatments before human clinical trials.
Recreating the Devastation of Spinal Cord Injury in the Lab
For the first time, scientists have successfully engineered human spinal cord organoids that faithfully reproduce the hallmark biological responses to traumatic injury. These miniature, stem-cell-derived organs, when subjected to simulated trauma, exhibited key pathological features observed in actual human SCIs. This includes widespread cell death, a robust inflammatory response orchestrated by immune cells, and the formation of glial scars. Glial scarring, a dense accumulation of reactive astrocytes and other glial cells, poses a formidable barrier to nerve regeneration, physically and chemically impeding the regrowth of severed axons.
The Northwestern team meticulously guided induced pluripotent stem cells over several months to develop into complex spinal cord tissue. Crucially, they were the first to incorporate microglia, the resident immune cells of the central nervous system, into these organoids. This addition significantly enhanced the model’s realism, allowing it to accurately mimic the intricate inflammatory cascade triggered by SCI. "It’s kind of a pseudo-organ," explained Samuel I. Stupp, the study’s senior author and a distinguished professor at Northwestern University. "We were the first to introduce microglia into a human spinal cord organoid, so that was a huge accomplishment. It means that our organoid has all the chemicals that the resident immune system produces in response to an injury. That makes it a more realistic, accurate model of spinal cord injury."
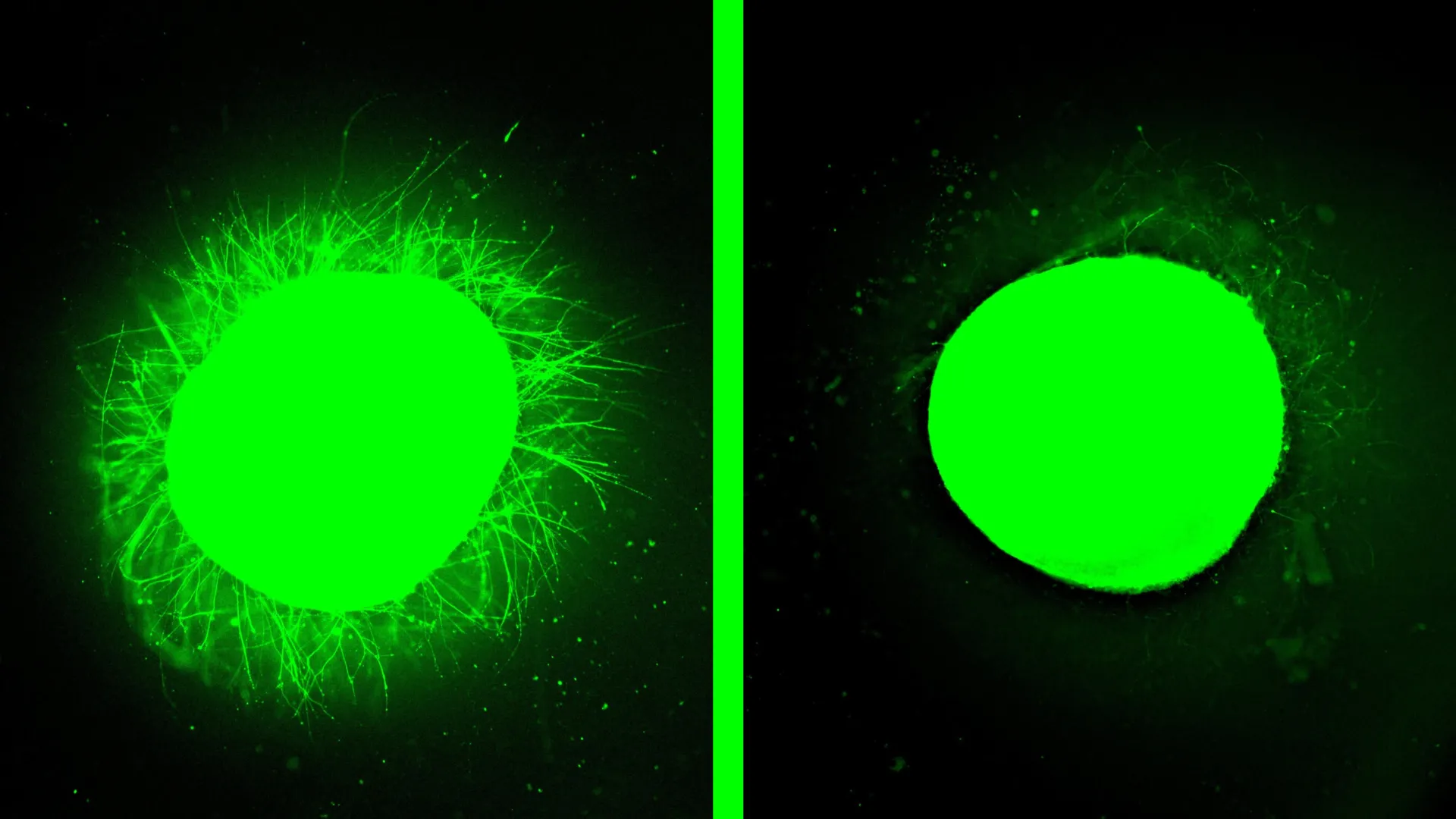
These advanced organoids, measuring several millimeters across, were mature enough to withstand and accurately model traumatic damage. The researchers simulated two common injury types: a direct cut with a scalpel, mimicking surgical lacerations, and a compressive contusion injury, akin to trauma sustained in accidents. Both injury models resulted in observable cell death and the characteristic formation of glial scars, complete with the overproduction of chondroitin sulfate proteoglycans, molecules known to inhibit nerve regeneration.
The Promise of "Dancing Molecules" Therapy
The study then put a promising regenerative therapy, "dancing molecules," to the test within this advanced human organoid model. This therapy, first introduced in 2021, belongs to a class of supramolecular therapeutic peptides (STPs) that leverage the controlled motion of large molecular assemblies to activate cellular receptors and stimulate the body’s innate repair mechanisms.
When applied to the damaged organoids, the results were nothing short of dramatic. The "dancing molecules" therapy significantly reduced the glial scar tissue, making it barely detectable. More importantly, it stimulated substantial neurite outgrowth – the regrowth of axons and dendrites, the vital extensions that enable neurons to communicate. This observed regeneration closely mirrored the positive outcomes previously seen in animal studies, providing powerful validation for the therapy’s potential in humans.
The "dancing molecules" are delivered as a liquid injection. Upon administration, they rapidly self-assemble into a three-dimensional nanofiber scaffold that closely resembles the natural extracellular matrix of the spinal cord. The dynamic movement of these molecules within this scaffold is key to their therapeutic effect. "Given that cells themselves and their receptors are in constant motion, you can imagine that molecules moving more rapidly would encounter these receptors more often," Stupp stated in prior research. "If the molecules are sluggish and not as ‘social,’ they may never come into contact with the cells." This concept of dynamic molecular motion is crucial for effectively interacting with the constantly shifting cellular receptors involved in tissue repair.
A Timeline of Innovation and Validation
The journey leading to this breakthrough began with the foundational work on supramolecular therapeutic peptides. In 2021, initial studies demonstrated the potential of "dancing molecules" in animal models. These early experiments revealed that a single injection administered 24 hours after a severe SCI in mice enabled them to regain mobility within four weeks. Crucially, formulations exhibiting faster molecular motion consistently outperformed slower versions, underscoring the direct correlation between molecular dynamism and enhanced bioactivity and cellular signaling.
Building upon this success, the Northwestern team focused on developing a more accurate human model. The creation of human spinal cord organoids, first explored by various research groups for basic biological studies, was advanced significantly by the Northwestern team’s incorporation of microglia and their ability to engineer mature, injury-susceptible structures. This development paved the way for the current study, which specifically aimed to validate the "dancing molecules" therapy in a human context. The publication in Nature Biomedical Engineering on February 11, 2024, marks the culmination of years of dedicated research and development.
The Significance of Human Organoids in Research
The development of organoids has revolutionized biological research, offering miniature, simplified yet highly representative models of complex organs. Grown from induced pluripotent stem cells, these structures mimic the architecture, cellular diversity, and functional characteristics of their in vivo counterparts. This makes them invaluable tools for studying disease mechanisms, testing therapeutic interventions, and understanding developmental processes.
Compared to traditional methods, organoids offer several distinct advantages. They allow researchers to accelerate the pace of discovery and reduce the costs associated with extensive animal experimentation and the lengthy timelines of human clinical trials. While previous spinal cord organoid models have contributed to fundamental understanding, the Northwestern team’s creation represents a leap forward for injury research due to its maturity, size, and the inclusion of key immune components.
Analysis of Implications and Future Directions
The implications of this research are profound. The ability to accurately model human spinal cord injury in vitro provides an unprecedented opportunity to screen and optimize potential therapies with a much higher degree of confidence. This could significantly reduce the attrition rate of promising treatments in later-stage clinical trials, saving valuable time and resources.
The "dancing molecules" therapy, having recently received Orphan Drug Designation from the U.S. Food and Drug Administration (FDA), is now poised for further investigation. The successful validation in the human spinal cord organoid model strengthens the rationale for advancing this therapy towards human clinical trials. Such trials are the ultimate test, but this pre-clinical data offers a strong signal of hope.
Looking ahead, the Northwestern team plans to engineer even more sophisticated organoid models. Future iterations will aim to replicate chronic SCIs, which are characterized by more established and resistant scar tissue. This will allow for the development and testing of therapies tailored to these more challenging conditions. Furthermore, the researchers envision a future where these miniature spinal cords could contribute to personalized medicine. By generating implantable tissue from a patient’s own stem cells, the risk of immune rejection could be significantly minimized, paving the way for truly individualized regenerative treatments.
Samuel I. Stupp, who also directs the Center for Regenerative Nanomedicine (CRN) at Northwestern, articulated the excitement surrounding this advancement. "One of the most exciting aspects of organoids is that we can use them to test new therapies in human tissue," he stated. "Short of a clinical trial, it’s the only way you can achieve this objective. We decided to develop two different injury models in a human spinal cord organoid and test our therapy to see if the results resembled what we previously saw in the animal model. After applying our therapy, the glial scar faded significantly to become barely detectable, and we saw neurites growing, resembling the axon regeneration we saw in animals. This is validation that our therapy has a good chance of working in humans."
The research was supported by the Center for Regenerative Nanomedicine at Northwestern University and a generous gift from the John Potocsnak Family for spinal cord injury research, underscoring the critical role of philanthropic support in driving scientific innovation in this vital area of medical research.