The landscape of gynecological oncology is facing a potential paradigm shift following a groundbreaking study led by researchers at Nagoya University in Japan. For decades, ovarian cancer has earned a reputation as a "silent killer," characterized by its ability to spread with devastating speed before clinical symptoms appear. Published in the prestigious journal Science Advances, the new research identifies a previously unknown biological mechanism: ovarian cancer cells do not migrate through the abdomen in isolation. Instead, they hijack healthy mesothelial cells to form aggressive hybrid clusters that act as specialized "drilling rigs," facilitating rapid tissue invasion and providing a shield against standard chemotherapy treatments.
Ovarian cancer remains the most lethal of all gynecological malignancies. According to the World Cancer Research Fund, there are more than 300,000 new cases diagnosed globally each year, with a disproportionately high mortality rate compared to breast or cervical cancers. The primary reason for this high fatality rate is the timing of diagnosis. Approximately 70% to 80% of patients are diagnosed at Stage III or IV, after the cancer has already undergone transcoelomic metastasis—a process where cells break off from the primary tumor and spread throughout the peritoneal cavity.
The Mechanism of Transcoelomic Spread
To understand the significance of the Nagoya University discovery, it is essential to look at how ovarian cancer differs from other common malignancies. In cancers such as lung or breast cancer, the primary mode of metastasis is hematogenous or lymphatic. Tumor cells enter the bloodstream or the lymphatic system, which act like highways with predictable routes. This allows clinicians to monitor the spread through blood tests and imaging of specific lymph nodes.
Ovarian cancer, however, largely bypasses the vascular system. Instead, it utilizes the fluid-filled space of the abdominal cavity. When cancer cells detach from the ovaries or fallopian tubes, they enter the peritoneal fluid, also known as ascites. This fluid is in constant, unpredictable motion, driven by the rhythmic movements of the diaphragm during breathing and the peristaltic contractions of the intestines. This "floating phase" has long been a black box for scientists. While it was known that the cancer eventually attaches to the lining of the stomach, liver, and intestines, the specific dynamics of how these floating cells survive and eventually penetrate healthy tissue remained a mystery until now.
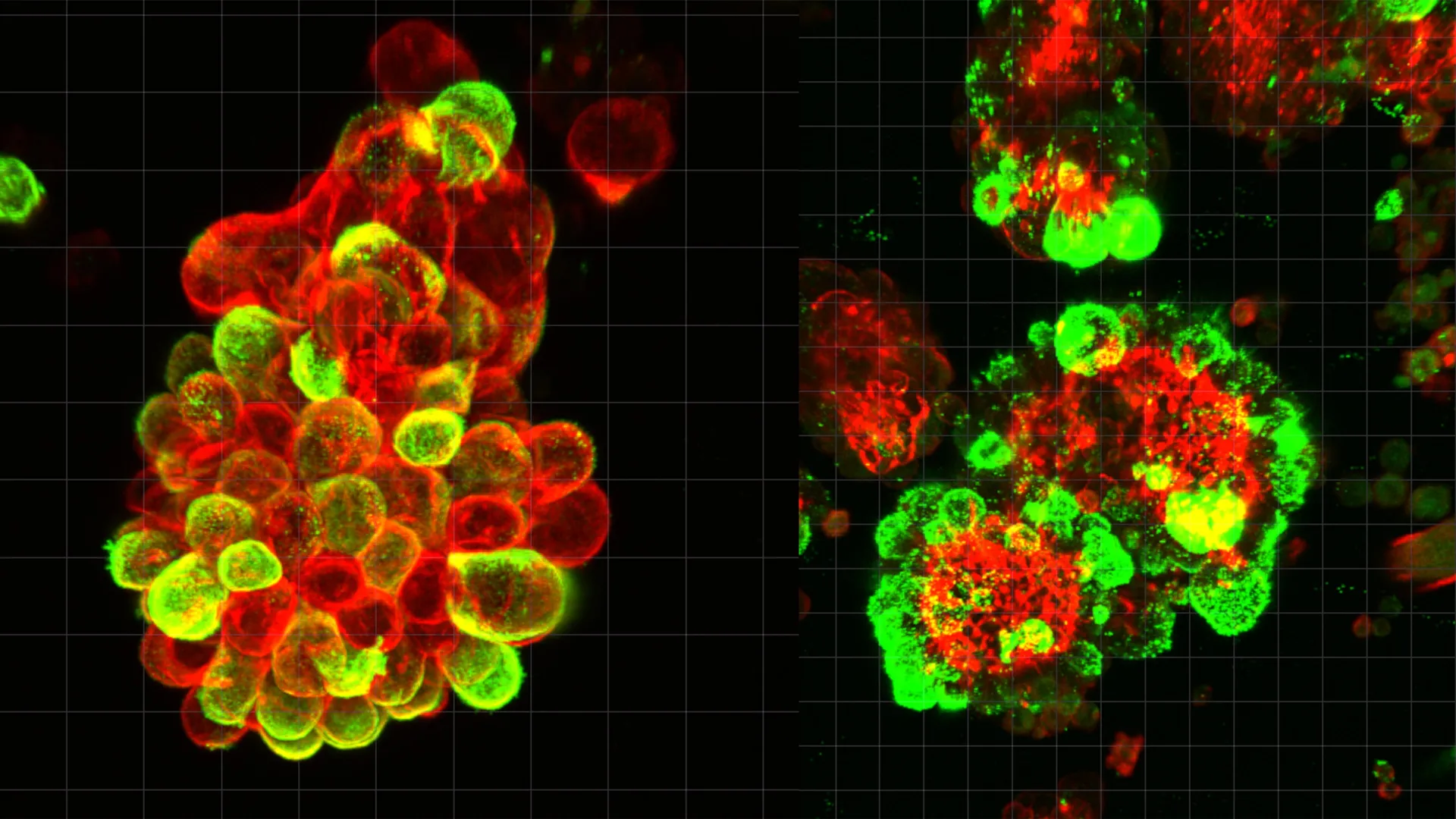
The Discovery of Hybrid Cell Clusters
The research team, led by Dr. Kaname Uno, began their investigation by analyzing abdominal fluid samples taken directly from ovarian cancer patients. Using advanced microscopy and single-cell RNA sequencing, they observed that the cancer cells were rarely alone. Instead, they were found in compact, spherical clusters alongside mesothelial cells.
Mesothelial cells are a vital part of the body’s internal defense system; they form a smooth, protective monolayer that lines the peritoneal cavity and internal organs, allowing them to slide against each other without friction. The Nagoya study revealed a dark irony: the very cells meant to protect the abdominal organs are recruited by the cancer to facilitate its spread.
The data indicates that approximately 60% of the cancer cell spheres found in patient ascites are these hybrid clusters. The process begins when the primary tumor releases a potent signaling protein called Transforming Growth Factor-beta 1 (TGF-β1). This molecule acts as a chemical "instruction," forcing nearby mesothelial cells that have shed into the abdominal fluid to undergo a transformation. Once recruited into the cluster, the mesothelial cells change their physical structure and behavior.
Invadopodia: The Biological Drill
The most striking finding of the study involves the development of "invadopodia." These are specialized, spike-like protrusions that emerge from the hijacked mesothelial cells. While the cancer cells themselves remain relatively passive during the floating phase, the mesothelial cells become the "engine" of the invasion.
"The cancer cells manipulate the mesothelial cells to do the heavy lifting of tissue invasion," explained Dr. Uno. "The cancer cells undergo very few genetic or molecular changes themselves. They simply ride along and migrate through the openings that the mesothelial cells create using these invadopodia."
These spikes function like biological drills, capable of puncturing the protective lining of abdominal organs. Once the hybrid sphere makes contact with an organ, the mesothelial cells lead the charge, breaking through the surface and allowing the cancer cells to take root and form secondary tumors. This collaborative effort explains why ovarian cancer can colonize the entire abdominal cavity with such frightening efficiency.
Chronology of Ovarian Cancer Progression
The research allows for a more detailed timeline of how the disease progresses from a localized tumor to a systemic crisis:
- Primary Tumor Development: The cancer originates in the ovaries or fallopian tubes, often growing unnoticed for months.
- Exfoliation: As the tumor grows, individual cells and small groups break off into the peritoneal cavity.
- Recruitment Phase: The detached cancer cells release TGF-β1 into the surrounding ascites fluid.
- Hybridization: Floating mesothelial cells respond to the TGF-β1 signal and bind to the cancer cells, forming mixed spheres.
- Activation: The mesothelial cells within the sphere develop invadopodia.
- Dissemination: Peritoneal fluid movement carries these activated spheres throughout the abdomen.
- Invasion: Upon landing on an organ, the invadopodia drill into the tissue, establishing a metastatic site.
- Chemoresistance: These spheres create a microenvironment that shields the interior cancer cells from the effects of carboplatin and paclitaxel, the standard chemotherapy drugs used for the disease.
Clinical Background: The Motivation Behind the Research
The study’s lead author, Dr. Kaname Uno, brought a unique clinical perspective to the laboratory. Before becoming a researcher at Nagoya University’s Graduate School of Medicine, Uno spent eight years as a practicing gynecologist. His shift from the clinic to the lab was driven by a specific, tragic case.
One of Dr. Uno’s patients had undergone a routine gynecological screening that showed no signs of abnormality. However, only three months later, she returned with severe symptoms and was diagnosed with advanced-stage ovarian cancer. Despite aggressive treatment, the disease had progressed too far to be contained.
"The fact that current diagnostic tools could not detect anything just weeks before the cancer became terminal was a profound shock," Dr. Uno stated in his reflections on the study. This experience highlighted the inadequacy of current screening methods, such as the CA-125 blood test and transvaginal ultrasounds, which often fail to catch the disease during its "floating" or early metastatic phases.
Supporting Data and Experimental Validation
To ensure the findings were robust, the Nagoya University team employed a multi-faceted experimental approach. Beyond observing patient samples, they utilized mouse models to replicate the spread of the disease. When mice were injected with hybrid clusters, the rate of peritoneal seeding and tumor growth was significantly higher than in mice injected with cancer cells alone.
Furthermore, the team used single-cell transcriptomics to map the gene expression profiles of the cells within the clusters. This confirmed that the mesothelial cells were expressing genes associated with mesenchymal transition and proteolytic activity—essentially the genetic blueprint for invasion. The data also showed that the hybrid clusters were significantly less sensitive to apoptosis (programmed cell death) when exposed to chemotherapy, explaining why many patients experience a recurrence of the disease even after initially successful treatment cycles.
Implications for Future Treatment and Diagnostics
The identification of the TGF-β1 signaling pathway and the role of mesothelial cells offers several new avenues for therapeutic intervention. Current treatments are almost exclusively "cytotoxic," meaning they aim to kill the cancer cells directly. However, if the cancer cells are protected within hybrid spheres and are using mesothelial cells as "shields" and "drills," then cytotoxic drugs may never reach their target effectively.
Future therapies could focus on:
- Pathway Inhibition: Developing drugs that block the TGF-β1 signal, preventing the recruitment of mesothelial cells in the first place.
- Anti-Invadopodia Agents: Targeting the formation of the spike-like structures to prevent the clusters from anchoring to organs.
- Liquid Biopsies of Ascites: Instead of relying solely on blood tests, doctors could analyze small samples of peritoneal fluid to look for these hybrid clusters. This could serve as a highly accurate biomarker for disease progression or to monitor how well a patient is responding to a specific treatment regimen.
Broader Impact on Oncology
This research contributes to a growing body of evidence that cancer is not just a disease of "rogue cells," but a complex system that subverts the body’s own healthy cells to create a supportive "niche." The discovery that mesothelial cells—cells that are supposed to be part of the body’s structural defense—can be turned into accomplices suggests that other types of "protective" cells in different organs might be similarly compromised by other forms of cancer.
As the medical community moves toward personalized medicine, understanding the specific cellular partnerships at play in a patient’s body will be crucial. The work at Nagoya University provides a new roadmap for tackling one of the most challenging obstacles in women’s health, offering hope that the "silent killer" may one day be silenced by targeted, innovative science.