The field of oncology and immunology is witnessing a paradigm shift as researchers at Northwestern University demonstrate that the physical architecture of a vaccine is just as critical as its chemical composition. For over a decade, scientists led by nanotechnology pioneer Chad A. Mirkin have been investigating the nuances of "structural nanomedicine," a discipline that posits the spatial arrangement of antigens and adjuvants at the nanoscale can determine the ultimate efficacy of a therapeutic intervention. Their latest findings, published on February 11 in the journal Science Advances, reveal that subtle adjustments to the orientation of a single cancer-targeting peptide can significantly amplify the immune system’s ability to eradicate tumors, particularly those driven by the human papillomavirus (HPV).
This breakthrough comes at a pivotal time in cancer research. While traditional vaccine development has long focused on identifying the "right" ingredients—the specific proteins or genetic sequences that signal the body to attack—Northwestern’s research suggests that the pharmaceutical industry may have been overlooking a fundamental variable: geometry. By optimizing the way these ingredients are displayed to immune cells, the team has successfully transformed a modest immune response into a potent, tumor-destroying force.
The Science of Spherical Nucleic Acids and Structural Nanomedicine
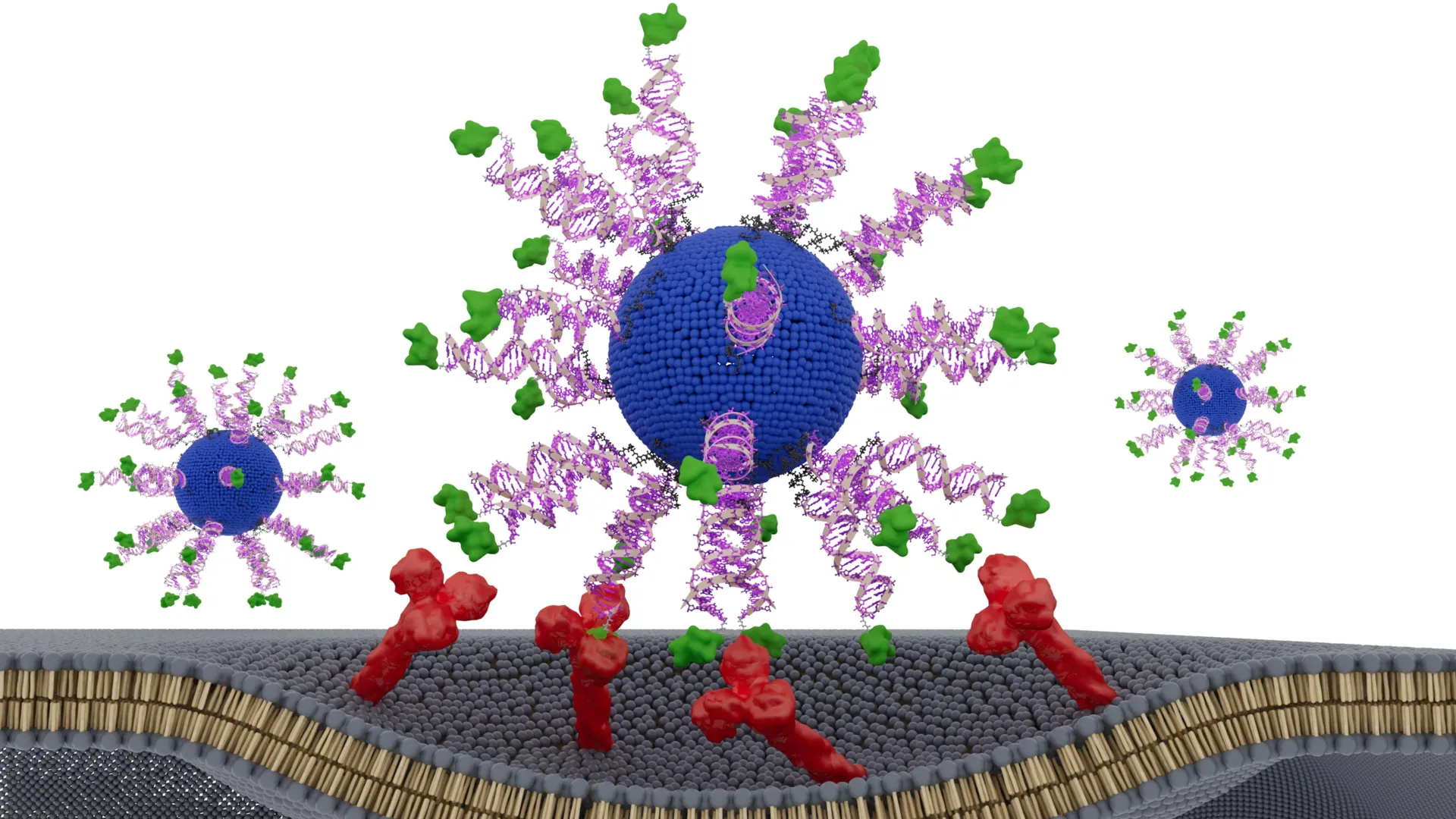
At the heart of this research is the Spherical Nucleic Acid (SNA), a platform technology invented by Chad A. Mirkin. Unlike traditional linear DNA or RNA, SNAs consist of a dense shell of highly organized genetic material arranged in a globular shape around a nanoparticle core. This unique geometry allows SNAs to enter cells more efficiently than their linear counterparts, making them ideal vehicles for delivering immunotherapy.
In the context of cancer vaccines, the SNA acts as a scaffold. It carries both the antigen (the target the immune system should attack) and the adjuvant (the "alarm signal" that activates the immune system). In their recent study, the Northwestern team focused on therapeutic vaccines for HPV-positive cancers. HPV is a major global health concern, linked to nearly all cervical cancers and a rapidly increasing number of head and neck squamous cell carcinomas. While prophylactic vaccines like Gardasil are highly effective at preventing infection, they offer no benefit to patients who have already developed HPV-driven malignancies. Therapeutic vaccines aim to fill this gap by training the body’s CD8+ "killer" T cells to recognize and destroy existing tumor cells.
The researchers discovered that by simply changing where and how a specific HPV-derived peptide was attached to the SNA, they could fundamentally alter the vaccine’s performance. They tested three primary configurations: one where the peptide was sequestered inside the nanoparticle, and two where it was displayed on the surface. For the surface-bound versions, they compared attachment via the N-terminus (one end of the protein chain) versus the C-terminus (the opposite end).
Experimental Findings and Quantitative Success
The results of the study were striking and consistent across multiple models. The configuration that presented the HPV antigen on the surface of the SNA, attached via its N-terminus, outperformed all other designs. This specific arrangement triggered an immune response that was significantly more robust, producing up to eight times more interferon-gamma—a critical signaling protein that coordinates the immune system’s attack on cancer cells.
When tested in "humanized" animal models—mice engineered to have immune systems that mimic human responses—the N-terminus surface-bound vaccine led to a marked reduction in tumor growth and a substantial increase in survival rates. Perhaps more importantly, the researchers validated these findings using actual tumor samples from patients with head and neck cancer. In these human samples, the optimized SNA vaccine increased the killing of cancer cells by a factor of two to three compared to less optimized versions.
"The immune system is incredibly sensitive to the geometry of molecules," explained Dr. Jochen Lorch, a professor of medicine at Northwestern’s Feinberg School of Medicine and co-leader of the study. Lorch, who serves as the medical oncology director of the Head and Neck Cancer Program, noted that the improvement did not stem from higher dosages or more complex ingredients. Instead, it was the result of presenting the same components in a "smarter" way, allowing immune cells to process the antigen more efficiently and mount a more focused counterattack.
Moving Beyond the Blender Approach in Vaccine Design
For decades, the standard protocol for vaccine formulation has been what Mirkin describes as the "blender approach." In this traditional method, antigens and adjuvants are mixed together in a vial, resulting in a formulation where the components are randomly distributed. While this has been effective for many infectious diseases, it often falls short in the complex environment of oncology, where tumors employ sophisticated mechanisms to evade immune detection.
Mirkin points to the current generation of mRNA COVID-19 vaccines as an example of highly successful but structurally heterogeneous medicines. "No two particles are exactly the same in those formulations," Mirkin observed. "While they are impressive and extremely useful, we can do better. To create the most effective cancer vaccines, we will have to move toward precision at the nanoscale."
Structural nanomedicine represents the "bottom-up" construction of medicines. By controlling the exact placement of every atom and molecule within a nanoparticle, researchers can ensure that every dose is identical and optimized for maximum efficacy. This level of precision minimizes "off-target" effects, potentially reducing the toxicity and side effects often associated with aggressive immunotherapies.
A Growing Portfolio of SNA-Based Therapeutics
The recent HPV study is the latest in a series of successes for the Northwestern team. Using the principles of structural nanomedicine, they have already developed SNA vaccine candidates for several other difficult-to-treat cancers, including:
- Melanoma: Early studies showed that structured SNAs could induce regression in advanced skin cancer models.
- Triple-Negative Breast Cancer: One of the most aggressive forms of breast cancer, which currently lacks targeted therapies.
- Prostate and Colon Cancers: Research is ongoing to identify the optimal structural configurations for these common malignancies.
- Merkel Cell Carcinoma: A rare but deadly skin cancer where SNA vaccines have shown preclinical promise.
The impact of this technology extends beyond the laboratory. Currently, seven SNA-based drugs are in various stages of human clinical trials for a range of diseases. Furthermore, the underlying technology has been integrated into more than 1,000 commercial products, demonstrating the scalability and versatility of the SNA platform.
The Role of Artificial Intelligence in Future Development
As the complexity of vaccine design increases, Mirkin anticipates that artificial intelligence (AI) and machine learning will play a central role in identifying the "ideal" structure for new medicines. There are thousands of variables involved in the construction of an SNA—ranging from the density of the DNA shell to the specific chemical linkers used to attach antigens.
"The promise of structural nanomedicine is being able to identify from the myriad possibilities the configurations that lead to the greatest efficacy and least toxicity," Mirkin said. AI can process vast datasets from previous experiments to predict which structural combinations are most likely to succeed, significantly shortening the timeline for drug development and reducing the costs associated with failed clinical trials.
This data-driven approach could also breathe new life into "failed" drug candidates. Mirkin suggests that many previous vaccine trials may have failed not because the ingredients were ineffective, but because they were delivered in a sub-optimal configuration. By restructuring these existing components using SNA technology, scientists may be able to transform once-discarded molecules into potent therapeutic agents.
Broader Implications and Economic Impact
The shift toward structural nanomedicine has profound implications for the global pharmaceutical industry. As the cost of developing new drugs continues to skyrocket—often exceeding $2 billion per successful drug—the ability to improve efficacy through structural optimization rather than the discovery of entirely new chemical entities offers a more sustainable path forward.
Furthermore, the precision of SNAs could lead to more "personalized" cancer vaccines. By taking a biopsy of a patient’s tumor, identifying the specific mutations (neoantigens) present, and rapidly incorporating those into a structurally optimized SNA, clinicians could provide a bespoke treatment tailored to the individual’s unique cancer profile.
The research was a collaborative effort involving several prominent institutions and was supported by the National Cancer Institute, the Lefkofsky Family Foundation, and the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. As the team moves forward, their focus remains on refining the SNA platform and expanding its application to a wider array of infectious diseases and autoimmune disorders.
Conclusion: A New Era of Precision Immunotherapy
The findings from Northwestern University underscore a fundamental truth in biology: form dictates function. By proving that the orientation and position of a single peptide can be the difference between a failing vaccine and a life-saving treatment, Mirkin and his colleagues have provided a roadmap for the next generation of immunotherapy.
"The whole concept of structural nanomedicines is a major train roaring down the tracks," Mirkin concluded. "We have shown that structure matters—consistently and without exception." As the medical community moves away from the "blender approach" and toward the precision of structural nanomedicine, the prospect of effective, low-toxicity therapeutic vaccines for cancer moves closer to reality. The transition from random mixtures to "bottom-up" engineered medicines marks the beginning of a new era in which the architecture of a vaccine is as carefully designed as the medicine itself.