The Evolution of Structural Nanomedicine at Northwestern
The study represents the culmination of years of rigorous investigation into the architecture of vaccines. For decades, the pharmaceutical industry has relied on what Dr. Mirkin calls the "blender approach" to vaccine formulation. In this traditional model, antigens—the molecules that teach the immune system what to attack—and adjuvants—the compounds that stimulate the immune response—are mixed together in a liquid medium. While this method has been effective for many infectious diseases, it often falls short in the complex environment of oncology, where tumors employ sophisticated mechanisms to evade immune detection.
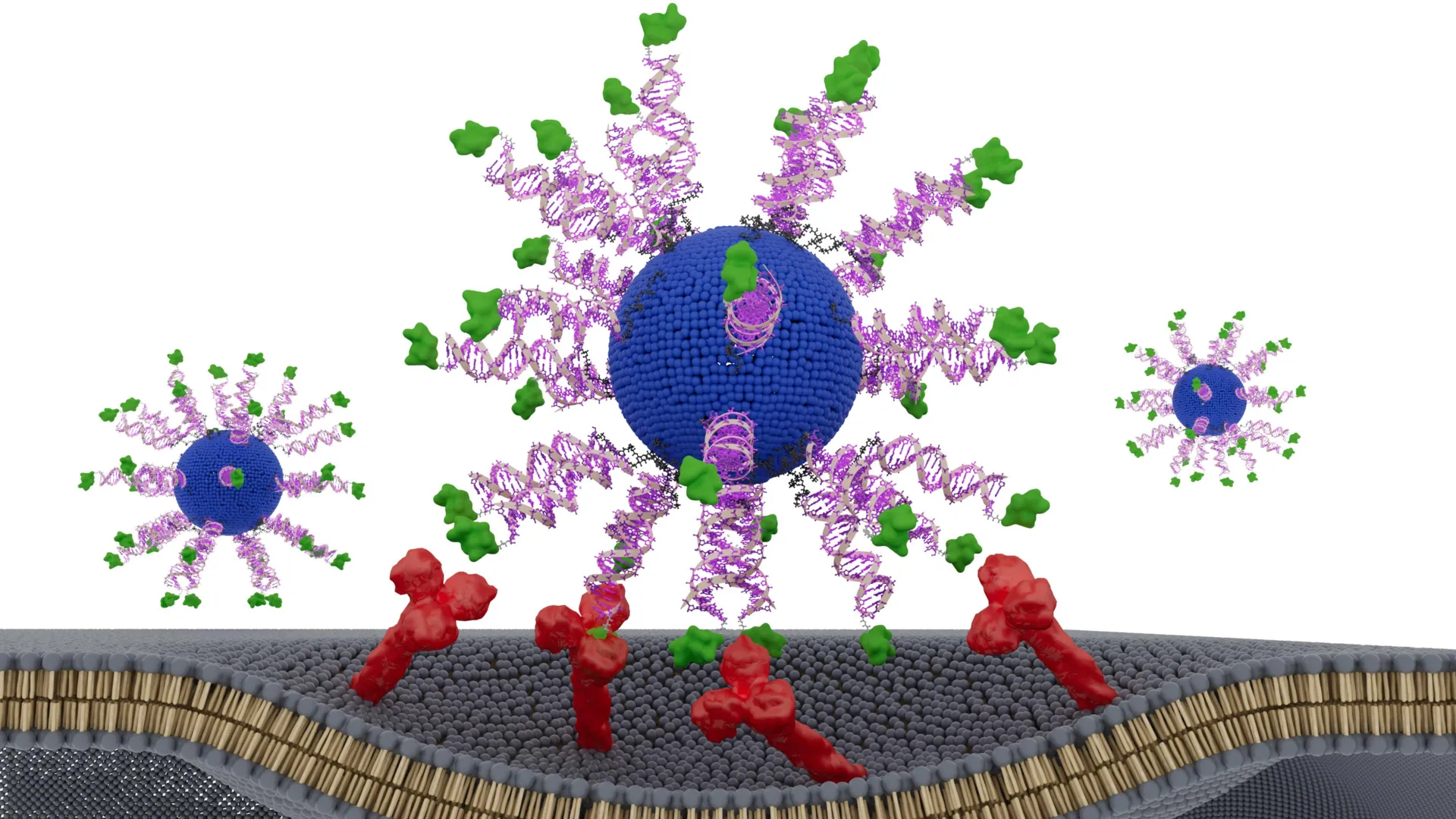
The Northwestern team’s breakthrough rests on the development of Spherical Nucleic Acids (SNAs). Invented by Mirkin, SNAs are globular structures consisting of a dense shell of DNA or RNA strands organized around a central core. Unlike linear genetic material, which is often degraded by the body before it can reach its target, the 3D structure of SNAs allows them to naturally enter immune cells and trigger a robust activation. This unique geometry serves as a programmable scaffold, allowing scientists to place different medicinal components with atomic-level precision.
Addressing the Global Burden of HPV-Positive Cancers
While the primary focus of the study was the structural optimization of vaccines, the practical application targeted one of the most significant viral contributors to global cancer rates: human papillomavirus (HPV). HPV is the most common sexually transmitted infection in the world and is the primary cause of nearly all cervical cancers. Furthermore, the medical community has observed a sharp increase in the incidence of HPV-positive head and neck cancers, particularly oropharyngeal squamous cell carcinoma.
Current preventive vaccines, such as Gardasil 9, are highly effective at preventing initial infection. However, they offer no therapeutic benefit to the millions of individuals who already harbor HPV-driven malignancies. For these patients, the immune system has already failed to clear the virus, and the resulting tumors are often resistant to standard therapies. The Northwestern study sought to bridge this gap by developing a therapeutic vaccine capable of "re-training" the immune system—specifically CD8+ "killer" T cells—to recognize and destroy existing HPV-positive cells.
Methodology: Comparing Geometric Configurations
To determine the impact of structure on vaccine performance, the research team engineered three distinct versions of an SNA-based vaccine. Each version utilized the exact same chemical composition: a lipid core, immune-stimulating DNA sequences (adjuvants), and a specific peptide derived from the HPV E7 protein (the antigen). The only variable was the physical placement and orientation of the E7 peptide.
The three designs tested were:
- Internalized Peptide: The antigen was sequestered within the core of the nanoparticle, hidden from immediate cellular recognition.
- Surface-Exposed (C-terminus): The antigen was attached to the surface of the SNA via its C-terminus, one end of the protein chain.
- Surface-Exposed (N-terminus): The antigen was attached to the surface via its N-terminus, the opposite end of the protein chain.
The team evaluated these configurations using humanized animal models—mice engineered to possess human-like immune components—and primary tumor samples harvested from patients at the Northwestern Medicine Head and Neck Cancer Program.
Quantifying the Results: A Breakthrough in Potency
The experimental data revealed a stark contrast in performance based solely on the peptide’s orientation. The version of the vaccine that presented the antigen on the surface via its N-terminus outperformed the other configurations across every metric.
Specifically, the N-terminus surface vaccine triggered a surge in the production of interferon-gamma, a critical signaling protein that coordinates the body’s attack on cancer. Compared to the other designs, this configuration produced up to eight times more interferon-gamma. This increase in signaling translated directly to cellular performance; the CD8+ T cells generated by this specific vaccine were significantly more aggressive in seeking out and lysing HPV-positive tumor cells.
In animal trials, the N-terminus vaccine led to a marked reduction in tumor volume and a significant extension of survival rates. Perhaps more importantly, the tests conducted on actual human patient samples showed a twofold to threefold increase in cancer cell killing capacity. These results underscore the sensitivity of the immune system to molecular geometry. As Dr. Jochen Lorch, the medical oncology director of the Head and Neck Cancer Program at Northwestern Medicine, noted, the immune system does not just see ingredients; it "reads" the shape and accessibility of those ingredients.
Moving Beyond the "Blender Approach"
The implications of this study extend far beyond HPV. By proving that the structural organization of a nanovaccine can determine its success or failure, the Northwestern team has challenged the status quo of vaccine manufacturing.
"The COVID-19 vaccines are a beautiful example—no two particles are the same," Mirkin stated during the announcement of the findings. "While very impressive and extremely useful, we can do better." Mirkin’s critique of the current state of "less structured medicines" highlights a major opportunity for the pharmaceutical industry. If existing vaccines can be redesigned with structural precision, they could potentially achieve higher efficacy with lower dosages, thereby reducing the risk of side effects and toxicity.
The Northwestern International Institute for Nanotechnology has already begun applying this structural logic to a wide array of other cancers. Preclinical studies are currently underway for SNA vaccines targeting:
- Melanoma
- Triple-negative breast cancer
- Prostate cancer
- Colon cancer
- Merkel cell carcinoma
To date, seven different SNA-based drugs have moved into human clinical trials, and the technology has been integrated into over 1,000 commercial products, signaling a rapid transition from laboratory discovery to industrial application.
The Role of Artificial Intelligence in Future Vaccine Design
As the field of structural nanomedicine grows, the number of possible molecular configurations becomes nearly infinite. With thousands of variables—including particle size, DNA density, peptide orientation, and core composition—manually testing every iteration is impossible.
Mirkin envisions a future where artificial intelligence (AI) and machine learning become the primary architects of vaccine design. By feeding data from studies like this one into AI models, researchers can predict which structural arrangements will yield the most potent immune responses before a single nanoparticle is synthesized in the lab. This "bottom-up" approach to medicine would allow for the rapid development of personalized cancer vaccines tailored to the specific molecular signatures of an individual’s tumor.
Analysis: Resurrecting Failed Clinical Candidates
One of the most profound implications of the Northwestern study is the potential to revisit previously "failed" vaccine candidates. In the history of oncology, many therapeutic vaccines have shown promise in early stages but failed to meet clinical endpoints in Phase II or Phase III trials.
The research suggests that many of these failures may not have been due to the ingredients themselves, but rather their haphazard delivery. By restructuring these existing components into optimized SNA configurations, scientists may be able to "rescue" billions of dollars in research and development and bring life-saving treatments to market that were previously discarded.
Conclusion and Institutional Support
The study, titled "E711-19 placement and orientation dictate CD8+ T cell response in structurally defined spherical nucleic acid vaccines," was a collaborative effort involving the Weinberg College of Arts and Sciences, the McCormick School of Engineering, and the Northwestern University Feinberg School of Medicine.
The research received significant backing from the National Cancer Institute, the Lefkofsky Family Foundation, and the Robert H. Lurie Comprehensive Cancer Center. As the medical community moves toward more precise and personalized therapies, the principles of structural nanomedicine established at Northwestern are likely to serve as the foundation for the next generation of immunotherapy. The transition from the "blender approach" to a blueprint-based approach marks a new era in the fight against cancer—one where the architecture of the cure is just as important as the cure itself.