A groundbreaking study, published in the prestigious journal Nature Communications, is shedding new light on a persistent and perplexing challenge in cancer therapy: why the same drug can yield dramatically different results for different patients. Researchers at the MRC Laboratory of Medical Sciences (LMS), led by Dr. Louise Fets, have meticulously investigated the behavior of PARP inhibitors, a vital class of targeted therapies, within ovarian tumor samples. Their findings pinpoint cellular compartments known as lysosomes as unexpected and significant reservoirs, profoundly influencing drug efficacy and patient outcomes.
The Enigma of Differential Drug Response in Oncology
The advent of targeted cancer therapies has revolutionized oncology in recent decades, offering hope and improved survival rates for countless individuals. Among these advancements, PARP inhibitors have emerged as a transformative treatment for ovarian cancer, demonstrating remarkable efficacy in a significant subset of patients. However, the clinical reality is often more complex. A substantial portion of patients either do not respond to these drugs or develop resistance over time, leading to disease recurrence. The fundamental requirement for these targeted agents to be effective is their ability to accumulate within cancer cells at concentrations sufficient to induce cell death. Yet, the intricate mechanisms governing drug distribution within the complex tumor microenvironment and at the cellular level have remained a significant knowledge gap for scientists.
This pioneering research directly addresses this deficit by demonstrating that the success of a cancer drug hinges not solely on its initial delivery to the tumor, but critically on its subsequent distribution and accumulation within the tumor mass and, crucially, within individual cancer cells. To meticulously examine this process, the research team utilized an innovative approach involving ex vivo tumor explants. These are thin slices of ovarian tumors, carefully harvested from patients and maintained in a viable state in the laboratory. By exposing these living tumor samples to PARP inhibitors, scientists could directly observe and quantify the drug’s movement through authentic human tumor tissue in real-time.
Advanced Imaging Techniques Illuminate Drug Distribution
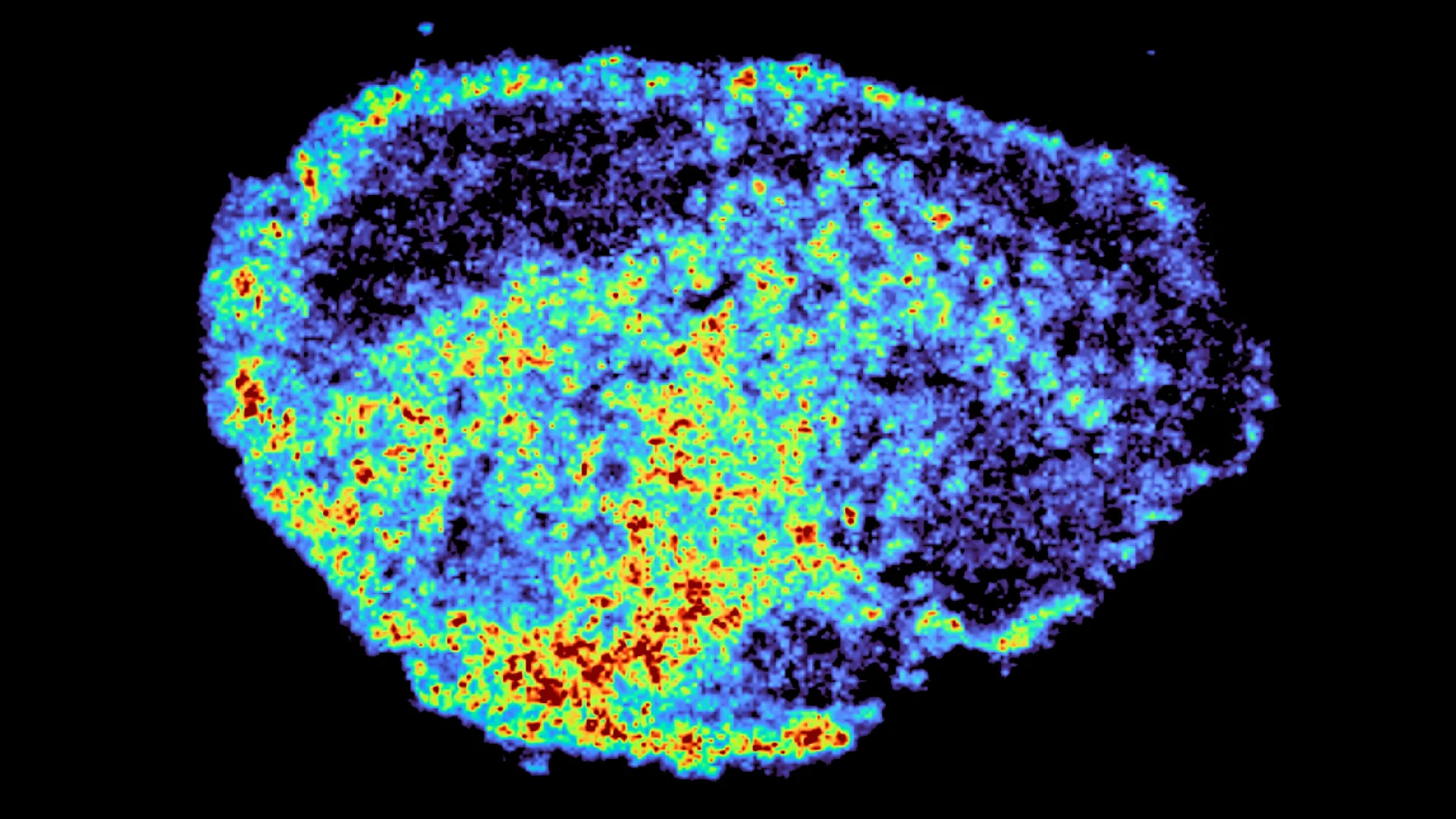
The study employed a powerful combination of cutting-edge techniques to map drug penetration with unprecedented detail. Mass spectrometry imaging (MSI) was instrumental in generating high-resolution maps that precisely identified the locations and concentrations of drug molecules within the tumor slices. This spatial information was then integrated with spatial transcriptomics, a technology that simultaneously analyzes gene expression patterns within the same tissue sections. This dual approach allowed researchers to correlate areas of high and low drug accumulation with specific cellular activities and genetic profiles, revealing intricate relationships between drug presence and tumor biology.
"A novel aspect of this study was the use of mass spectrometry imaging to directly measure and visualize drug uptake in patient tumour tissue," stated Dr. Zoe Hall, a senior author on the study and Associate Professor at Imperial’s Department of Metabolism, Digestion and Reproduction. "Through the spatial mapping of drug molecules, we could pinpoint regions of high and low drug and compare gene expression, from the same tissue slice, using spatial transcriptomics." The results were striking, showcasing significant variability in drug distribution not only between different tumors but also between individual patients, even when identical drug doses were administered. This heterogeneity underscores the inherent complexity of drug behavior within the tumor landscape.
Lysosomes: The Unseen Architects of Drug Accumulation
The study’s most compelling revelation centers on the unexpected role of lysosomes. These ubiquitous organelles, often described as the cell’s "recycling centers," were found to actively sequester and retain certain PARP inhibitors. Instead of dispersing evenly throughout the cell, these drugs were observed to accumulate within lysosomes, creating intracellular pockets of high drug concentration.
This lysosomal sequestration transforms these organelles into dynamic, slow-release reservoirs. They effectively "hold onto" the drug, gradually releasing it over time. This process leads to differential drug exposure across the cellular population within a tumor – some cells experience prolonged and higher drug exposure, while others are exposed to significantly lower levels. The study further elucidated that this lysosomal trapping is not a universal phenomenon for all PARP inhibitors. Specifically, drugs like rucaparib and niraparib were found to be significantly influenced by this lysosomal mechanism, whereas olaparib appeared to be less affected.
"We were surprised to see large variability in drug accumulation at the single-cell level," commented Dr. Carmen Ramirez Moncayo, the study’s first author and a Postdoctoral Researcher at the LMS. "This variability was driven by the build-up of a drug in lysosomes, which are acting as reservoirs, increasing the exposure of cancer cells to drugs, by storing and releasing the drug when needed." This insight provides a tangible explanation for why some cancer cells might be more effectively targeted by certain PARP inhibitors than others, even within the same tumor.
Implications for Personalized Cancer Therapy
The implications of this research for the future of cancer treatment are profound. PARP inhibitors are currently a cornerstone in the management of ovarian, breast, and prostate cancers, and their therapeutic potential is being actively explored in a growing number of other cancer types. A deeper understanding of the cellular mechanisms governing drug storage and distribution, such as the lysosomal sequestration identified in this study, opens the door to more sophisticated and personalized treatment strategies.
By unraveling these intricate cellular processes, clinicians may be able to predict which patients are most likely to benefit from specific PARP inhibitors and identify potential strategies to overcome or prevent treatment resistance and subsequent relapse. "By understanding how drugs are taken up into cells, we can understand whether this influences why cancer drugs work for some people and not for others," explained Dr. Louise Fets, a senior author and Head of the LMS’ Drug Transport and Tumour Metabolism Group. "Eventually, we hope to be able study the molecular signature of a patient’s tumor to help to tailor therapeutic approaches in a more personalized way."
This personalized approach could involve stratifying patients based on their tumor’s lysosomal activity or exploring combination therapies that might enhance drug release from lysosomes or bypass this sequestration mechanism altogether. The development of predictive biomarkers that indicate a tumor’s propensity for lysosomal drug trapping could become a critical tool in optimizing treatment selection.
Bridging the Gap to Clinical Application
It is important to acknowledge that this foundational research was conducted using tumor tissue maintained outside the body. In a living patient, drug delivery is mediated by the bloodstream, and the often disorganized vascular networks within tumors can introduce further complexities to drug distribution. These anomalies in tumor vasculature can exacerbate uneven drug penetration, potentially compounding the effects of intracellular sequestration.
Therefore, the next critical steps in this research trajectory involve translating these findings into more clinically relevant models. Future studies will leverage animal models and expand investigations to larger patient cohorts. These efforts will aim to elucidate the intricate interplay between systemic drug delivery, the physical structure of the tumor microenvironment, and the intracellular lysosomal storage mechanisms in real-world clinical settings. Furthermore, understanding these processes in the context of relapsed cancers, where treatment resistance is often a significant hurdle, will be paramount.
The research was made possible through substantial funding from several key organizations, including the Medical Research Council and Cancer Research UK. Additional support was provided by a PhD studentship from the Integrative Toxicology Training Partnership, administered by the MRC Toxicology Unit, and a Victoria’s Secret Global Fund for Women’s Cancers Career Development Award, in partnership with Pelotonia and the American Association for Cancer Research (AACR). This collaborative funding landscape underscores the critical importance and broad recognition of this research area within the global oncology community. By unraveling the complex journey of cancer drugs within the tumor, this study represents a significant leap forward in the quest for more effective and personalized cancer therapies.