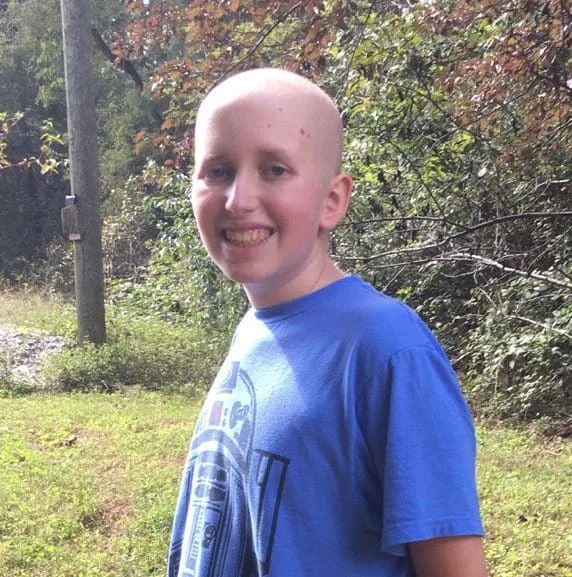
The reality of pediatric oncology remains one of the most significant challenges in modern medicine, characterized by a dichotomy between high survival rates in some leukemias and devastatingly poor outcomes in others. For the family of Luke Getchell, a 13-year-old diagnosed in July 2018, this reality was defined by a diagnosis of acute myeloid leukemia (AML) complicated by a FLT3-ITD genetic alteration. This specific mutation shifted his prognosis from a standard clinical challenge to a high-risk battle against a highly aggressive form of blood cancer. Despite the rigor of modern protocols, Luke’s 14-month journey serves as a poignant case study in the limitations of current cytotoxic therapies and the critical necessity for targeted, less toxic research initiatives.
Clinical Overview of Acute Myeloid Leukemia and the FLT3-ITD Mutation
Acute myeloid leukemia is a cancer of the blood and bone marrow characterized by the rapid overproduction of abnormal myeloblasts. While it is less common than acute lymphoblastic leukemia (ALL) in children, it is significantly more difficult to treat. Luke’s diagnosis was further complicated by the presence of a FLT3-ITD (Fms-like tyrosine kinase 3 – internal tandem duplication) mutation.
In a healthy biological system, the FLT3 gene plays a vital role in the proliferation and survival of hematopoietic stem cells. However, when an internal tandem duplication occurs, the FLT3 receptor is constitutively activated—meaning it is permanently "switched on." This leads to the uncontrolled division of leukemia cells and a significantly higher risk of relapse. Historically, pediatric patients with FLT3-ITD mutations face a lower five-year survival rate compared to those without the mutation, often necessitating more intensive interventions, including bone marrow transplants and high-dose chemotherapy.
The Patient Profile: A Life Beyond the Diagnosis
Before his diagnosis, Luke Getchell was known in his Richmond, Virginia, community for his empathetic nature and diverse interests. A dedicated enthusiast of Lego building, Luke utilized the medium as both a creative outlet and a cognitive distraction during his subsequent hospitalizations. His passion for the culinary arts led him to host "Chopped" style competitions in his family kitchen, showcasing a sophisticated palate and a flair for gastronomy.
Following his passing, Luke’s family and the medical community at VCU Health established "Luke’s Lego-cy," a permanent display of his Lego models located at the Children’s Wonder Tower at VCU Health. This installation serves not only as a memorial but as a reminder of the children behind the clinical statistics, humanizing the sterile environment of a pediatric oncology ward.
A Timeline of Treatment and Clinical Complications
Luke’s medical journey began in the summer of 2018 and spanned over a year of intensive inpatient and outpatient care. The standard of care for AML involves aggressive induction chemotherapy designed to clear the blood and bone marrow of leukemic blasts.
July 2018 – January 2019: Induction and Consolidation
During the initial six months of treatment, Luke underwent multiple rounds of high-dose chemotherapy. While these drugs are designed to target rapidly dividing cancer cells, they are non-specific and cause significant systemic damage. Luke experienced a series of severe secondary health complications, including:
- Sepsis: A life-threatening systemic inflammatory response to infection, common in immunocompromised oncology patients.
- Colon Infection: Resulting from the destruction of the mucosal lining of the gastrointestinal tract.
- Cardiotoxicity: Permanent heart damage, a known long-term side effect of certain anthracycline chemotherapies used in AML protocols.
February 2019: The Remission and Immediate Relapse
By early 2019, clinical tests indicated that Luke had reached a state of no detectable leukemia. This period of remission, however, lasted only one week. In FLT3-ITD cases, "minimal residual disease" (MRD) often persists below the threshold of standard detection, allowing the cancer to rebound with increased resistance to previous therapies. When Luke’s blood counts began to fluctuate just seven days later, subsequent testing confirmed a rapid and aggressive relapse.
March 2019 – October 2019: Terminal Progression
The post-relapse phase was marked by a decline in physiological stability. Luke’s body struggled with the cumulative toxicity of his treatments and the progression of the disease. His clinical profile during this time included:
- Graft-versus-Host Disease (GVHD): A condition where donated stem cells or bone marrow attack the recipient’s body.
- Secondary Diabetes: Induced by high doses of prednisone and other corticosteroids used to manage inflammation and GVHD.
- Physical Decline: By September 2019, the malignancy and treatment side effects resulted in the loss of vision and the inability to walk.
On October 20, 2019, at the age of 14, Luke Getchell died from complications related to his disease and its treatment.
The Systemic Impact of Treatment Toxicity
The Getchell family’s experience highlights a harrowing truth in pediatric oncology: the "cure" is often nearly as destructive as the disease. The family described the trauma of witnessing their child suffer from bleeding of the eyes, severe thrush, and the psychological burden of administering oral medications that represent a final, desperate hope for survival.
Current treatments for AML rely heavily on decades-old cytotoxic frameworks. While these protocols have saved many lives, they often leave survivors with lifelong "late effects," including secondary cancers, organ failure, and cognitive impairments. For high-risk patients like Luke, the narrow therapeutic window between efficacy and fatal toxicity is a constant challenge for oncologists.
National Funding and the Pediatric Research Gap
The struggle to find better treatments for children like Luke is exacerbated by a significant disparity in research funding. According to data from the National Cancer Institute (NCI), only a small fraction of the federal budget for cancer research is dedicated specifically to pediatric cancers. Most funding is directed toward adult cancers, such as breast, lung, and prostate cancer, which have larger patient populations.
This funding gap forces pediatric oncologists to rely on "off-label" use of adult medications or older protocols that may not be optimized for a child’s developing physiology. Organizations like CureSearch for Children’s Cancer aim to bridge this gap by funding targeted research that focuses specifically on the molecular drivers of childhood malignancies.
Fueling Future Breakthroughs: The Work of Dr. Elvin Wagenblast
In response to the limitations of current therapies, Childhood Cancer Awareness Month initiatives are focusing on raising funds for innovative research, such as that conducted by Dr. Elvin Wagenblast at Mount Sinai. Dr. Wagenblast is a recipient of the CureSearch Young Investigator Program, which supports early-career scientists dedicated to pediatric oncology.
Dr. Wagenblast’s research utilizes advanced gene-editing technologies, including CRISPR-Cas9, to model the earliest stages of leukemia development in human blood stem cells. By understanding the "how" and "why" of leukemia initiation—specifically in the context of genetic mutations like FLT3-ITD—his team aims to:
- Identify specific vulnerabilities in leukemia cells that can be targeted without harming healthy tissue.
- Develop precision medicines that reduce the reliance on broad-spectrum, toxic chemotherapy.
- Uncover why certain leukemias are resistant to standard treatments, potentially preventing relapses like the one Luke experienced.
The current goal of raising $25,000 for this research is part of a broader effort to ensure that the next generation of children diagnosed with AML has access to therapies that are both more effective and less debilitating.
Broader Implications and the Path Forward
The story of Luke Getchell is a call to action for the medical community, policymakers, and the public. It underscores the fact that "survival" should not be the only metric of success; the quality of life during and after treatment is equally paramount. The shift toward precision medicine—tailoring treatment to the genetic profile of the individual patient’s tumor—offers the best hope for achieving this balance.
The implications of failing to fund this research are clear: families will continue to face the "stomach-turning reality" of watching their children endure archaic treatments. However, through the support of investigators like Dr. Wagenblast and the preservation of legacies like Luke’s, the trajectory of pediatric oncology can be altered.
As Childhood Cancer Awareness Month continues, the focus remains on the urgent need for innovation. The memory of Luke Getchell, his Legos, and his culinary spirit serve as a catalyst for a future where a diagnosis of FLT3-ITD AML is no longer a grim prognosis, but a manageable condition met with targeted, humane, and effective medical intervention. Making a gift in Luke’s memory is more than a gesture of sympathy; it is an investment in the scientific breakthroughs that will define the future of pediatric survival.