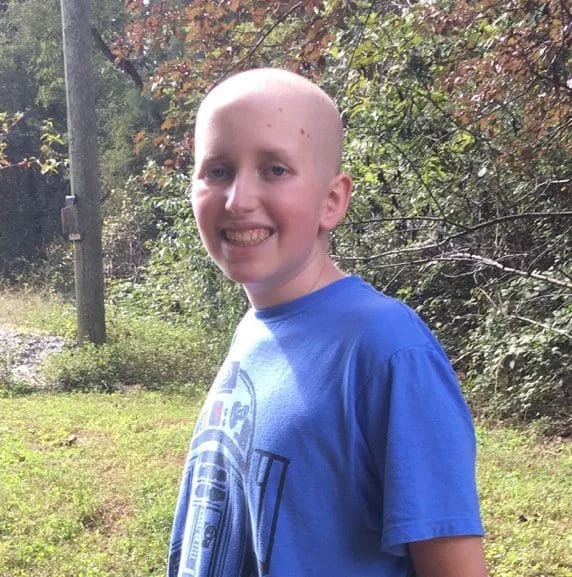
The diagnosis of childhood cancer represents a profound shift in the trajectory of a family’s life, transforming a standard upbringing into a high-stakes medical odyssey. For the family of Luke Getchell, this reality began in July 2018 when the 13-year-old was diagnosed with acute myeloid leukemia (AML). The severity of the diagnosis was compounded by the presence of a specific genetic alteration known as FLT3-ITD (FMS-like tyrosine kinase 3 with internal tandem duplication). In the field of pediatric oncology, this marker is recognized as a harbinger of a poor prognosis, indicating an aggressive form of the disease that is historically resistant to conventional therapies and prone to early relapse.
Luke Getchell was characterized by those who knew him as an empathetic and kind teenager with a passion for creative expression and culinary arts. A dedicated enthusiast of Lego building, his intricate models served as a testament to his patience and focus. Today, many of these creations are preserved in the "Luke’s Lego-cy" display within the Children’s Wonder Tower at VCU Health in Richmond, Virginia. Beyond his hobbies, Luke was known for his love of penguins and his "Chopped" style cooking competitions hosted in his family’s kitchen. However, these youthful pursuits were largely eclipsed by a fourteen-month battle against a malignancy that tested the limits of modern medical intervention.
A Chronology of Treatment and Clinical Complications
The clinical journey for Luke Getchell began immediately following his July 2018 diagnosis. For over a year, he and his family navigated the complexities of the inpatient environment, where he was subjected to intensive chemotherapy regimens. While these treatments are designed to eradicate leukemic cells, their high levels of toxicity often result in significant collateral damage to healthy organ systems.
Throughout his treatment, Luke suffered from a series of secondary health crises precipitated by the very medications intended to save him. These complications included a severe colon infection, bouts of sepsis—a life-threatening systemic response to infection—and permanent heart damage, a common side effect of anthracycline-based chemotherapies used in AML protocols. Despite these harrowing setbacks, there was a brief window of clinical optimism. In early 2019, approximately six months after the initiation of treatment, diagnostic tests indicated no detectable signs of leukemia.
This period of remission offered the Getchell family a temporary reprieve. However, the respite lasted only one week. Follow-up blood work revealed a rapid rise in cell counts, prompting immediate hospitalization and further testing. The results confirmed a relapse, with the cancer spreading more aggressively than before. The medical team was forced to deliver a grim prognosis, as the options for salvage therapy in the context of a FLT3-ITD relapse are limited and often involve even higher levels of toxicity.
The Physical Toll of Relapse and Palliative Transition
The secondary phase of Luke’s illness was marked by a rapid physiological decline. Following the relapse, his body faced a constellation of complications, including internal bleeding, severe dermatological issues, and graft-versus-host disease (GVHD)—a condition where donor immune cells attack the recipient’s body, often following a bone marrow transplant. Additionally, the high doses of prednisone required to manage inflammation led to the development of secondary diabetes.
By September 2019, fourteen months after the initial diagnosis, the oncology team at VCU Health recommended a transition to hospice care. This decision marked the shift from curative intent to a focus on comfort and quality of life. The decline in the final weeks was precipitous; Luke lost his mobility and his vision. On October 20, 2019, at the age of 14, he passed away. His experience highlights the brutal reality of pediatric AML, where the treatment can be as devastating as the disease itself, sometimes leading to ocular hemorrhaging and systemic organ failure.
Understanding Pediatric AML and the FLT3-ITD Mutation
Acute myeloid leukemia is a cancer of the blood and bone marrow characterized by the overproduction of immature white blood cells (blasts). While leukemia is the most common form of childhood cancer, AML accounts for only about 20% of pediatric leukemia cases, yet it is responsible for a disproportionate number of deaths due to its aggressive nature.
The FLT3-ITD mutation found in Luke’s case is a significant focus of current oncological research. FLT3 is a gene that provides instructions for making a protein that spans the cell membrane, playing a critical role in the survival and proliferation of hematopoietic stem cells. When an internal tandem duplication (ITD) occurs, the FLT3 receptor is constitutively activated, meaning it stays "turned on" and signals the cells to divide uncontrollably. Statistics show that pediatric patients with the FLT3-ITD mutation face a significantly higher risk of relapse and a lower five-year survival rate compared to those without the mutation.
The traditional "one-size-fits-all" approach of high-dose chemotherapy often fails these patients because the underlying genetic drivers of their cancer are not specifically addressed. Furthermore, the long-term "late effects" for survivors of such toxic treatments can include infertility, cognitive impairment, and secondary malignancies, underscoring the desperate need for targeted, less toxic therapies.
Current Research Initiatives and the Role of CureSearch
In response to the limitations of current treatment protocols, organizations like CureSearch for Children’s Cancer are focusing on funding high-impact research. One such initiative is the work being conducted by Dr. Elvin Wagenblast at Mount Sinai. As a recipient of the CureSearch Young Investigator Program, Dr. Wagenblast and his team are investigating the fundamental origins of childhood leukemia.
The research focuses on the "how" and "why" of leukemogenesis—the process by which normal cells transform into cancer cells. By using advanced genomic tools and CRISPR technology, researchers aim to identify the earliest genetic mutations that trigger the disease. Understanding these early steps is crucial for developing "smart" drugs that can target leukemic cells while sparing healthy tissue. The goal is to move away from broad-spectrum cytotoxic agents and toward precision medicine that can effectively neutralize mutations like FLT3-ITD without the devastating side effects Luke Getchell endured.
CureSearch has set a fundraising goal of $25,000 this Childhood Cancer Awareness Month to support Dr. Wagenblast’s research. This funding is vital, as pediatric cancer research historically receives significantly less federal funding than adult cancer research, despite the fact that childhood survivors face a lifetime of potential health complications.
Broader Implications for Pediatric Oncology
The story of Luke Getchell is a poignant reminder of the gaps in current medical capabilities. While significant strides have been made in treating pediatric acute lymphoblastic leukemia (ALL), AML remains a formidable challenge. The medical community’s reaction to cases like Luke’s is one of increasing urgency. There is a growing consensus among pediatric oncologists that the "ceiling" of chemotherapy has been reached; increasing dosages further only increases mortality from toxicity rather than improving cure rates.
The implications of this shift are twofold. First, there is a push for international collaboration in clinical trials to gather enough data on rare mutations like FLT3-ITD. Second, there is an emphasis on the "survivorship" aspect of oncology—ensuring that children who do survive do not do so at the cost of their long-term health.
The Getchell family’s decision to share Luke’s story and his "Lego-cy" serves a dual purpose: it humanizes the cold statistics of oncology and provides a tangible focal point for advocacy. The "Luke’s Lego-cy" display at VCU Health serves as both a memorial and a silent plea for progress. It reminds medical professionals and the public alike that behind every clinical trial and every genetic mutation is a child who deserved a future.
Conclusion and Call to Action
Childhood Cancer Awareness Month serves as a period of reflection on the progress made and the work that remains. The horrors described by the Getchell family—the struggle to administer pills that represent the last hope for survival, the physical trauma of chemotherapy, and the ultimate loss of a child—are realities that the pediatric medical community is striving to eliminate.
By funding innovative research like that of Dr. Wagenblast, the goal is to ensure that future families do not have to endure the same "heartbreaking, stomach-turning reality." The pursuit of safe and effective treatment options is not merely a scientific endeavor but a moral imperative. Contributions made in memory of Luke Getchell are directed toward a future where a diagnosis of AML with a FLT3-ITD mutation is no longer a "grim prognosis," but a manageable and curable condition. Through continued investment in targeted research, the medical community honors the memory of children like Luke by working to ensure that their "Lego-cy" is one of hope and scientific triumph.