The biological foundation of a healthy pregnancy is established long before a woman reaches reproductive age, originating during her own fetal development within her mother’s womb. A groundbreaking study led by Neil Hunter, a professor in the Department of Microbiology and Molecular Genetics at the University of California, Davis, has identified a critical network of proteins that ensures the integrity of this process. Published in the journal Nature, the research illuminates the complex "choreography" required to manage the DNA-containing chromosomes within developing egg and sperm cells. When this molecular sequence falters, the resulting chromosomal abnormalities can lead to infertility, miscarriage, or the birth of children with significant genetic conditions.
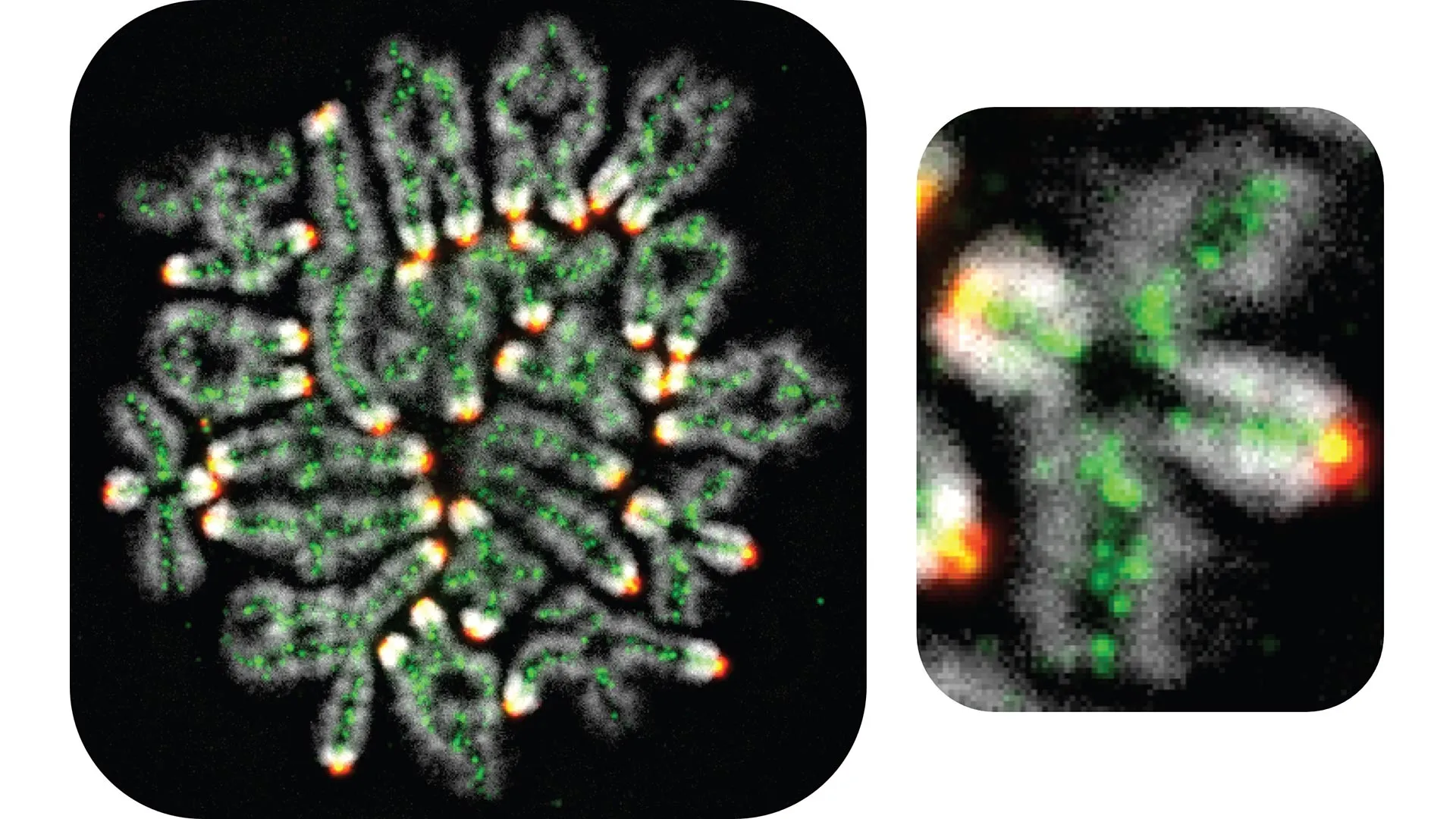
At the heart of this discovery is the process of meiosis, the specialized cell division that produces gametes—eggs and sperm. Unlike somatic cells, which contain two sets of chromosomes (one from each parent), gametes must contain exactly one set. To achieve this, matching pairs of homologous chromosomes must find each other, align, and undergo a physical exchange of genetic material. This exchange, known as a "crossover," is not merely a mechanism for genetic diversity; it is a structural necessity that keeps chromosome pairs tethered together until the cell is ready to divide. The Hunter lab’s findings provide a detailed map of how cells protect these fragile connections over time, a challenge that is particularly acute in human females.
The Evolutionary Mechanics of Chromosomal Crossovers
The fundamental processes of life are often so essential that they remain virtually unchanged across millions of years of evolution. To uncover the secrets of human reproduction, Professor Hunter’s team turned to budding yeast (Saccharomyces cerevisiae), a single-celled organism that has served as a cornerstone of genetic research for decades. Despite the vast biological distance between yeast and humans, the proteins governing chromosome recombination are remarkably conserved. Hunter noted that every protein studied in the yeast model has a direct counterpart in human biology, making these findings directly applicable to clinical understanding of human fertility.
The research focused on a specific intermediate structure known as the "double Holliday junction." Named after the British geneticist Robin Holliday, who first proposed the model of DNA strand exchange in 1964, these junctions represent the moment when DNA strands from two different chromosomes are intertwined. For a successful crossover to occur, these junctions must be stabilized and then precisely cut, or "resolved," to join the maternal and paternal DNA. If the junctions are dismantled prematurely or incorrectly, the chromosomes fail to form the necessary connections, leading to "aneuploidy"—a condition where cells have an abnormal number of chromosomes.
The Biological Timeline: A Decades-Long Stasis
The stakes of maintaining these chromosomal connections are significantly higher in females due to a unique biological timeline. In males, the production of sperm begins at puberty and continues throughout life, with the entire process of meiosis completing in a matter of weeks. However, in females, the initial stages of egg development—including the formation of crossovers—occur entirely within the fetal ovary. By the time a female infant is born, her entire lifetime supply of immature eggs is already present, but these cells are arrested in a state of "suspended animation."
These immature eggs remain in this dormant state for decades, until they are individually activated during ovulation. Only then does the cell complete its division. "Maintaining the crossover connections over many years is a major challenge for immature egg cells," Hunter explained. If the crossovers, which were formed before the woman was even born, degrade or fail during these decades of waiting, the chromosomes will not segregate correctly when the egg finally matures. This age-related degradation is a primary reason why the risk of chromosomal disorders, such as Down syndrome, increases as a woman ages.
Breakthrough Methodology: Real-Time Genetics
The discovery of the protein network responsible for safeguarding these junctions required a level of detail previously unattainable in the field. Hunter and his lead researcher, Shangming Tang—now an assistant professor at the University of Virginia—developed a technique referred to as "real-time genetics." This approach allowed the team to manipulate cells in a synchronized state, targeting specific proteins for degradation at precise moments during the formation of the double Holliday junction.
By systematically removing individual proteins and observing the impact on DNA resolution, the researchers built a comprehensive picture of the cellular defense system. They identified a key role for a protein complex called cohesin. While cohesin was already known for its role in holding sister chromatids together, Hunter’s team discovered that it also acts as a shield for the double Holliday junction. Cohesin prevents an enzyme known as the STR complex (or the Bloom complex in humans) from inappropriately "unwinding" or dismantling the junctions before they can be converted into crossovers.
This protective mechanism is essential because the STR complex is naturally designed to suppress crossovers in most cells to prevent genomic instability. However, in the specific context of making eggs and sperm, crossovers are required. The research demonstrates how the cell selectively disables the STR complex’s "anti-crossover" activity at the right place and time to ensure reproductive success.
Supporting Data and Clinical Implications
The implications of failing to protect these junctions are well-documented in clinical statistics. Chromosomal abnormalities are the leading cause of pregnancy loss, accounting for approximately 50% of all first-trimester miscarriages. Furthermore, aneuploidy is the most common cause of developmental disabilities in humans. Down syndrome, caused by an extra copy of chromosome 21 (Trisomy 21), occurs in approximately 1 in every 700 births in the United States. Other conditions, such as Edwards syndrome (Trisomy 18) and Patau syndrome (Trisomy 13), also result from errors in chromosome segregation during meiosis.
The Hunter lab’s findings provide a new framework for understanding the molecular origins of these conditions. By identifying the specific proteins that "protect" the blueprint of the next generation, scientists can begin to develop better diagnostic tools to assess egg quality and fertility. Moreover, this research has significant overlaps with cancer biology. Many of the proteins involved in meiotic recombination, such as the Bloom complex, are also involved in DNA repair within somatic cells. Mutations in these proteins are known to increase cancer risk, suggesting that the "guardians" of our reproductive cells are also vital for preventing the genetic mutations that lead to tumors.
Collaborative Research and Future Directions
The success of this project was the result of a multi-year effort involving a diverse team of researchers. Beyond the senior leadership, seven undergraduate students from the UC Davis College of Biological Sciences contributed to the work, highlighting the university’s commitment to integrating high-level research with student education. The team utilized an array of advanced scientific facilities at UC Davis, including the Proteomics Core Facility and the MCB Light Microscopy Imaging Facility, to visualize molecular events with unprecedented clarity.
Funding for this extensive study was provided by the National Institutes of Health (NIH) and the Howard Hughes Medical Institute (HHMI), with additional support from several cancer research foundations, including the American Cancer Society and the Damon Runyon Cancer Foundation. This broad base of support reflects the interdisciplinary importance of the work, spanning the fields of reproductive health, genetics, and oncology.
Looking forward, the Hunter lab aims to investigate how these protective protein networks might be influenced by environmental factors or the natural aging process. If the "glue" that holds these chromosome pairs together can be strengthened or preserved, it could lead to revolutionary treatments for infertility or methods to reduce the incidence of genetic disorders.
Conclusion: A Fundamental Understanding of Life
The study published in Nature marks a significant milestone in our understanding of the most fundamental transition in biology: the passing of genetic information from one generation to the next. By detailing the choreography of proteins that safeguard chromosome crossovers, Neil Hunter and his team have provided a missing piece of the puzzle in reproductive science.
The discovery that a complex network of "guardians" must protect our DNA strands from being dismantled by the cell’s own machinery underscores the delicate balance of life. As researchers continue to bridge the gap between yeast models and human clinical applications, the hope is that these molecular insights will eventually translate into better outcomes for families and a deeper appreciation for the intricate processes that occur within the womb, years before a new life even begins. Factual, objective, and meticulously detailed, this research serves as a testament to the power of basic science to illuminate the most complex mysteries of human existence.