The landscape of global health diagnostics is on the precipice of a significant transformation as researchers at Arizona State University (ASU) announce the development of a breakthrough tool capable of identifying a wide array of illnesses with unprecedented speed and affordability. Known as NasRED (Nanoparticle-Supported Rapid Electronic Detection), the device utilizes a single drop of blood to deliver laboratory-quality results within 15 minutes, at a production cost of approximately two dollars per test. This innovation addresses a critical gap in the medical infrastructure, offering a portable solution for the detection of pathogens ranging from the virus that causes COVID-19 to more elusive threats like Ebola, HIV, and Lyme disease.
The development, led by Chao Wang, an associate professor at ASU’s Biodesign Center for Molecular Design and Biomimetics and the School of Electrical, Computer and Energy Engineering, represents a technical milestone in bioengineering. By combining the sensitivity of sophisticated laboratory equipment with the ease of a common rapid antigen test, the ASU team has created a platform that could democratize high-end diagnostics. The findings of their latest study, which demonstrates the tool’s pinpoint accuracy in distinguishing COVID-19 from other respiratory infections, were recently published in the prestigious journal ACS Nano.
The Technical Framework of NasRED
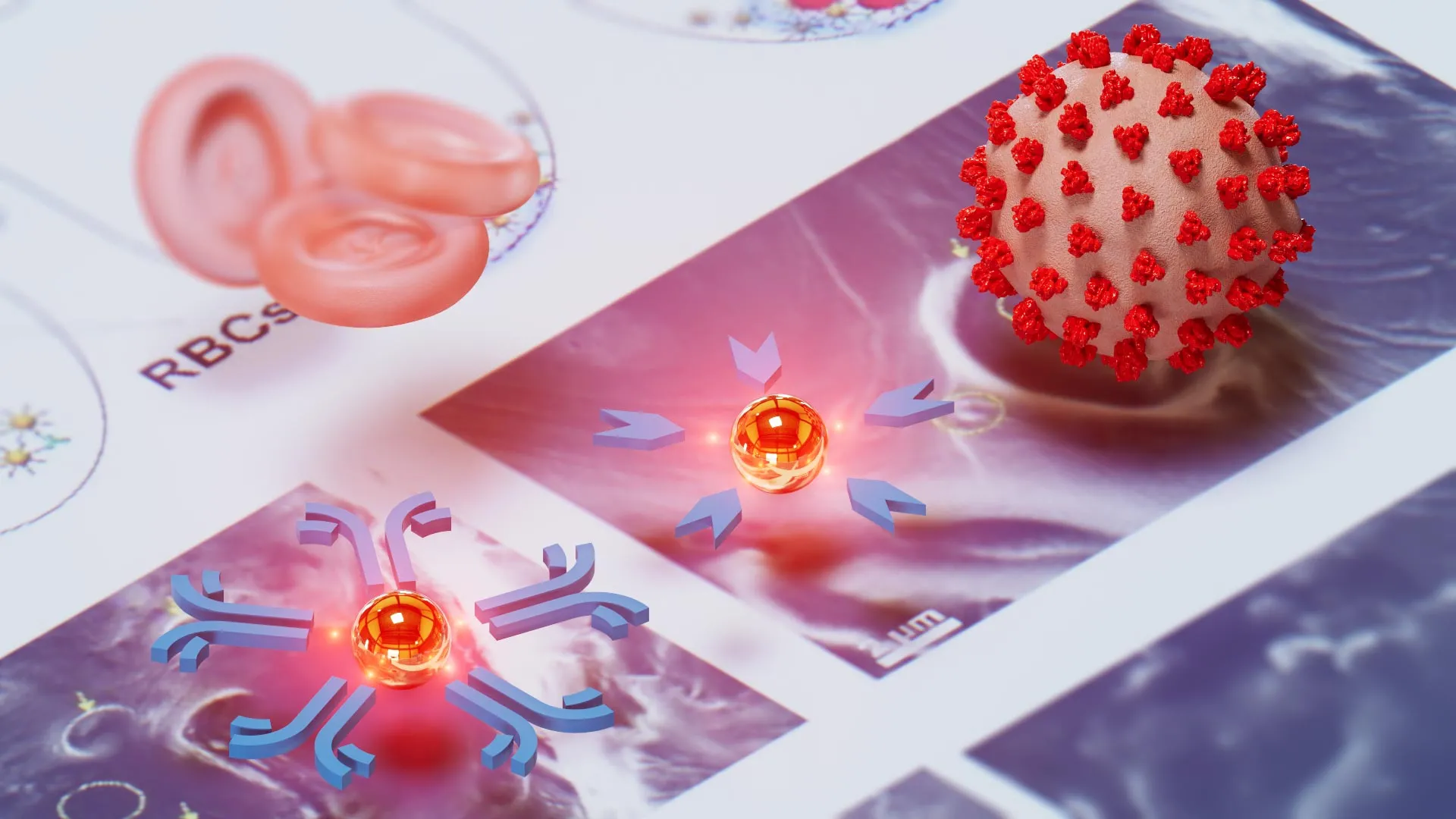
At the core of the NasRED system is the innovative use of gold nanoparticles, which are engineered to interact with extremely low concentrations of disease-related proteins. The process begins by coating these microscopic gold particles with specific molecules tailored to recognize certain diseases. These coatings typically consist of either antibodies, which act as biological magnets to latch onto viral or bacterial proteins, or antigens, which attract the antibodies naturally produced by an infected person’s immune system.
The diagnostic process is elegantly simple. A tiny sample of bodily fluid—such as blood, saliva, or nasal discharge—is introduced into a solution containing these functionalized nanoparticles. If the targeted pathogen or its associated immune response is present, the nanoparticles bind to the markers and settle at the bottom of the testing tube. In a negative sample, the particles remain suspended throughout the liquid.
To interpret the results, the researchers developed a custom electronic detector. A small LED light beam is passed through the upper portion of the liquid. If the fluid is clear—indicating that the nanoparticles have precipitated to the bottom due to a positive reaction—the detector senses a higher intensity of light. This electronic quantification removes the ambiguity often associated with the faint lines seen on traditional rapid tests, providing a definitive and highly sensitive readout.
Unprecedented Sensitivity and Comparison to Existing Standards
The sensitivity of the NasRED platform is perhaps its most remarkable feature. The ASU team reported that the device can detect disease markers at the "attomolar" range. To provide a perspective on this level of precision, Chao Wang likened it to detecting a single drop of ink dispersed within 20 Olympic-sized swimming pools. This allows the tool to identify an infection even when only a few hundred molecules are present in a sample, a concentration roughly 100,000 times lower than the threshold required by many standard laboratory assays.
When compared to existing "gold standard" diagnostic methods, NasRED shows significant advantages in both performance and efficiency. Currently, the medical community relies heavily on Polymerase Chain Reaction (PCR) and Enzyme-Linked Immunosorbent Assay (ELISA) tests. While accurate, these methods are often hindered by:
- Cost: PCR and ELISA require expensive reagents and machinery that can cost tens of thousands of dollars.
- Time: Turnaround times for these tests can range from several hours to several days, especially if samples must be transported to a centralized lab.
- Expertise: Operating these systems requires specialized training and a controlled laboratory environment.
The ASU study indicates that NasRED is approximately 3,000 times more sensitive than traditional ELISA tests. Furthermore, it requires 16 times less sample volume and delivers results nearly 30 times faster. Unlike PCR, which requires complex sample preparation to amplify genetic material, NasRED can detect viral loads directly from bodily fluids, streamlining the entire diagnostic pipeline.
Addressing the Global Burden of Infectious Disease
The motivation behind the NasRED project is rooted in the staggering statistics of global morbidity. Infectious diseases remain one of the primary causes of death worldwide, claiming over 10 million lives annually. In low-income countries, these diseases are the leading cause of mortality, often due to a lack of early detection and intervention.
The "diagnostic gap" is a well-documented phenomenon in global health. In many regions, the absence of reliable testing means that outbreaks of diseases like Ebola or African swine fever are often well-established before health authorities can intervene. A portable, two-dollar test that requires no specialized training could allow frontline health workers in remote areas to conduct immediate screenings, effectively halting the spread of contagion at the source.
The impact is not limited to developing nations. In the United States, diagnostic errors contribute to the death or permanent disability of approximately 800,000 people each year, according to data published in BMJ Quality & Safety. Many of these errors involve missed infections or vascular events that are treatable if identified early. For high-risk populations, such as intravenous drug users or those in underserved urban areas, the barriers to consistent testing—such as cost and the need for clinical appointments—can lead to late-stage diagnoses of HIV or Hepatitis C (HCV). NasRED offers a way to bring the laboratory to the patient, facilitating regular screening in non-traditional settings.
Chronology of Development and Collaborative Research
The journey to the current NasRED device has been one of iterative refinement. An earlier version of the technology was successfully utilized to detect Ebola virus particles in minute blood samples. Building on that foundation, the research team, which includes Yeji Choi, Seyedsina Mirjalili, Ashif Ikbal, and several other ASU scholars, expanded the platform’s capabilities to address the COVID-19 pandemic.
The team’s ability to pivot between different pathogens is a result of the system’s modular design. Because the gold nanoparticles can be coated with a variety of different proteins, the same electronic hardware can be used to test for a multitude of conditions. To date, the researchers have demonstrated the platform’s efficacy in detecting:
- Respiratory Viruses: Including SARS-CoV-2.
- Bacterial Toxins: Such as Shiga toxin-producing E. coli.
- Chronic Disease Markers: Proteins related to Alzheimer’s disease and various cancer biomarkers.
- Vector-Borne Illnesses: Specifically Lyme disease.
- Agricultural Threats: Such as African swine fever, which has devastating impacts on global food security.
Chao Wang’s recent recognition as a Bay Area Lyme Foundation Emerging Leader Award recipient underscores the project’s momentum. He intends to use the high sensitivity of NasRED to tackle the notorious difficulty of diagnosing early-stage Lyme infection, which often eludes conventional testing.
Future Implications and the Path to Home Testing
While the current iteration of NasRED requires small benchtop equipment for the mixing and spinning of samples, the ASU team is already looking toward further miniaturization. The ultimate goal is to automate the process entirely, creating a device that could be used as a home test.
The implications of such a device are profound. Unlike current home COVID tests, which are often criticized for their lower sensitivity compared to PCR, a home-based NasRED unit would provide clinical-grade accuracy. This could lead to a new era of "real-time" health monitoring, where individuals can screen themselves for a variety of conditions before symptoms even become severe.
Beyond individual health, the data generated by widespread use of such tools could revolutionize public health surveillance. Real-time, localized data on disease prevalence would allow for more targeted interventions and a better understanding of how diseases move through populations.
In a broader sense, the NasRED platform represents a shift toward "point-of-care" diagnostics. By removing the logistical hurdles of the centralized laboratory, medical providers can make faster, more informed decisions. Whether in a busy urban hospital trying to triage patients or a rural clinic with limited resources, the ability to confirm a diagnosis in 15 minutes for the price of a cup of coffee is a paradigm shift that could save countless lives.
As the research moves toward commercialization and regulatory approval, the ASU team remains focused on the modularity of their invention. The capacity to "swap in" different proteins ensures that NasRED will remain a relevant tool against both current health threats and future emerging pathogens. In a world increasingly defined by its vulnerability to infectious outbreaks, the development of NasRED provides a vital new line of defense in the global public health arsenal.