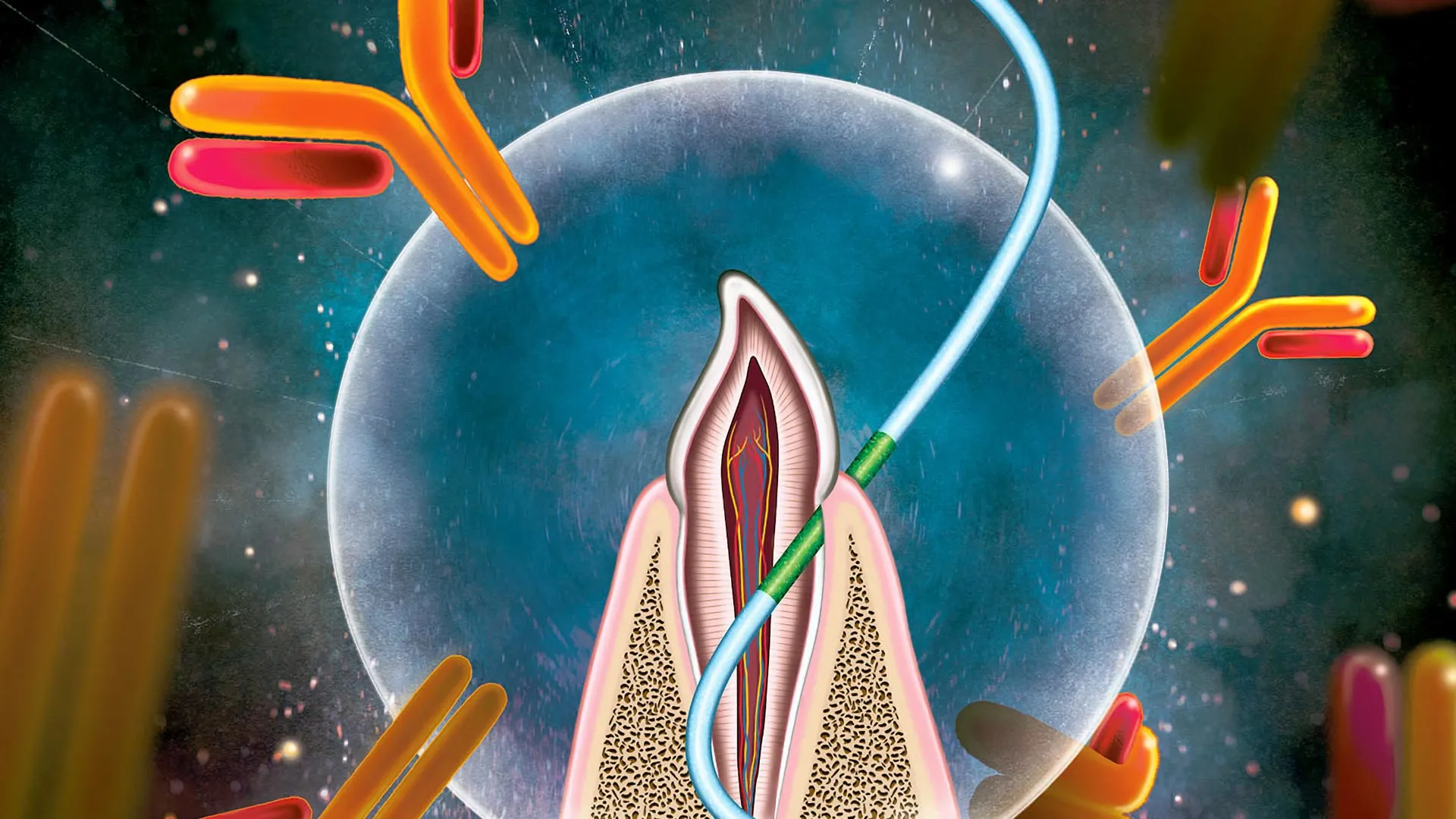
In a significant departure from traditional immunization techniques, a multidisciplinary team of researchers has successfully demonstrated a novel vaccine delivery method that utilizes dental floss to administer vaccines through the tissue located between the teeth and gums. This pioneering approach, detailed in a study published in the journal Nature Biomedical Engineering, leverages a specific anatomical site known as the junctional epithelium to stimulate a robust immune response. Unlike conventional intramuscular injections, which primarily generate antibodies within the bloodstream, this flossing technique has been shown to induce high levels of antibodies on mucosal surfaces, including the linings of the nose and lungs, providing a critical first line of defense against airborne pathogens like influenza and COVID-19.
The Science of Mucosal Immunity and the Junctional Epithelium
To understand the significance of this breakthrough, one must first examine the mechanics of the human immune system. Most current vaccines are administered via a needle into muscle tissue. While highly effective at producing systemic immunity—antibodies circulating in the blood—these injections often fail to produce a strong "mucosal" response. Mucosal surfaces are the primary entry points for the vast majority of human pathogens. When a virus is inhaled, it first encounters the lining of the respiratory tract. If the body has already stationed antibodies at these "front doors," it can neutralize the threat before it ever enters the bloodstream.
Harvinder Singh Gill, the Ronald B. and Cynthia J. McNeill Term Professor in Nanomedicine at North Carolina State University and the study’s corresponding author, emphasizes the strategic importance of this approach. According to Gill, when a vaccine is introduced directly to a mucosal surface, the body responds by producing antibodies both systemically and locally. This dual-layered defense is essential for preventing infection rather than merely mitigating the severity of the disease once it has taken hold.
The research focuses on the junctional epithelium, a specialized and highly permeable layer of tissue situated at the base of the gingival sulcus—the pocket between the tooth and the gum. While most epithelial tissues, such as the skin or the lining of the intestines, act as formidable barriers to prevent foreign substances from entering the body, the junctional epithelium is unique. Its primary biological function is to allow immune cells to pass from the body into the mouth to combat oral bacteria. The researchers hypothesized that if immune cells could move out through this tissue, vaccine molecules could move in.
Chronology of the Research and Experimental Design
The development of this technology followed a rigorous chronological path, moving from theoretical modeling to animal testing and eventually to human usability trials. The research began at Texas Tech University, where Gill and his team, including lead author Rohan Ingrole, sought to identify a mucosal delivery site that avoided the pitfalls of existing methods.
Existing mucosal delivery systems, such as nasal sprays, face significant hurdles. The nasal cavity is designed to filter out particles, meaning many vaccine formulations are simply blocked by the nasal barrier. Furthermore, there have been long-standing safety concerns regarding intranasal delivery, as the proximity to the olfactory nerves presents a theoretical risk of the vaccine or its components reaching the brain. The oral cavity, specifically the area under the tongue (sublingual delivery), has been the "gold standard" for oral mucosal vaccination, yet it often requires high doses and yields inconsistent results.
In the first phase of the study, the researchers turned to an animal model using lab mice. They applied a peptide-based influenza vaccine to unwaxed dental floss and used it to treat the mice’s teeth. To establish a baseline of efficacy, they compared the results of the floss-based delivery against two other methods: nasal administration and sublingual administration.
The results were definitive. The floss-based method produced a mucosal antibody response that was far superior to the sublingual method. Perhaps more importantly, the flossing technique provided protection against the flu virus that was comparable to the nasal delivery method, which is widely considered the most effective mucosal route currently available.
Broad Compatibility with Vaccine Technologies
A critical component of the study involved testing whether the junctional epithelium could accept various types of vaccine platforms. Modern medicine relies on several distinct technologies, and a delivery method is only as useful as the cargo it can carry. The researchers tested the floss-based delivery with three prominent classes of vaccines:
- Proteins: Often used in traditional vaccines, such as those for hepatitis B.
- Inactivated Viruses: Used in conventional flu shots.
- mRNA: The revolutionary technology used in the most prominent COVID-19 vaccines.
In every instance, the application via the junctional epithelium resulted in robust antibody production in both the bloodstream and across the body’s mucosal layers. This suggests that the "floss-vaccine" could be a universal platform capable of delivering a wide array of immunizations.
Furthermore, the team investigated environmental factors that might affect the vaccine’s efficacy in a real-world setting. In the mouse models, researchers found that the consumption of food or water immediately following the "vaccination flossing" did not diminish the immune response. This finding is significant for human application, as it suggests the method is resilient to the typical activities of daily life.
Transitioning to Human Application: The Floss Pick Study
While traditional dental floss worked for lab mice, the researchers recognized that manual flossing with vaccine-coated string would be impractical and potentially messy for human patients. To bridge the gap between the lab and the clinic, they adapted the technology for use with a "floss pick"—a plastic tool with a pre-strung piece of floss held between two prongs.
To test the viability of this delivery mechanism in humans, the team conducted a study involving 27 participants. The goal was not yet to vaccinate them, but to determine if a person could accurately deposit a substance into the junctional epithelium using a floss pick. The researchers coated the floss with a fluorescent food dye and instructed the participants to floss as they normally would.
The data revealed that approximately 60% of the dye was successfully deposited into the gum pocket. This high rate of deposition suggests that the floss pick is a viable and practical tool for self-administration of vaccines. For public health officials, the prospect of a self-administered vaccine that does not require a trained clinician to give an injection could be transformative for mass vaccination campaigns.
Implications for Public Health and Global Logistics
The implications of a floss-based vaccine delivery system are far-reaching. From a psychological perspective, it addresses the widespread issue of needle phobia, which remains a significant barrier to vaccine uptake globally. By transforming a medical procedure into a familiar part of a daily hygiene routine, health providers could potentially increase vaccination rates in hesitant populations.
From a logistical standpoint, the benefits are equally compelling. Traditional injectable vaccines generate a massive amount of medical waste in the form of "sharps" (needles and syringes), which require specialized disposal protocols to prevent the spread of bloodborne pathogens. A floss-based system would eliminate this waste stream entirely. Additionally, if the vaccine formulations used on the floss can be engineered for shelf-stability at room temperature—a goal many nanomedicine researchers are currently pursuing—it could revolutionize vaccine distribution in developing nations where the "cold chain" (refrigerated transport) is difficult to maintain.
Expert Analysis and Future Challenges
Despite the promising results, the researchers and independent medical analysts emphasize that several hurdles remain. One of the most prominent concerns is the health of the oral cavity itself. Professor Gill noted that the study must still determine how gum disease (gingivitis or periodontitis) might affect the absorption of the vaccine. Inflammation or bleeding in the gums could potentially alter the permeability of the junctional epithelium, either hindering the vaccine’s entry or causing it to enter the bloodstream too rapidly.
There is also the question of age-appropriateness. This method is inherently unsuitable for infants and toddlers who have not yet developed teeth or the necessary gum structure. Therefore, it would likely serve as a complementary tool rather than a total replacement for pediatric injections.
Independent immunologists have reacted to the study with cautious optimism. Dr. Richard Compans of Emory University, a co-author of the paper, suggests that the ability to target the gingival sulcus opens a new frontier in "localized systemic" immunization. Public health experts have noted that if clinical trials prove successful, this could be the key to managing seasonal respiratory surges more effectively by providing a "booster" that people can use at home at the start of flu season.
Conclusion and Path to Clinical Trials
The research, supported by the National Institutes of Health (NIH) and the Whitacre Endowed Chair at Texas Tech, has already led to a patent filing by Gill, Ingrole, and Akhilesh Kumar Shakya. The next steps involve moving toward Phase I clinical trials to evaluate the safety and immunogenicity of the floss-based delivery in humans.
If the technology moves from the laboratory to the pharmacy shelf, it could represent one of the most significant shifts in vaccine administration since the invention of the bifurcated needle for smallpox. By turning a routine dental habit into a life-saving medical intervention, the researchers have not only found a new way to deliver medicine but have also reimagined the role of mucosal surfaces in the global fight against infectious disease. The "floss-vaccine" stands as a testament to the power of looking at familiar biological structures—like the space between our teeth—through a revolutionary new lens.