A kidney cancer diagnosis often ushers in a period of profound uncertainty, fear, and a cascade of urgent questions for individuals and their loved ones. However, in the face of such challenges, it is crucial to recognize that the landscape of kidney cancer treatment is undergoing a transformative shift, largely driven by the remarkable advancements in immunotherapy. This innovative approach is not only providing new options but also empowering patients to make informed, impactful decisions about their care. The journey against cancer is rarely walked alone, and the scientific community, healthcare providers, and patient advocacy groups stand united in the ongoing fight, continually pushing the boundaries of what is possible.
Understanding Kidney Cancer: A Comprehensive Overview
Kidney cancer originates when cells within the kidneys begin to grow uncontrollably, eventually forming a malignant tumor. These vital, bean-shaped organs, nestled on either side of the spine just below the rib cage, are approximately the size of a fist and perform several indispensable functions for overall bodily health. Primarily, the kidneys are responsible for filtering waste products and excess water from the blood, producing urine, and maintaining the body’s fluid and electrolyte balance. Beyond filtration, they play a critical role in controlling blood pressure through hormone production and stimulating red blood cell production, essential for oxygen transport.

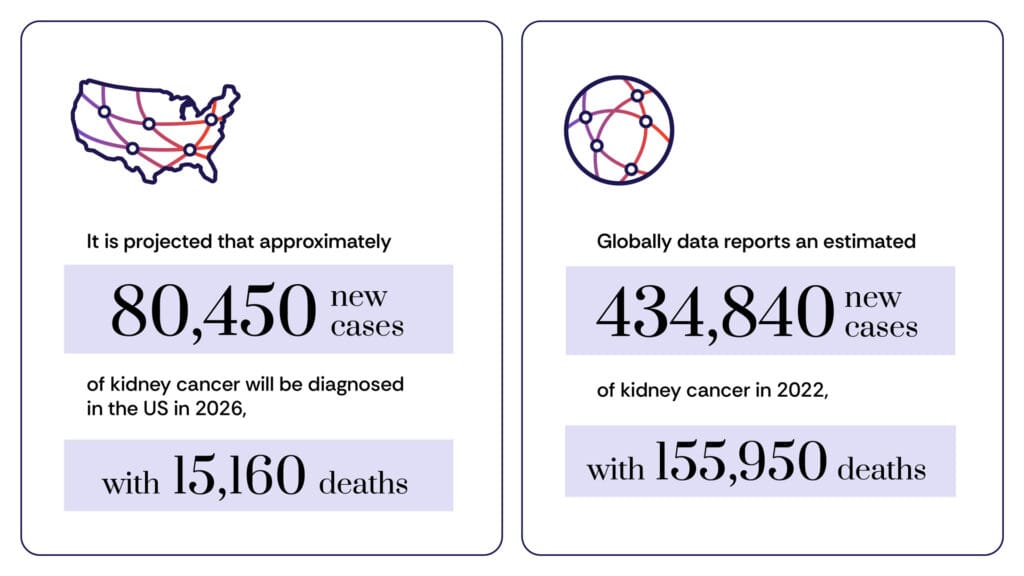
Kidney cancer encompasses several distinct subtypes, with Renal Cell Carcinoma (RCC) being the most common, accounting for approximately 9 out of 10 kidney cancer diagnoses. Within RCC, clear cell RCC is the predominant histological subtype, followed by papillary RCC and chromophobe RCC, each with unique biological characteristics and treatment responses. According to the American Cancer Society (ACS), kidney and renal pelvis cancers are among the ten most common cancers in the U.S., with an estimated 82,290 new cases and 14,310 deaths anticipated in 2023 alone. Globally, the incidence is also significant, with the World Health Organization’s GLOBOCAN project reporting over 430,000 new cases and 180,000 deaths worldwide in 2020, reflecting a growing public health concern. The disease disproportionately affects older individuals, with the average age at diagnosis being 65, and it is observed to be nearly twice as prevalent in men compared to women, a disparity that researchers continue to investigate, potentially linking it to environmental exposures, hormonal factors, or lifestyle differences.
Several factors are known to increase the risk of developing kidney cancer. Prominent among these are smoking, which significantly elevates risk; obesity, a modifiable risk factor linked to numerous health issues; and hypertension (high blood pressure). Genetic predispositions also play a role, with certain inherited syndromes like von Hippel-Lindau disease, Birt-Hogg-Dubé syndrome, and hereditary papillary renal cell carcinoma substantially increasing susceptibility. Additionally, long-term dialysis treatment, occupational exposure to certain substances (such as cadmium or asbestos), and a history of certain medications can contribute to risk. Early-stage kidney cancer often presents without noticeable symptoms, making routine check-ups and vigilance for subtle signs crucial. As the disease progresses, common symptoms may include blood in the urine (hematuria), persistent back or side pain, a lump in the abdomen, unexplained weight loss, and chronic fatigue. Diagnosis typically involves a combination of physical examination, urine tests, blood tests, and advanced imaging techniques such as CT scans, MRI, or ultrasound, often followed by a biopsy to confirm the cancer type and guide treatment decisions.
The Evolving Landscape of Kidney Cancer Treatment: From Traditional Approaches to Targeted Therapies
For decades, the primary treatment modalities for kidney cancer, especially localized disease, centered around surgical intervention. Nephrectomy, the surgical removal of part or all of the kidney, remains the cornerstone of curative treatment for many patients. For those with localized disease, partial nephrectomy (removing only the tumor and a margin of healthy tissue) is often preferred to preserve kidney function, particularly in cases of smaller tumors. For larger or more complex tumors, a radical nephrectomy (removal of the entire kidney) may be necessary. Ablative techniques like radiofrequency ablation or cryoablation are also used for smaller tumors, especially in patients who are not surgical candidates. Radiation therapy and chemotherapy, while standard for many other cancer types, historically had limited efficacy against kidney cancer, particularly RCC, which is known for its inherent resistance to conventional cytotoxic chemotherapy agents, offering minimal survival benefits for advanced disease.

The advent of targeted therapies in the early 2000s marked the first significant paradigm shift in the management of advanced kidney cancer. These drugs, primarily tyrosine kinase inhibitors (TKIs) such as Sunitinib, Pazopanib, and Axitinib, were designed to block specific molecular pathways that promote cancer growth and blood vessel formation (angiogenesis). By inhibiting these pathways, TKIs could slow tumor progression and, in some cases, shrink tumors, offering a substantial improvement over the limited options available previously for metastatic disease. The approval of Sorafenib in 2005, followed by Sunitinib in 2006, heralded a new era, providing the first systemic treatments with demonstrable survival benefits for metastatic RCC. While these targeted therapies provided a lifeline for many, their benefits were often temporary, with patients frequently developing resistance over time, necessitating a continuous search for more durable and effective treatments. This unmet need, coupled with a deeper understanding of cancer immunology, paved the way for the exploration and eventual integration of immunotherapy into the standard of care for kidney cancer.
Immunotherapy: Harnessing the Body’s Own Defenses Against Cancer
Immunotherapy represents a revolutionary approach to cancer treatment, fundamentally differing from traditional methods by leveraging the patient’s own immune system to identify and eliminate cancer cells. The immune system, a complex network of cells, tissues, and organs, is designed to protect the body from foreign invaders like bacteria, viruses, and abnormal cells, including cancerous ones. However, cancer cells are often adept at evading this immune surveillance. They can develop mechanisms to "hide" from immune cells by mimicking healthy cells or actively suppress immune responses by exploiting immune checkpoints, allowing them to grow and spread unchecked.
Immunotherapy works by "unmasking" these cancer cells and "unleashing" the full potential of the immune system against them. This can involve various strategies, but for kidney cancer, immune checkpoint inhibitors (ICIs) have emerged as the most impactful class of therapies. These drugs target specific proteins, or "checkpoints," on immune cells (like T-cells) or cancer cells that act as brakes on the immune system. By blocking these inhibitory signals, ICIs essentially release the brakes, allowing the T-cells to become activated, recognize cancer cells as foreign, and launch a robust attack. The beauty of this approach lies in its potential for long-lasting responses, as the immune system can develop a "memory" of the cancer, potentially reducing the chance of recurrence even after treatment cessation in some cases.
The Transformative Role of Immune Checkpoint Inhibitors in Kidney Cancer Treatment
The introduction of immune checkpoint inhibitors fundamentally altered the prognosis for many patients with advanced kidney cancer. The first major breakthrough occurred in 2015 with the U.S. Food and Drug Administration (FDA) approval of nivolumab (Opdivo), a PD-1 inhibitor, for advanced RCC patients who had previously received anti-angiogenic therapy. This landmark approval, based on the CheckMate 025 clinical trial, demonstrated improved overall survival compared to everolimus, a targeted therapy, marking a pivotal moment in the treatment landscape.
Since then, the therapeutic landscape has expanded significantly, with several ICIs receiving approval for kidney cancer, both as monotherapies and, more critically, in combination regimens:
- PD-1/PD-L1 Inhibitors: These drugs block the programmed death-1 (PD-1) protein on T-cells or its ligand, PD-L1, on cancer cells. Key examples include Nivolumab (Opdivo), Pembrolizumab (Keytruda), and Avelumab (Bavencio). By interrupting this pathway, T-cells are reactivated to attack the tumor.
- CTLA-4 Inhibitors: These drugs block the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) on T-cells. Ipilimumab (Yervoy) is the prime example. CTLA-4 acts as an early brake on T-cell activation, and blocking it can enhance the initial immune response.
The most significant advancements have come from combination therapies. Recognizing that targeting multiple immune pathways could yield synergistic effects, researchers explored various combinations:

- ICI-ICI Combinations: The combination of nivolumab (PD-1 inhibitor) and ipilimumab (CTLA-4 inhibitor) was approved in 2018 for intermediate- and poor-risk advanced RCC, based on the CheckMate 214 trial, demonstrating superior overall survival and durable responses compared to sunitinib. This dual checkpoint blockade unleashes the immune system at different stages of the immune response, leading to more potent anti-tumor activity.
- ICI-TKI Combinations: This strategy combines an immune checkpoint inhibitor with a targeted therapy (tyrosine kinase inhibitor). Examples include Pembrolizumab + Axitinib (approved 2019, based on KEYNOTE-426), Nivolumab + Cabozantinib (approved 2021, based on CheckMate 9ER), Avelumab + Axitinib (approved 2019, based on JAVELIN Renal 101), and Lenvatinib + Pembrolizumab (approved 2021, based on CLEAR trial). These combinations have become front-line standards of care for many patients with advanced RCC, consistently showing improved response rates, progression-free survival, and overall survival compared to TKIs alone. The rationale is that TKIs can not only directly inhibit tumor growth but also modulate the tumor microenvironment, making it more amenable to immune attack. They can reduce immunosuppressive cells, enhance antigen presentation, and normalize tumor vasculature, thus synergizing with ICIs.
The impact of these combination therapies has been profound. For patients with metastatic kidney cancer, once considered a rapidly fatal disease with limited treatment options, the median overall survival has significantly extended, and a subset of patients achieves long-term remission, a scenario that was almost unimaginable a

