The landscape of oncology has undergone a seismic shift over the last decade, primarily driven by the advent of T cell immunotherapies. These "living drugs" leverage the body’s own immune system to identify and eliminate malignant cells, offering hope where traditional chemotherapy and radiation have failed. However, despite the clinical success of treatments like CAR-T cell therapy, a profound mystery has persisted at the heart of immunology: how exactly does the T cell receptor (TCR), the primary "antenna" of the immune system, trigger an internal alarm when it detects a threat? For years, this molecular mechanism remained obscured, leading to a bottleneck in the development of therapies for solid tumors.
New research from The Rockefeller University has finally illuminated this process, revealing that the TCR operates like a molecular "jack-in-the-box." Published in the journal Nature Communications, the study provides the first high-resolution look at the TCR in its natural, resting state within a membrane environment. The findings contradict years of scientific assumptions and provide a vital roadmap for engineering more sensitive, effective, and versatile cancer treatments.
The Critical Role of T Cell Receptors in Modern Medicine
To appreciate the significance of this discovery, one must understand the role T cells play in the human body. T cells are the primary executioners of the adaptive immune system. They patrol the body, scanning the surfaces of other cells for signs of infection or cancerous transformation. This scanning is performed by the T cell receptor (TCR), a complex protein structure embedded in the T cell’s outer membrane.
When a TCR encounters an antigen—a small fragment of a protein presented by a Human Leukocyte Antigen (HLA) molecule on the surface of another cell—it must decide whether that cell is a friend or a foe. If an antigen is recognized as "foreign" or "dangerous," the TCR sends a signal into the interior of the T cell, activating a cascade of biochemical events that leads to the destruction of the target cell.
While T cell immunotherapies have been remarkably effective against "liquid" cancers, such as certain types of leukemia and lymphoma, they have struggled to penetrate and destroy solid tumors, such as sarcomas, lung cancer, or pancreatic cancer. One reason for this failure is the lack of a fundamental understanding of TCR signaling. Without knowing how the receptor switches from "off" to "on," scientists have been limited in their ability to "tune" these receptors to recognize the subtle signals emitted by solid tumor cells.
A Decadelong Structural Mystery
The quest to understand the TCR’s structure is not new. For decades, structural biologists have used various techniques to map the individual components of the TCR complex. We have known that the TCR is not a single protein but a sophisticated assembly of eight different protein chains. However, capturing these eight proteins in their active and inactive states proved to be an immense challenge.
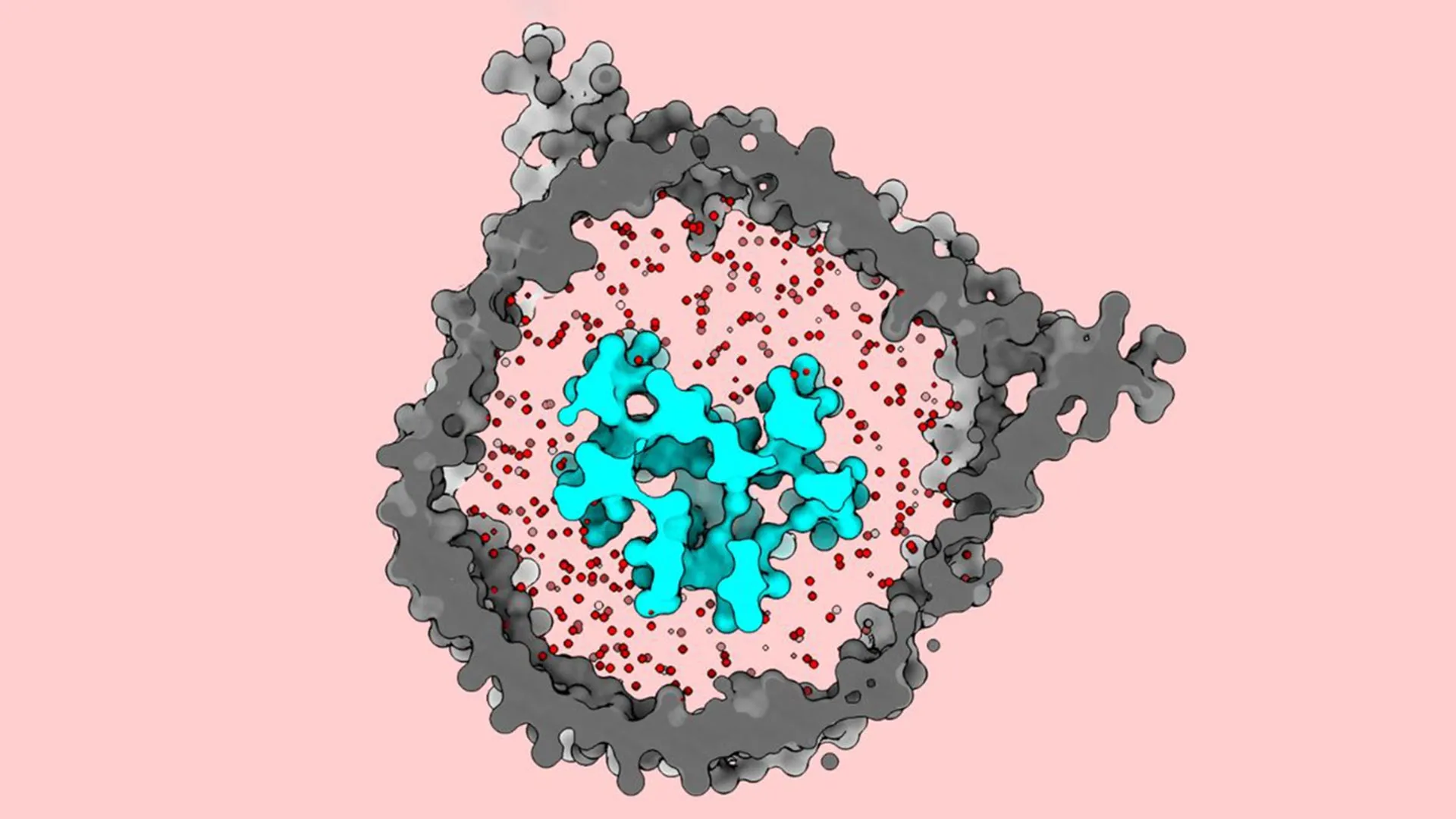
Previous attempts to visualize the TCR relied on detergents to extract the proteins from the cell membrane. While this allowed scientists to use cryo-electron microscopy (cryo-EM) to see the receptor, the process of stripping away the surrounding lipids—the fats that make up the cell membrane—altered the receptor’s shape. These earlier studies consistently showed the TCR in an "open" or extended configuration. Consequently, the prevailing scientific consensus was that the TCR remained in an open state even when it was dormant, and that activation occurred through some other, perhaps less dramatic, mechanism.
This "open-by-default" model puzzled researchers like Ryan Notti, an instructor in clinical investigation at Rockefeller and a physician at Memorial Sloan Kettering Cancer Center. Notti, who specializes in treating sarcomas, noted that many of his patients did not respond to current immunotherapies. If the TCR was always open, why was it so difficult to trigger an immune response against certain cancers?
The Breakthrough: Recreating the Native Milieu
The breakthrough came when Thomas Walz, a world-renowned expert in cryo-EM and head of the Laboratory of Molecular Electron Microscopy at Rockefeller, decided to revisit the TCR structure using a more sophisticated approach. Rather than using detergents to isolate the receptor, Walz’s team utilized "nanodiscs"—tiny, disc-shaped sections of synthetic membrane that mimic the environment of a living cell.
"We can change the biochemical composition, the thickness of the membrane, the tension and curvature, the size—all kinds of parameters that we know have an influence on the embedded protein," Walz explained. By carefully selecting a lipid mixture that mirrored the native T cell membrane, the researchers were able to reinsert the TCR into a stable, life-like environment.
The result was a scientific revelation. Under the cryo-EM, the TCR did not appear open and extended as it had in previous studies. Instead, in its dormant state, the receptor was tightly coiled and compact. It was only when the researchers introduced an antigen-presenting molecule that the receptor underwent a massive conformational change, springing upward and outward.
"The data that were available when we began this research depicted this complex as being open and extended in its dormant state," says Notti. "But we found that it does, springing open like a sort of jack-in-the-box."
Chronology of the Discovery
The journey to this discovery began with Ryan Notti’s transition from a Ph.D. in structural microbiology to a career in clinical oncology. Disturbed by the clinical limitations of immunotherapy in his sarcoma patients, Notti approached Walz with a proposal to apply Rockefeller’s high-end imaging capabilities to the TCR signaling problem.
- Phase I: Membrane Engineering (2021-2022): The team spent months perfecting the lipid composition of the nanodiscs. They discovered that using standard "model" lipids was insufficient; the TCR only maintained its natural "closed" state when placed in a specific mixture of lipids that resembled the complex environment of a human T cell.
- Phase II: Protein Assembly: Assembling the eight-protein TCR complex within the nanodisc was a significant biochemical hurdle. The researchers had to ensure that each component—the TCR alpha and beta chains, and the CD3 epsilon, gamma, delta, and zeta subunits—were correctly oriented.
- Phase III: Cryo-EM Imaging: Using the state-of-the-art facilities at Rockefeller, the team captured thousands of images of the TCR in both its resting and activated states.
- Phase IV: Data Analysis and Publication (2023-2024): The structural data confirmed the "jack-in-the-box" mechanism, leading to the publication of their findings in Nature Communications.
Analyzing the "Jack-in-the-Box" Mechanism
The "jack-in-the-box" analogy is more than just a catchy phrase; it describes a fundamental principle of biological signaling called "allosteric regulation." In its closed state, the TCR is under a form of structural tension, held in place by the lipids of the cell membrane. This prevents the internal signaling components (the CD3 chains) from interacting with the machinery inside the cell that triggers an immune response.
When the TCR binds to an antigen, the energy from that binding event overcomes the restraint of the membrane, allowing the receptor to "spring" open. This extension physically moves the signaling components into a position where they can be phosphorylated by enzymes inside the T cell.
This discovery explains why earlier studies were misleading. By removing the membrane with detergents, previous researchers had essentially "removed the lid" of the jack-in-the-box, allowing the receptor to spring open prematurely. This led to the false conclusion that the receptor was always extended.
Clinical Implications: Re-Engineering the Next Generation of Therapies
The implications of this discovery for cancer treatment are profound. By understanding the physical movement required for TCR activation, bioengineers can now design synthetic receptors with higher precision.
1. Tuning the Activation Threshold
One of the greatest challenges in immunotherapy is the "goldilocks" problem: if a T cell is too sensitive, it may attack healthy tissue (autoimmunity); if it is not sensitive enough, it will ignore cancer cells. "One could imagine using our insights to re-engineer the sensitivity of those receptors by tuning their activation threshold," says Notti. By modifying the "spring" of the jack-in-the-box, scientists could make T cells more likely to trigger when they encounter the weak signals produced by solid tumors.
2. Expanding Scope to Solid Tumors
Sarcomas and other solid tumors often "hide" from the immune system by presenting very few antigens. A more sensitive, better-engineered TCR could potentially detect these elusive targets. This is particularly relevant for "Adoptive T Cell Therapy," where a patient’s T cells are removed, genetically modified to recognize a specific cancer, and then infused back into the patient.
3. Vaccine Design and Infectious Disease
Beyond cancer, this structural data is invaluable for vaccine development. Vaccines work by teaching T cells to recognize specific antigens. "People in the field can now use our structures to see refined details about the interactions between different antigens… and ways to optimize it," says Walz. This could lead to more effective vaccines for viruses that currently evade the immune system, such as HIV or certain strains of influenza.
Official Responses and Scientific Consensus
The scientific community has greeted the Rockefeller study with significant interest. Experts in the field of structural immunology note that this research settles a long-standing debate regarding the "conformational change" model versus the "clustering" model of T cell activation. While clustering (the grouping together of multiple TCRs) may still play a role, the Rockefeller study provides definitive evidence that a major structural shift within a single TCR is a primary trigger.
Thomas Walz, who has spent decades studying macromolecular complexes, described this as "some of the most important work to ever come out of my lab." The study underscores the necessity of "basic science"—the fundamental study of biological structures—in driving clinical breakthroughs.
Conclusion: A New Era of Immunological Precision
The discovery of the TCR’s "jack-in-the-box" mechanism marks a turning point in our understanding of the human immune system. By proving that the cell membrane acts as a vital regulator of immune signaling, the Rockefeller team has corrected decades of scientific misconceptions and opened a new frontier in drug design.
As the medical community moves toward "Next-Generation" immunotherapies, the ability to manipulate the TCR’s structural state offers a level of precision previously thought impossible. For patients with sarcomas and other difficult-to-treat cancers, this basic science discovery provides a tangible hope for more effective, tailored treatments in the near future. The "jack-in-the-box" has finally been understood, and with that knowledge comes the power to harness the immune system with unprecedented accuracy.