Over the past decade, T cell immunotherapy has transitioned from a radical experimental concept to a cornerstone of modern oncology, offering hope where traditional chemotherapy and radiation have failed. By harnessing the body’s own immune system to identify and eradicate malignant cells, these therapies have achieved remarkable remissions in blood cancers. However, despite this clinical success, a fundamental mystery has persisted: how exactly do T cell receptors (TCRs) translate the detection of a cancer marker into a cellular "call to arms" at the molecular level? This lack of mechanical understanding has served as a significant bottleneck, particularly as researchers struggle to adapt these therapies for solid tumors and rare cancers that remain stubbornly resistant to current treatments.
In a landmark study published in Nature Communications, scientists at The Rockefeller University have finally provided a high-resolution answer to this question. Using advanced cryo-electron microscopy (cryo-EM), the team revealed that the TCR functions through a "jack-in-the-box" mechanism—a dynamic structural shift that was previously invisible to researchers using older laboratory techniques. This discovery not only corrects long-standing misconceptions about T cell activation but also provides a literal blueprint for engineering more sensitive and effective cancer treatments.
The Molecular Mystery of T Cell Activation
The T cell receptor is an extraordinarily complex protein machinery embedded in the cell membrane. Its primary role is to scan other cells for signs of infection or malignancy. It does this by interacting with human leukocyte antigen (HLA) complexes, which present small fragments of proteins—antigens—on the cell surface. If the TCR recognizes an antigen as "foreign" or "dangerous," it triggers a signaling cascade that transforms the T cell from a passive observer into a potent killer.
For years, structural biologists struggled to capture the TCR in its natural state. Early models suggested the receptor was a relatively static structure. Because membrane proteins are notoriously difficult to isolate, previous studies often used detergents to strip the TCR from the fatty lipid bilayer of the cell membrane. While this allowed for imaging, it inadvertently altered the protein’s behavior. The resulting images showed the TCR in an "open" or extended configuration, leading scientists to believe that the receptor did not undergo significant structural changes upon binding to an antigen.
"The T cell receptor is really the basis of virtually all oncological immunotherapies, so it’s remarkable that we use the system but really have had no idea how it actually works," says Thomas Walz, head of the Laboratory of Molecular Electron Microscopy at Rockefeller and a world-renowned expert in cryo-EM. "This is some of the most important work to ever come out of my lab."
A Methodological Breakthrough: Recreating the Native Milieu
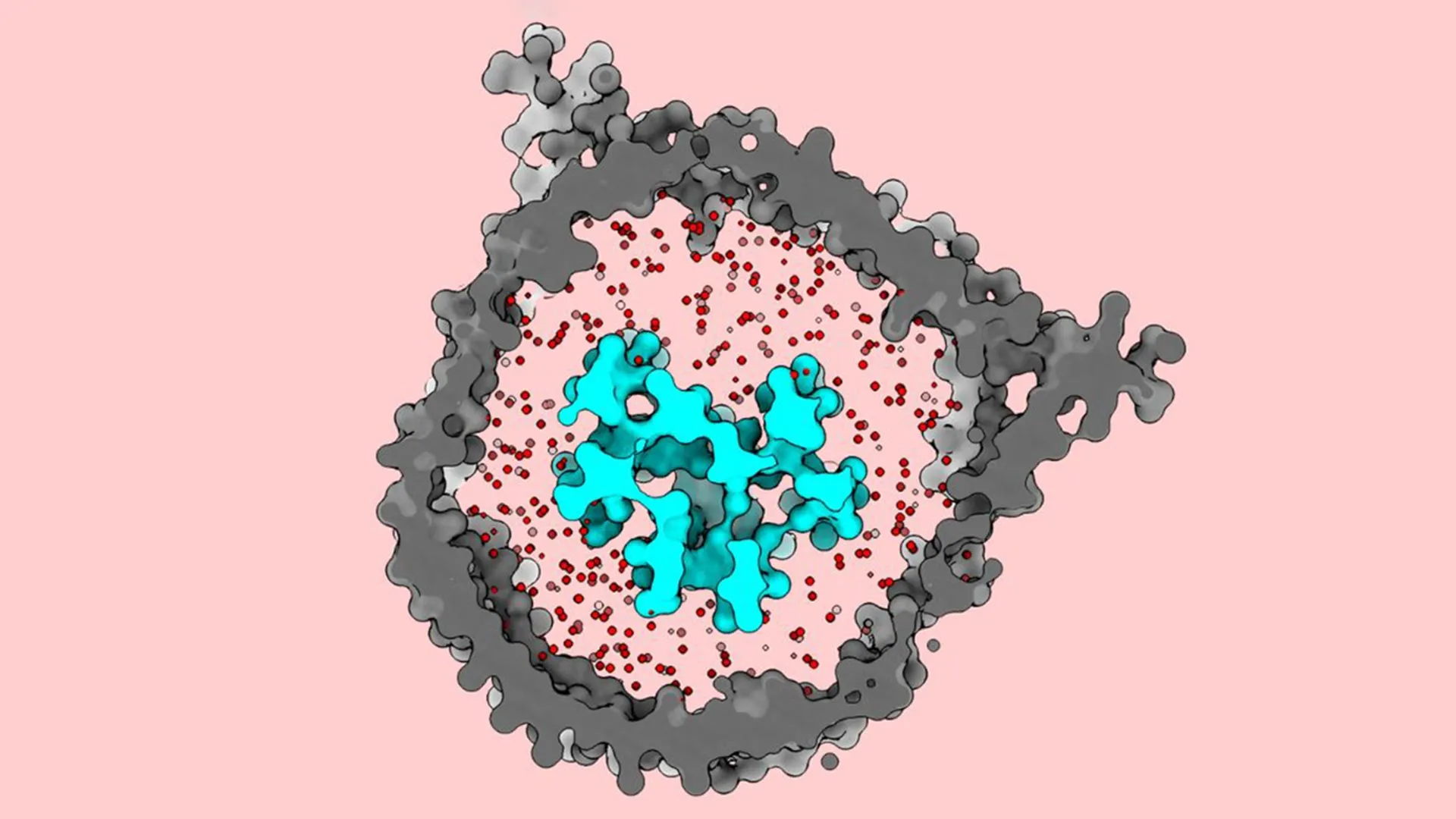
The breakthrough at Rockefeller was driven by a shift in methodology. Rather than stripping the TCR away from its environment, the researchers, led by first author Ryan Notti, sought to rebuild the receptor’s natural surroundings in a test tube. Notti, an instructor in clinical investigation and a physician-scientist at Memorial Sloan Kettering Cancer Center, recognized that the lipid environment of the cell membrane was likely essential to the receptor’s function.
The team utilized "nanodiscs"—tiny, disc-shaped sections of synthetic membrane held together by scaffold proteins. By carefully selecting a lipid mixture that mimicked the specific composition of a human T cell membrane, they were able to reinsert the full eight-protein TCR complex into a stable, native-like environment.
"We can change the biochemical composition, the thickness of the membrane, the tension and curvature, the size—all kinds of parameters that we know have an influence on the embedded protein," Walz explains. This meticulous reconstruction allowed the researchers to observe the TCR as it truly exists on the surface of a living cell.
The Jack-in-the-Box Discovery
When the team visualized the TCR within these nanodiscs using cryo-EM, the results were startling. Contrary to previous detergent-based studies, the inactive TCR appeared compact and tightly folded against the cell membrane. It was not "pre-opened"; rather, it was held in a spring-loaded, dormant state by the surrounding lipids.
Upon encountering an antigen-presenting HLA molecule, the receptor underwent a dramatic conformational change. It "sprung" open, extending outward from the membrane. This motion acts as the physical trigger that transmits a signal from the outside of the cell to the inside, notifying the T cell’s internal machinery to begin an immune response.
"The data that were available when we began this research depicted this complex as being open and extended in its dormant state," says Notti. "But we found that it does, springing open like a sort of jack-in-the-box. The intact membrane keeps the receptor in a closed position until activation occurs. In earlier studies, detergent may have removed this restraint, allowing the receptor to open prematurely."
Historical Context and the Evolution of Immunotherapy
To understand the weight of this discovery, one must look at the timeline of immunotherapy. The field has evolved through several distinct eras:
- Late 19th Century: William Coley first attempts to stimulate the immune system against tumors using bacterial toxins.
- The 1980s-90s: The discovery of Interleukin-2 and the first attempts at adoptive cell transfer.
- 2011: The FDA approval of Ipilimumab, the first checkpoint inhibitor, which "unmasked" cancer cells to the immune system.
- 2017: The landmark approval of CAR-T cell therapies for leukemia and lymphoma.
While CAR-T therapies (Chimeric Antigen Receptor T cells) have been revolutionary, they often bypass the natural TCR by using a synthetic receptor. However, natural TCR-based therapies are considered the "holy grail" for treating solid tumors because they can detect a much wider array of internal cancer proteins than CAR-T cells can. The inability to see how the natural TCR "switches on" has been a primary reason why TCR-based therapies have seen slower progress in the clinic.
Bridging the Gap Between the Lab and the Clinic
For Ryan Notti, the research is deeply personal. As an oncologist treating patients with sarcomas—cancers that arise in the bones and soft tissues—he frequently witnesses the limitations of current immunotherapies. Sarcomas are notoriously difficult to treat because they are often "immunologically cold," meaning they do not naturally attract T cell activity.
"Determining how information gets from outside the cell to the inside, where signaling turns on the T cell, is crucial," Notti says. "Many of my sarcoma patients were not benefiting from T cell immunotherapies. This new fundamental understanding of how the signaling system works may help re-engineer that next generation of treatments."
By understanding the "activation threshold" of the TCR—how much force or what specific binding affinity is required to trigger the jack-in-the-box mechanism—scientists can now look toward "tuning" these receptors. If a patient’s T cells are too sluggish to recognize a tumor, researchers might engineer receptors with a lower "spring" tension, making them easier to activate. Conversely, for autoimmune diseases where T cells are overactive, the mechanism could be modified to keep the receptor in its closed, dormant state.
Supporting Data and Technical Challenges
The technical execution of the study was a feat of molecular engineering. The TCR is not a single protein but a complex assembly of eight different subunits: two ligand-binding chains (TCRα and TCRβ) and six signaling subunits (the CD3 complex consisting of epsilon, delta, gamma, and zeta chains).
The researchers noted that getting all eight proteins to assemble correctly within a nanodisc was one of the most challenging aspects of the project. Furthermore, the choice of lipids proved decisive. The T cell membrane is rich in specific sterols and phospholipids that influence protein rigidity. When the team experimented with "model" lipids that lacked the complexity of native membranes, the TCR failed to maintain its closed state, proving that the membrane itself is an active participant in immune signaling, not just a passive container.
Broader Implications for Vaccine Design and Beyond
The implications of the Rockefeller study extend beyond cancer. Thomas Walz highlights that this structural data is a goldmine for vaccine researchers. Vaccines work by teaching T cells to recognize specific viral or bacterial antigens. By seeing the "refined details" of the interactions between antigens and the TCR, scientists can design vaccines that trigger a more robust and precise immune response.
Furthermore, the study provides a new lens through which to view "immune exhaustion." In chronic diseases like HIV or persistent solid tumors, T cells often become "exhausted" and stop responding. It is possible that the mechanical "spring" of the TCR becomes damaged or permanently stuck over time. Understanding the physical state of the receptor in these conditions could lead to therapies that "reset" the immune system.
Analysis: A New Era of Molecularly Informed Oncology
The discovery of the TCR’s jack-in-the-box mechanism marks the end of the "black box" era of T cell signaling. For years, immunotherapy was a game of trial and error—engineering cells and hoping they worked in the complex environment of the human body. With these new high-resolution images, the field moves into the era of precision molecular engineering.
As the oncology community digests these findings, the focus will likely shift toward "adoptive T cell therapy" (ACT). Unlike CAR-T, which uses a "one-size-fits-all" synthetic hook to grab cancer cells, TCR-based ACT can be tailored to the specific genetic mutations of an individual’s tumor. By optimizing the "opening" mechanism of these tailored receptors, researchers hope to finally break the barrier that has kept immunotherapy from being effective in the majority of cancer types.
In the words of Walz, the marriage of basic science and clinical application is what makes this discovery so potent. While the "jack-in-the-box" is a simple analogy, it represents a profound shift in our ability to manipulate the very machinery of life to fight disease. The next generation of cancer treatments will not just be about finding the right target; it will be about understanding the molecular handshake that tells a cell it is time to fight.