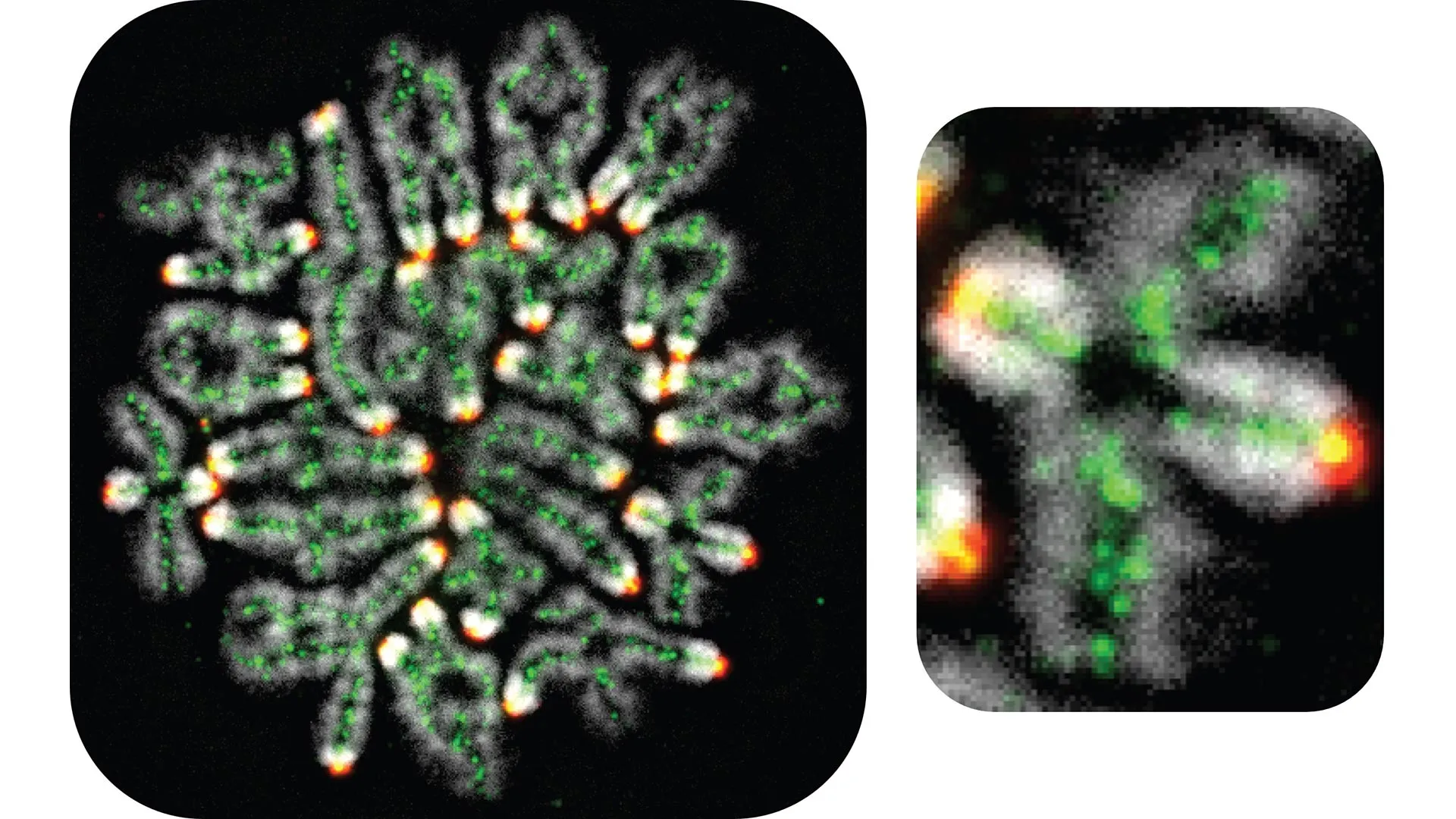
The biological foundations of a successful pregnancy are established long before an individual reaches reproductive age, originating in a series of intricate molecular events during fetal development. For women, the health of future pregnancies is inextricably linked to the quality of egg cells that begin forming while they are still in their own mother’s womb. At the heart of this process is the precise cutting, splicing, and sorting of DNA-containing chromosomes. When this molecular choreography falters, the consequences are profound, leading to infertility, pregnancy loss, or the birth of children with significant genetic conditions.
In a landmark study published in the journal Nature, a research team led by Neil Hunter, a professor in the Department of Microbiology and Molecular Genetics at the University of California, Davis, has revealed a major discovery regarding the mechanisms that safeguard this delicate process. By mapping the interaction of specific proteins that manage chromosome pairs, the team has provided a new understanding of how the body ensures that egg and sperm cells receive the correct genetic "dosage."
The Fundamental Mechanics of Meiosis and Crossovers
To understand the significance of the UC Davis discovery, one must first look at the unique way the body produces gametes—the egg and sperm cells. Unlike somatic cells, which contain 46 chromosomes (23 pairs), gametes must contain only 23 single chromosomes so that when they fuse during fertilization, the resulting embryo has the correct total of 46.
This reduction is achieved through a specialized form of cell division called meiosis. Early in this process, matching (homologous) chromosomes—one inherited from the mother and one from the father—must find each other and pair up. Once aligned, these chromosomes undergo a process known as "crossing over." During a crossover, the parental chromosomes break and exchange segments of DNA.
This exchange serves two vital purposes. First, it ensures genetic diversity, creating a unique mixture of traits in the offspring. Second, and perhaps more critically for the immediate success of a pregnancy, these crossovers act as physical "tethers" that keep the chromosome pairs together. Without these connections, the cell cannot accurately distribute one chromosome of each pair to the daughter cells. If a pair loses its connection, it may segregate incorrectly, leading to aneuploidy—a state where a cell has the wrong number of chromosomes.
The Vulnerability of the Female Reproductive Timeline
One of the most striking aspects of human biology is the timeline of egg development. While males begin producing sperm at puberty and continue throughout their lives, a female is born with her entire lifetime supply of immature eggs. These eggs begin the process of meiosis and form their crossovers while the female is still a fetus.
Once the crossovers are established, the egg cells enter a state of "suspended animation" or meiotic arrest. They remain in this state for decades, until they are called upon during ovulation. Only then does the cell complete the division process, finally separating the chromosome pairs that were tethered together years or even decades earlier.
"Maintaining the crossover connections over many years is a major challenge for immature egg cells," explains Professor Hunter. If the molecular tethers fail during this long wait, the resulting egg may have an extra copy of a chromosome or be missing one entirely. This is a primary cause of Down syndrome (Trisomy 21), where an extra copy of chromosome 21 leads to cognitive impairments and physical health challenges. It is also the leading cause of miscarriages, as many chromosomal imbalances are incompatible with life.
Decoding the Double Holliday Junction
The UC Davis study focused on a critical intermediate structure in the crossover process known as the "double Holliday junction." This occurs when matching DNA strands from two chromosomes are twined together over a short distance, forming a four-armed structure that must be carefully resolved to complete the exchange.
For years, the exact mechanism that ensures these junctions are resolved into crossovers—rather than simply being dismantled—remained a mystery. Using "real-time genetics" in budding yeast (Saccharomyces cerevisiae), Hunter’s team was able to watch these molecular events unfold with unprecedented detail. Yeast serves as an ideal model organism because the fundamental proteins involved in DNA repair and recombination have been conserved through billions of years of evolution.
"The chromosome structures that we studied have changed very little across evolution," Hunter noted. "Every protein that we looked at in yeast has a direct counterpart in humans."
The Discovery: A Protein Shield for Genetic Integrity
The research identified a sophisticated network of proteins that function as a protective "chaperone" system for the double Holliday junction. A key player in this process is a protein complex called cohesin. The team discovered that cohesin, along with other associated proteins, acts to protect the junction from being prematurely or incorrectly dismantled by an enzyme known as the STR complex (referred to as the Bloom complex in humans).
The Bloom complex is a powerful molecular machine capable of unwinding DNA. While it has important roles in other types of DNA repair, its activity must be strictly regulated during meiosis. If the Bloom complex acts too early or without oversight, it dissolves the double Holliday junction before a crossover can be finalized. By "protecting" the junction, cohesin ensures that the physical tether between chromosomes is successfully established.
This discovery highlights a critical checkpoint in the life cycle of an egg. If the proteins designed to protect these junctions are deficient or dysfunctional, the "tethering" fails, setting the stage for reproductive complications later in life.
Methodology: Real-Time Genetics and Collaborative Effort
The breakthroughs achieved by the Hunter lab were made possible through innovative techniques that allowed researchers to manipulate protein levels in living cells with high precision. By engineering yeast cells so that specific proteins would degrade at a chosen moment, the team could observe the immediate impact on DNA recombination.
This "real-time" approach allowed the researchers to move beyond traditional observational studies and actually test the functional necessity of each protein in the network. The project was a massive undertaking that spanned several years and involved a diverse team of scientists.
Lead author Shangming Tang, who began the work as a postdoctoral fellow at UC Davis and is now an assistant professor at the University of Virginia, was joined by a group of seven undergraduate students from the UC Davis College of Biological Sciences. This involvement of undergraduate researchers—Jennifer Koo, Mohammad Pourhosseinzadeh, Emerald Nguyen, Natalie Liu, Christopher Ma, Hanyu Lu, and Monica Lee—underscores the university’s commitment to integrating high-level research with education.
Clinical Implications and the Future of Reproductive Medicine
The implications of this research extend far beyond the laboratory. By identifying the specific proteins that safeguard chromosome crossovers, scientists can now look for mutations or variations in these same proteins in humans who experience unexplained infertility or recurrent pregnancy loss.
In the context of modern reproductive medicine, where the average age of first-time mothers is increasing in many developed nations, understanding why chromosome tethers fail over time is more important than ever. It is well-documented that the risk of chromosomal abnormalities increases with maternal age; this research provides a molecular framework for understanding why the "machinery" of the egg might become less stable as it sits in meiotic arrest.
Furthermore, the study offers potential insights into the Bloom syndrome, a rare genetic disorder characterized by short stature, immunodeficiency, and a high risk of cancer. Because the Bloom complex is a central figure in the crossover process, understanding its regulation provides a clearer picture of the systemic DNA instability that defines the syndrome.
Supporting Data and Institutional Backing
The research conducted by the Hunter lab is supported by a robust network of funding and scientific infrastructure. The National Institutes of Health (NIH) and the Howard Hughes Medical Institute (HHMI) provided primary funding, reflecting the study’s importance to public health and fundamental biology. Additional support came from the UC Davis Comprehensive Cancer Center and several cancer research foundations, highlighting the deep link between DNA repair mechanisms in reproduction and the errors that lead to malignancy.
The study also utilized state-of-the-art facilities at UC Davis, including the Proteomics Core Facility and the MCB Light Microscopy Imaging Facility. These tools allowed the team to visualize molecular interactions at a scale that was previously impossible, bridging the gap between theoretical genetics and observable biological phenomena.
Conclusion: A New Chapter in Genetic Research
The work of Neil Hunter and his colleagues represents a significant leap forward in our understanding of the very beginning of human life. By mapping the "choreography" of proteins that manage our DNA, the team has illuminated the hidden processes that occur within the fetal ovary—processes that ultimately determine the health and viability of the next generation.
As science continues to peel back the layers of meiotic division, the focus will likely shift toward diagnostic applications. Could we eventually screen for the robustness of a woman’s "crossover shield"? Could therapeutic interventions one day stabilize these connections in individuals at high risk for miscarriage? While those goals remain in the future, the discovery of the protein network that protects the double Holliday junction provides the essential foundation upon which those medical advancements will be built. For now, the study stands as a testament to the power of basic science and the enduring relevance of the humble yeast cell in solving the most complex mysteries of human life.