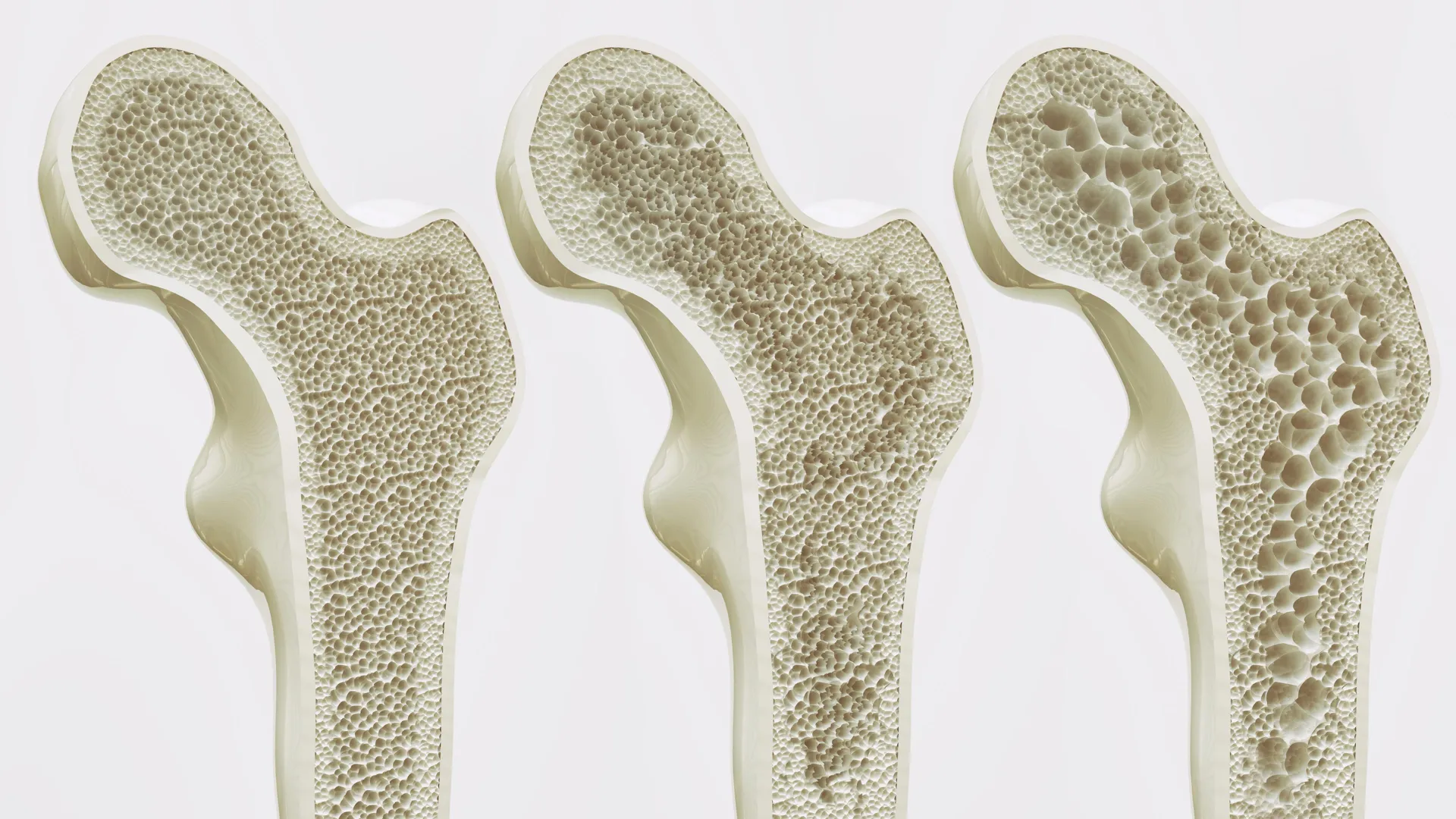
The persistent global challenge of osteoporosis, a debilitating condition characterized by weakened bones and increased fracture risk, has long driven a fervent search for safer and more effective therapeutic interventions. Affecting an estimated 200 million people worldwide, including approximately six million individuals in Germany alone, this "silent disease" predominantly impacts women, particularly after menopause, and poses a significant burden on healthcare systems and individual quality of life. Current treatments, while beneficial, often come with limitations in efficacy, potential side effects, or a narrow focus, highlighting the urgent need for innovative approaches that can not only halt bone deterioration but actively promote its regeneration. A groundbreaking study from Leipzig University has now unveiled a highly promising new biological target, the GPR133 receptor, and a compound, AP503, that could fundamentally reshape the future of bone health.
The Pervasive Threat of Osteoporosis: A Global Health Challenge
Osteoporosis represents a major public health concern, with its prevalence projected to rise further as global populations age. The condition leads to bones becoming porous and brittle, making them susceptible to fractures even from minor falls or stresses. Hip, spine, and wrist fractures are particularly common and can result in chronic pain, disability, loss of independence, and even increased mortality. In Europe, an osteoporotic fracture occurs every 30 seconds, leading to an estimated 3.5 million new fractures annually, costing the EU an estimated €37 billion per year in direct healthcare expenditures alone. Germany, as highlighted by the Leipzig researchers, bears a substantial portion of this burden, with millions suffering from the condition, often undiagnosed until a fracture occurs. The economic and social impact extends beyond direct medical costs, encompassing lost productivity, informal caregiving, and a significant reduction in patients’ quality of life.
The decline in bone mineral density (BMD) is a natural part of aging, exacerbated in women by the dramatic drop in estrogen levels following menopause. Estrogen plays a crucial role in maintaining bone density, and its absence accelerates bone loss, making postmenopausal women particularly vulnerable. Other risk factors include genetic predisposition, certain medical conditions, long-term use of specific medications (e.g., corticosteroids), sedentary lifestyles, and nutritional deficiencies. The complexity of these contributing factors underscores the necessity for multi-faceted and highly targeted therapeutic strategies.
Current Treatment Landscape: Progress and Persistent Gaps
Existing pharmacological treatments for osteoporosis broadly fall into two categories: anti-resorptive agents, which slow down bone breakdown, and anabolic agents, which promote bone formation. Anti-resorptive drugs, such as bisphosphonates (e.g., alendronate, risedronate), denosumab, and selective estrogen receptor modulators (SERMs), are widely used. Bisphosphonates integrate into the bone matrix and inhibit osteoclast activity, thereby reducing bone resorption. While generally effective in reducing fracture risk, they can have side effects such as gastrointestinal issues, and in rare cases, osteonecrosis of the jaw or atypical femoral fractures. Denosumab, a monoclonal antibody, targets RANKL, a protein essential for osteoclast formation, function, and survival, offering an alternative for patients who cannot tolerate bisphosphonates. However, its effects are reversible upon discontinuation, potentially leading to rapid bone loss.
Anabolic agents, like teriparatide and abaloparatide (parathyroid hormone analogues), stimulate new bone formation, primarily by enhancing osteoblast activity. These are generally reserved for patients with severe osteoporosis or those who have not responded to anti-resorptive therapies. While powerful, their use is often limited to a short duration (typically 18-24 months) due to concerns about potential side effects or a plateauing of efficacy, and they are considerably more expensive. Romosozumab, a newer anabolic agent, has a dual effect, increasing bone formation and decreasing bone resorption.
Despite these advancements, a significant unmet need remains for treatments that are highly effective, well-tolerated over the long term, and capable of both preventing further bone loss and actively rebuilding lost bone tissue, especially in a way that mimics natural physiological processes. This is where the recent findings from Leipzig University offer a beacon of hope, by identifying novel biological targets that can be leveraged for next-generation therapeutics.
Unveiling GPR133: A Key Regulator in Bone Metabolism
The Leipzig University study, led by Professor Ines Liebscher from the Rudolf Schönheimer Institute of Biochemistry at the Faculty of Medicine, pinpointed a specific receptor, GPR133, as a critical player in maintaining bone health. This receptor belongs to the larger and increasingly recognized family of adhesion G protein-coupled receptors (GPCRs). GPCRs are integral membrane proteins that act as cellular antennae, detecting a wide array of extracellular signals – from light and odors to hormones and neurotransmitters – and transmitting them into the cell to orchestrate various physiological processes. In fact, GPCRs are the targets of approximately one-third of all currently approved drugs, underscoring their immense pharmacological importance. However, the adhesion GPCR subgroup, while known for its roles in cell adhesion, migration, and development, has historically been less explored in terms of drug discovery compared to other GPCR families.
The new research from Leipzig provides compelling evidence that GPR133 plays a pivotal role in the dynamic process of bone building and maintenance. To elucidate its significance, researchers utilized animal models, specifically mice with genetic modifications that impaired the function of GPR133. These mice exhibited signs of low bone density early in life, strikingly similar to the presentation of osteoporosis in humans. This direct link between GPR133 dysfunction and bone fragility strongly implicated the receptor as a crucial regulator of bone mass.
Professor Liebscher explained, "If this receptor is impaired by genetic changes, mice show signs of loss of bone density at an early age — similar to osteoporosis in humans. Using the substance AP503, which was only recently identified via a computer-assisted screen as a stimulator of GPR133, we were able to significantly increase bone strength in both healthy and osteoporotic mice." This statement not only highlights the foundational genetic insight but also introduces the therapeutic potential of AP503, a compound capable of activating GPR133.
AP503: A Targeted Approach to Bone Regeneration
The discovery of AP503 represents a significant leap forward. This compound was identified through advanced computer-assisted screening methods. These sophisticated computational techniques allow scientists to rapidly screen vast libraries of chemical compounds for their ability to interact with and modulate specific biological targets, such as GPR133, without the need for laborious and time-consuming traditional laboratory experiments for every single compound. In this case, AP503 was found to act as a potent stimulator of GPR133, effectively "turning on" the receptor’s beneficial pathways.
The mechanism by which GPR133 and AP503 exert their effects lies in their ability to rebalance bone remodeling, a continuous lifelong process where old bone tissue is broken down and replaced with new tissue. This delicate equilibrium is maintained by two primary cell types: osteoblasts, which are responsible for forming new bone matrix, and osteoclasts, which resorb (break down) old or damaged bone. In healthy individuals, the activities of osteoblasts and osteoclasts are tightly coordinated. However, in conditions like osteoporosis, the balance is skewed, with bone resorption outpacing bone formation, leading to a net loss of bone mass.
The Leipzig team discovered that GPR133 is activated in bone tissue through two main pathways: interactions between nearby bone cells and physical forces, such as movement and mechanical pressure. Once activated, the receptor sends intracellular signals that significantly enhance the activity of osteoblasts, promoting new bone formation, while simultaneously reducing the activity of osteoclasts, thereby limiting bone breakdown. This dual action results in a net increase in bone density and strength, making the bone more resilient to damage and fractures.
Crucially, AP503 appears to mimic this natural activation process of GPR133. By stimulating the same signaling pathway, it effectively pushes the bone remodeling balance towards formation, leading to stronger, denser bone. This dual anabolic and anti-resorptive potential makes AP503 a highly attractive candidate for future therapeutic development. It suggests that such a compound could not only serve as a preventative measure to protect healthy bones but also as a restorative agent to rebuild bone in individuals already suffering from osteoporosis, including the large population of postmenopausal women.
A Decade of Pioneering Research: The Leipzig Legacy
This breakthrough is not an isolated discovery but the culmination of over a decade of dedicated research at Leipzig University, specifically focusing on the intricate biology of adhesion GPCRs. The work is an integral part of Collaborative Research Centre (CRC) 1423, titled "Structural Dynamics of GPCR Activation and Signaling." This highly interdisciplinary research consortium brings together leading scientists to unravel the fundamental mechanisms governing GPCR function, from their atomic structure to their complex signaling pathways, with the ultimate goal of translating these insights into novel therapeutic strategies.
Leipzig University has, through sustained effort and innovative research, established itself as an internationally recognized leader in the field of adhesion GPCRs. Their consistent contributions have significantly advanced the understanding of how these previously underappreciated receptors function and, critically, how they can be effectively targeted for the development of new therapies across a spectrum of diseases. The identification of GPR133’s role in bone metabolism is a testament to this enduring commitment to fundamental and translational research.
Beyond Bone: A Dual Benefit for Muscle and Bone Strength
The implications of this discovery extend beyond bone health alone, opening up even broader therapeutic avenues, particularly for aging populations. Earlier research conducted by the same Leipzig team had already revealed that activating GPR133 with AP503 also leads to an improvement in skeletal muscle strength. This synergistic effect is of immense significance.
Dr. Juliane Lehmann, lead author of the study and a researcher at the Rudolf Schönheimer Institute of Biochemistry, emphasized this point: "The newly demonstrated parallel strengthening of bone once again highlights the great potential this receptor holds for medical applications in an aging population."
Maintaining both muscle and bone strength is paramount for older adults. Sarcopenia (age-related muscle loss) and osteoporosis often co-exist, contributing significantly to frailty, increased risk of falls, and subsequent debilitating fractures. A treatment that could simultaneously bolster both musculoskeletal systems would offer unprecedented advantages. Such a therapy could not only reduce the incidence of fractures but also improve mobility, balance, and overall functional independence in older individuals, thereby enhancing their quality of life and potentially reducing the immense healthcare burden associated with age-related decline. The ability to counteract both bone loss and muscle weakness with a single therapeutic agent represents a paradigm shift in the management of age-related musculoskeletal disorders.
Future Directions and the Path to Clinical Translation
While the findings are exceptionally promising, the research team is now intensely focused on continuing to study AP503 and GPR133 in greater detail. The journey from preclinical discovery to approved human therapy is long and rigorous, involving extensive safety and efficacy testing. Ongoing projects aim to further elucidate the precise molecular mechanisms by which GPR133 regulates bone and muscle metabolism, explore its potential role in other physiological or pathological conditions, and refine the compound AP503 for optimal pharmacological properties.
The next critical steps will involve advanced preclinical studies, including long-term animal models, to confirm the sustained efficacy and safety of AP503. If these studies yield positive results, the compound could then progress to human clinical trials, a multi-phase process designed to assess its safety, dosage, and effectiveness in patients. This translational research will be crucial in determining whether AP503 or similar GPR133 activators can fulfill their potential as a new class of drugs for osteoporosis and related musculoskeletal disorders.
A New Horizon for Bone Health and Active Aging
The identification of GPR133 as a key regulator of bone strength and the subsequent discovery of AP503 as its stimulator mark a significant milestone in the fight against osteoporosis. For a disease often dubbed "silent" because bone loss progresses without noticeable symptoms until a debilitating fracture occurs, this breakthrough offers a new direction for treatment that moves beyond merely slowing deterioration. Future therapies derived from this research could actively rebuild bone, restore strength, and potentially reverse the damage already done, offering a more complete solution than many existing options.
The potential societal impact of such a therapy is profound. By providing a means to actively strengthen both bone and muscle, it holds the promise of promoting healthier, more active aging, reducing the incidence of falls and fractures, and ultimately extending the years of independent living for millions worldwide. While further research and rigorous clinical development are essential, the findings from Leipzig University offer a powerful glimpse into a future where maintaining strong, healthy bones and muscles throughout life may become far more achievable, transforming the landscape of care for an aging global population.