Ovarian cancer has earned a grim reputation as the "silent killer" of gynecological oncology, primarily because its symptoms are often vague until the disease has reached an advanced and frequently terminal stage. While the rapid progression of this malignancy has long been documented by clinicians, the precise biological mechanisms that allow it to colonize the abdominal cavity with such speed have remained a profound mystery. However, a groundbreaking study led by researchers at Nagoya University in Japan has finally decoded a critical aspect of this process. The research, published in the prestigious journal Science Advances, identifies a sophisticated "outsourcing" strategy where cancer cells do not work alone but instead hijack healthy cells to facilitate their spread and protect them from medical intervention.
The study reveals that ovarian cancer cells actively recruit mesothelial cells—the protective cells that form the lining of the abdominal cavity—to act as biological pioneers. These recruited cells form hybrid clusters with the cancer cells, creating a formidable partnership that navigates the abdominal environment more effectively than solitary malignant cells. Most significantly, these hybrid spheres exhibit a heightened resistance to traditional chemotherapy, explaining why many patients suffer relapses even after seemingly successful initial treatments.
The Architecture of a Hybrid Threat
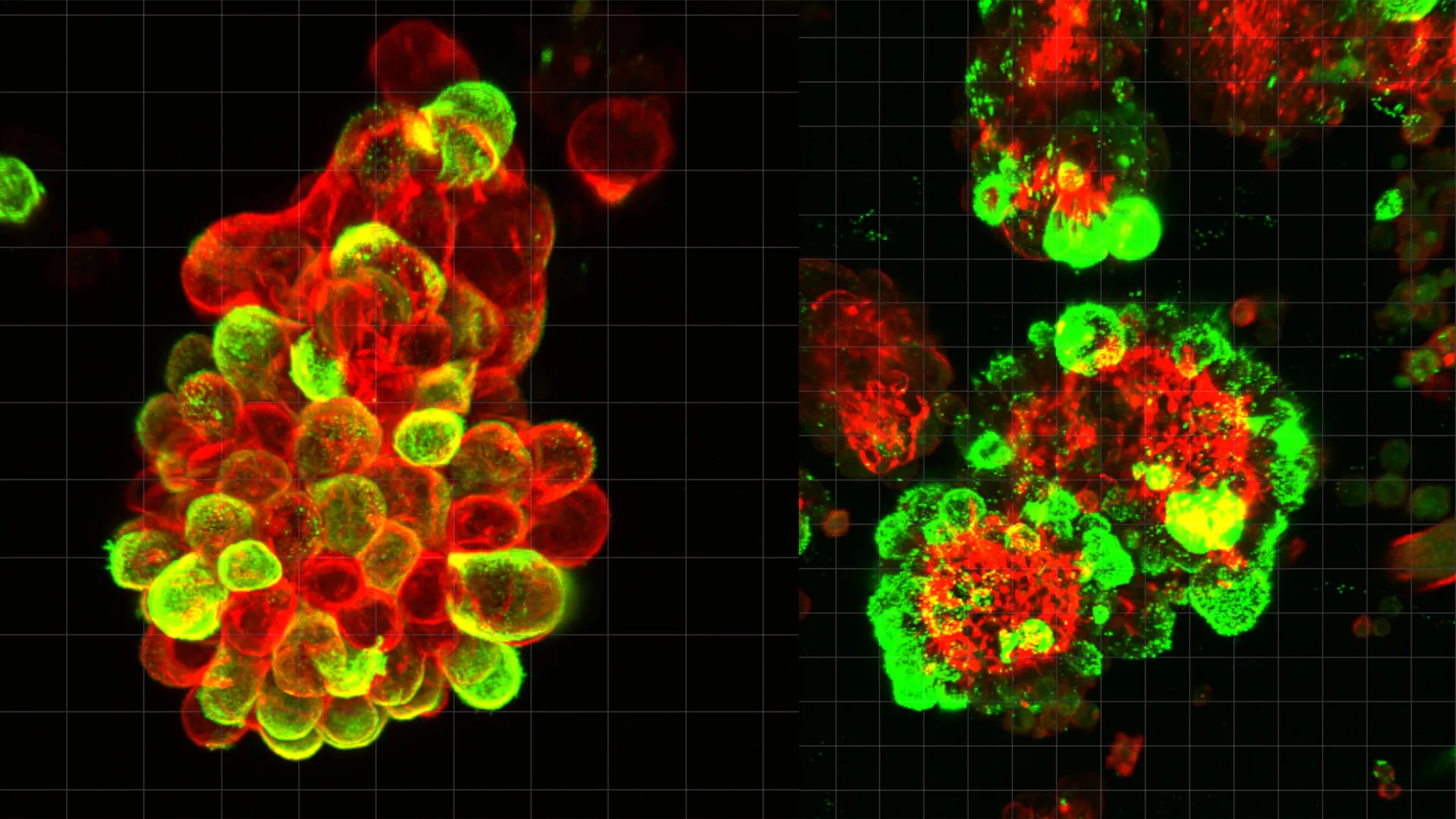
For decades, the prevailing scientific consensus suggested that ovarian cancer cells traveled through the abdominal cavity as individual units or simple clusters of malignant cells. To test this assumption, the Nagoya University team, led by Dr. Kaname Uno, meticulously analyzed abdominal fluid samples—known as ascites—from patients diagnosed with ovarian cancer. What they discovered was a complex cellular landscape that challenged previous models of metastasis.
The researchers found that cancer cells were rarely isolated. Instead, they were frequently found in "compact, mixed cell spheres" alongside mesothelial cells. According to the study’s data, approximately 60% of these floating cancer clusters contained recruited mesothelial cells. This was not a random association; it was a targeted biological recruitment.
The mechanism of this recruitment involves a specific signaling molecule known as Transforming Growth Factor-beta 1 (TGF-β1). The cancer cells secrete this protein, which acts as a molecular command, fundamentally altering the behavior and structure of the mesothelial cells. Under the influence of TGF-β1, the once-protective mesothelial cells undergo a transformation, developing sharp, spike-like protrusions called invadopodia. These structures act as biological "drill bits," capable of piercing through the tough membranes of organs within the abdomen, thereby clearing a path for the cancer cells to follow and embed themselves.
A Departure from Conventional Metastasis
To understand the significance of this discovery, it is necessary to contrast ovarian cancer’s behavior with that of other common malignancies. In cancers such as breast, lung, or prostate cancer, metastasis typically occurs through the hematogenous or lymphatic systems. Malignant cells break away from the primary tumor, enter the bloodstream or lymph vessels, and travel to distant organs like the brain, liver, or bones. Because these pathways are defined and the flow is somewhat predictable, doctors can often use blood tests or "liquid biopsies" to monitor the spread of the disease.
Ovarian cancer, however, utilizes a vastly different and more chaotic route. When cells detach from an ovarian tumor, they enter the peritoneal fluid that fills the space between abdominal organs. This fluid is in constant, unpredictable motion, driven by the rhythmic movements of the diaphragm during breathing and the peristaltic motions of the digestive tract.
This "floating phase" allows the cancer to reach almost any surface within the abdomen—the liver, the intestines, the stomach, and the omentum—without ever needing to enter the bloodstream. Until the publication of the Nagoya University study, the biological activity occurring during this floating phase was a "black box" for scientists. We now know that this is the period when the cancer cells are most active in forming their hybrid partnerships, preparing for the eventual invasion of new tissue.
The Role of Invadopodia in Tissue Colonization
The discovery of invadopodia in these hybrid clusters provides a clear explanation for the aggressive nature of ovarian cancer. When a solitary cancer cell lands on an organ, it must undergo significant internal changes to begin the process of invasion. However, by recruiting mesothelial cells that have already been "armed" with invadopodia, the cancer cell effectively bypasses this step.
The mesothelial cells, which are naturally native to the abdominal environment, act as the vanguard. They use their spike-like structures to breach the mesothelial lining of the target organ. Once the barrier is broken, the cancer cells follow through the opening. This collaborative effort makes the process of colonization significantly faster and more efficient.
Furthermore, the study highlighted a secondary, equally dangerous benefit of this partnership: chemoresistance. The hybrid spheres create a microenvironment that shields the internal cancer cells from the toxic effects of chemotherapy drugs. This protective "shielding" effect means that even if a treatment successfully kills off unattached cancer cells, the hybrid clusters remain viable, leading to the rapid recurrence of tumors that is so common in ovarian cancer cases.
Validation Through Advanced Microscopy and Genetic Analysis
The Nagoya University team utilized a multi-disciplinary approach to confirm their findings. Using advanced live-cell imaging and high-resolution microscopy, they were able to observe the formation of these hybrid clusters and the action of the invadopodia in real-time using patient-derived samples.
To ensure the clinical relevance of these observations, the researchers validated their findings using mouse models, where they could track the spread of the hybrid clusters through the peritoneal cavity. Additionally, they performed single-cell RNA sequencing to analyze gene activity within the clusters. This genetic analysis confirmed that the cancer cells themselves remained relatively stable in their genetic profile; they did not need to evolve into more invasive forms because they were successfully manipulating the mesothelial cells to perform the "heavy lifting" of tissue invasion.
Dr. Kaname Uno, the study’s lead author, noted the efficiency of this biological strategy. "The cancer cells undergo minimal genetic and molecular changes," Dr. Uno explained. "They simply migrate through the openings that the mesothelial cells create. It is a highly efficient way to spread without the metabolic cost of completely transforming their own cellular identity."
The Clinician’s Motivation: A Case for Early Detection
The impetus for this research was deeply personal for Dr. Uno. Before dedicating himself to laboratory research as a Visiting Researcher at Nagoya University’s Graduate School of Medicine, he spent eight years as a practicing gynecologist. During his clinical tenure, he encountered a patient whose case perfectly illustrated the limitations of current diagnostic capabilities.
The patient had undergone a routine gynecological screening and received a completely normal result. However, just three months later, she returned with symptoms that led to a diagnosis of advanced, Stage IV ovarian cancer. Despite the best efforts of the medical team, the disease had spread too far to be contained. The experience of seeing a patient go from "clear" to "terminal" in such a short window haunted Dr. Uno and drove him to investigate the underlying speed of ovarian cancer metastasis.
"Existing diagnostic tools failed to detect the disease early enough to save her life," Dr. Uno recalled. "That experience motivated me to investigate why ovarian cancer spreads so quickly and how it manages to escape the early detection methods we currently rely on."
Implications for Future Therapy and Diagnostics
The findings of this study have profound implications for the future of ovarian cancer treatment. Currently, the standard of care involves aggressive surgery followed by platinum-based chemotherapy. While this approach is often effective initially, the high rate of recurrence remains a major hurdle.
By identifying the TGF-β1 signaling pathway and the role of mesothelial cells, the Nagoya University study opens the door to several new therapeutic strategies:
- Targeting the Partnership: Future drugs could be designed to block the TGF-β1 signal, preventing cancer cells from "recruiting" mesothelial cells in the first place.
- Inhibiting Invadopodia: Therapies that specifically target the formation or function of the spike-like invadopodia could prevent the hybrid clusters from drilling into healthy tissue.
- Enhanced Monitoring: The presence of these hybrid cell clusters in abdominal fluid could serve as a new biomarker. By monitoring the concentration and activity of these clusters, doctors could more accurately predict how a patient’s disease will progress and tailor their treatment plans accordingly.
- Sensitizing Cells to Chemo: If the mechanism that allows these clusters to resist chemotherapy can be dismantled, existing drugs may become significantly more effective at eradicating the disease entirely.
Broader Impact on Gynecological Oncology
Ovarian cancer remains the deadliest gynecological malignancy worldwide. According to global health statistics, the five-year survival rate for ovarian cancer is significantly lower than that of breast or cervical cancer, largely due to the late stage of discovery. In the United States alone, the American Cancer Society estimates that over 13,000 women die from the disease annually.
The Nagoya University study provides a long-awaited biological explanation for the "silent" and "fast" nature of this cancer. By shifting the focus from the cancer cell in isolation to the cancer cell as a master of its environment, the research provides a new roadmap for oncology. It suggests that the key to defeating the most aggressive cancers may lie not just in attacking the malignant cells, but in protecting the healthy cells from being co-opted into the service of the tumor.
As the medical community moves toward "precision medicine," discoveries like these are essential. They allow for the development of treatments that are not only more effective but also more specific, potentially reducing the collateral damage associated with broad-spectrum chemotherapy. For the thousands of women diagnosed with ovarian cancer each year, this research offers a glimmer of hope that the "silent killer" may one day be silenced for good.