Northwestern University researchers have achieved a significant breakthrough in understanding and treating human spinal cord injuries by developing the most sophisticated lab-grown model to date. This innovative model, utilizing human spinal cord organoids, has successfully replicated the complex biological consequences of injury and demonstrated the potent regenerative capabilities of a promising new therapeutic approach known as "dancing molecules." The findings, published on February 11 in the prestigious journal Nature Biomedical Engineering, mark a critical step forward in the quest to restore function and improve recovery for individuals living with spinal cord damage.
A New Era in Spinal Cord Injury Research
For decades, the study of spinal cord injuries (SCIs) has been hampered by the limitations of existing models. Animal studies, while invaluable, do not always accurately reflect the intricate biological responses observed in humans. Furthermore, the ethical considerations and logistical challenges of human clinical trials mean that early-stage therapeutic development often relies on less direct methods. The advent of organoid technology has begun to bridge this gap, offering researchers the ability to study human tissue in a controlled laboratory environment.
The Northwestern team, led by Samuel I. Stupp, a Board of Trustees Professor at the university and a pioneer in regenerative materials science, has elevated this capability to an unprecedented level. Their human spinal cord organoids, derived from induced pluripotent stem cells, are not mere cellular aggregates; they are complex, multi-millimeter structures that mimic the architecture and cellular diversity of the human spinal cord. This advanced model has allowed researchers to recreate key features of SCI, including widespread cell death, significant inflammation, and the formation of glial scars.
Glial scarring is a particularly formidable obstacle in SCI recovery. Following injury, astrocytes, a type of glial cell, proliferate and form a dense, fibrous scar tissue. This scar acts as both a physical and chemical barrier, impeding the natural regeneration of severed nerve fibers, known as axons. The Northwestern model’s ability to faithfully reproduce this scarring phenomenon is a testament to its sophistication and its potential to accurately assess therapeutic interventions.
The Promise of "Dancing Molecules"
The true power of the Northwestern model was showcased when the researchers applied their novel "dancing molecules" therapy to the injured organoids. This therapy, a type of supramolecular therapeutic peptide (STP), was first introduced by Stupp’s lab in 2021. It leverages the controlled, dynamic movement of molecules to stimulate the body’s innate repair mechanisms.
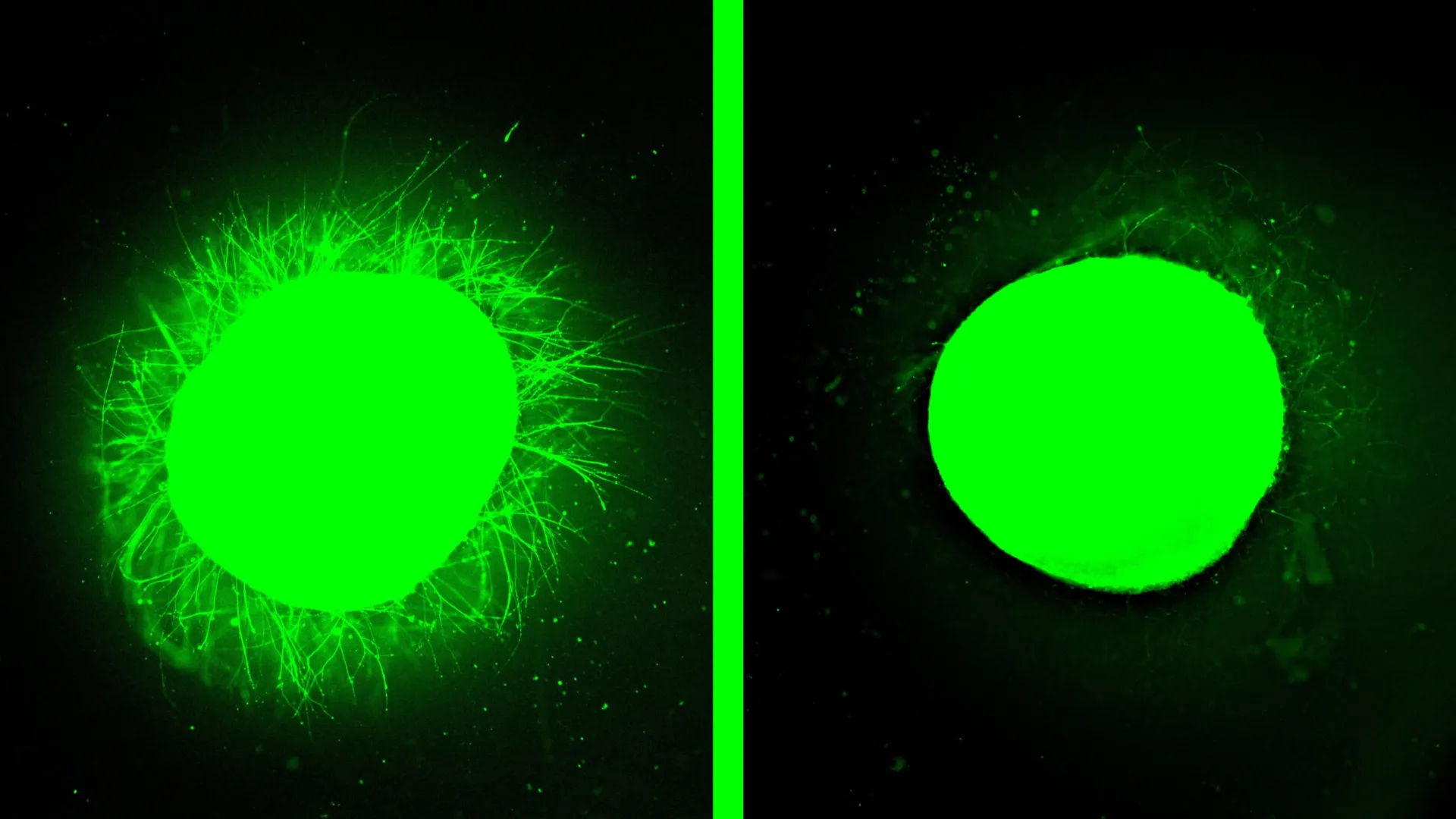
The results were nothing short of dramatic. Following treatment, the injured spinal cord organoids exhibited substantial neurite outgrowth, indicating that the long extensions that enable nerve cells to communicate were beginning to regrow. Crucially, the glial scar tissue, which had previously formed a substantial barrier, was significantly reduced, becoming barely detectable. This remarkable regeneration mirrors the positive outcomes observed in previous animal studies, where a single injection of the dancing molecules therapy led to significant functional recovery in mice.
"One of the most exciting aspects of organoids is that we can use them to test new therapies in human tissue," explained Stupp, the study’s senior author. "Short of a clinical trial, it’s the only way you can achieve this objective. We decided to develop two different injury models in a human spinal cord organoid and test our therapy to see if the results resembled what we previously saw in the animal model. After applying our therapy, the glial scar faded significantly to become barely detectable, and we saw neurites growing, resembling the axon regeneration we saw in animals. This is validation that our therapy has a good chance of working in humans."
Advancements in Organoid Technology
The development of these advanced spinal cord organoids involved several key innovations. Over several months, the research team meticulously guided stem cells to differentiate into the complex tissue found in the spinal cord, including neurons and astrocytes. A critical advancement was the incorporation of microglia, the resident immune cells of the central nervous system, into the organoid structure. This inclusion is vital for accurately modeling the inflammatory response that is a hallmark of spinal cord injury.
"It’s kind of a pseudo-organ," Stupp elaborated. "We were the first to introduce microglia into a human spinal cord organoid, so that was a huge accomplishment. It means that our organoid has all the chemicals that the resident immune system produces in response to an injury. That makes it a more realistic, accurate model of spinal cord injury."
This detailed cellular composition and structural organization allow the organoids to respond to injury in a manner that closely mirrors what occurs in a living human spinal cord. The ability to introduce specific types of trauma, such as lacerations or contusions, further enhances the model’s utility.
Understanding "Dancing Molecules"
The "dancing molecules" therapy represents a paradigm shift in regenerative medicine. Unlike traditional drug delivery methods that rely on static molecules, this therapy harnesses the power of dynamic molecular motion. The STPs, comprised of vast assemblies of molecules, are delivered as a liquid injection that rapidly self-assembles into a nanofiber scaffold, mimicking the extracellular matrix of the spinal cord.
The key to their efficacy lies in their ability to move dynamically within this scaffold. This motion allows the therapeutic molecules to efficiently interact with cell receptors, which are themselves in constant flux. "Given that cells themselves and their receptors are constantly in motion, you can imagine that molecules moving more rapidly would encounter these receptors more often," Stupp noted in a previous statement. "If the molecules are sluggish and not as ‘social,’ they may never come into contact with the cells."
This concept of enhanced molecular mobility leading to increased bioactivity and cellular signaling was first demonstrated in animal experiments. A single injection of the dancing molecules therapy, administered 24 hours post-injury, enabled mice to regain locomotion within four weeks. Further studies revealed that formulations with faster molecular motion yielded superior results, reinforcing the principle that dynamic movement is crucial for effective tissue repair.
Simulating Spinal Cord Trauma with Precision
To rigorously test the efficacy of the dancing molecules, the Northwestern researchers subjected their human spinal cord organoids to two distinct types of injury that are commonly encountered in human SCIs:
- Laceration Injury: Mimicking surgical trauma or penetrating wounds, some organoids were precisely cut with a scalpel.
- Compressive Contusion Injury: Replicating the blunt force trauma sustained in accidents like car crashes or falls, other organoids were subjected to controlled compression.
Both injury models successfully induced the characteristic biological responses of SCI, including widespread neuronal death and the formation of dense glial scars. The researchers were able to meticulously analyze these changes, observing the distinct morphology of reactive astrocytes within the glial scar and detecting the increased production of chondroitin sulfate proteoglycans, molecules known to inhibit nerve regeneration.
"We could distinguish between the astrocytes that are a part of normal tissue and the astrocytes in the glial scar, which are large and very densely packed," Stupp commented. "We also detected the production of chondroitin sulfate proteoglycans, which are molecules in the nervous system that respond to injury and disease."
A Glimpse of Functional Recovery
The therapeutic intervention with dancing molecules yielded compelling evidence of repair. The nanofiber scaffold not only helped to reduce inflammation and shrink the glial scarring but also demonstrably stimulated neurite extension. This regrowth of neurites, including axons, is pivotal for reconnecting severed neural pathways and restoring communication between nerve cells.
The ability to observe organized neuronal growth patterns within the organoids further underscores the therapy’s potential to promote functional recovery. In the context of SCI, severed axons lead to paralysis and loss of sensation. By encouraging axons to regrow and reconnect, therapies like the dancing molecules hold the promise of reversing these devastating consequences.
The Crucial Role of Molecular Dynamics
Stupp attributes the therapy’s success to its fundamental design principle: supramolecular motion. The ability of the molecules to move rapidly and even transiently detach from the nanofiber network appears to be critical for their interaction with the dynamic cellular environment. This was vividly illustrated in experiments conducted on healthy organoids.
"Before we even developed the injury model, we tested the therapy on a healthy organoid," Stupp recounted. "The dancing molecules spun out all these long neurites on the surface of the organoid but, when we used molecules that had less or no motion, we saw nothing. This difference was very vivid." This observation strongly suggests that the dynamic nature of the molecules is not merely a passive feature but an active driver of their therapeutic effect.
Future Directions and Broader Implications
The success of this study opens up exciting avenues for future research and development. The Northwestern team plans to engineer even more complex organoid models, including those capable of replicating chronic, long-standing injuries, which present unique challenges due to the more established and robust scar tissue.
Beyond the immediate therapeutic implications, the potential for personalized medicine is immense. As organoid technology advances, it may become possible to generate implantable spinal cord tissue from a patient’s own stem cells. This approach would significantly reduce the risk of immune rejection, a common complication in tissue transplantation, thereby paving the way for highly individualized treatment strategies.
The research was supported by the Center for Regenerative Nanomedicine at Northwestern University and a generous gift from the John Potocsnak Family, dedicated to advancing spinal cord injury research. This collaborative effort underscores the growing recognition of the urgency and importance of finding effective treatments for this debilitating condition. The development of this highly sophisticated human spinal cord injury model, coupled with the promising results of the dancing molecules therapy, represents a significant leap forward, offering renewed hope for millions affected by spinal cord injuries worldwide.