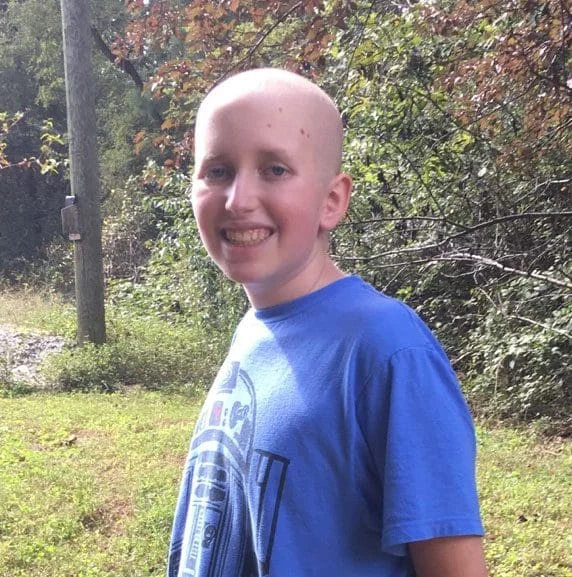
The diagnosis of pediatric cancer represents one of the most significant challenges in modern medicine, transforming the lives of families into a complex navigation of clinical trials, intensive therapies, and profound physiological strain. In July 2018, the Getchell family encountered this reality when 13-year-old Luke Getchell was diagnosed with acute myeloid leukemia (AML). The diagnosis was further complicated by the presence of a specific genetic alteration known as FLT3-ITD (Internal Tandem Duplication), a biomarker associated with a particularly aggressive disease course and a higher risk of relapse. Luke’s subsequent 14-month battle highlights not only the individual toll of hematologic malignancies but also the systemic need for more targeted, less toxic therapeutic interventions in pediatric oncology.
Clinical Chronology and Disease Progression
Luke Getchell’s medical journey began in the summer of 2018, marking the start of a rigorous clinical regimen designed to combat the rapid proliferation of leukemic cells. AML is characterized by the overproduction of immature white blood cells, which crowd out healthy blood cells in the bone marrow. The presence of the FLT3-ITD mutation in Luke’s case indicated a high leukemic burden and suggested that standard chemotherapy might be insufficient for long-term remission.
Throughout the latter half of 2018 and into early 2019, Luke underwent intensive cycles of chemotherapy. These treatments are designed to be cytotoxic, targeting rapidly dividing cells; however, their lack of specificity often leads to severe secondary health complications. Luke experienced a range of these adverse effects, including a serious colon infection, sepsis—a life-threatening systemic inflammatory response to infection—and measurable damage to his cardiac tissue.
By early 2019, approximately six months after his initial diagnosis, clinical tests indicated a complete remission, with no detectable signs of leukemia in his system. This period offered a brief window of clinical optimism for the Getchell family and his medical team at VCU Health in Richmond, Virginia. However, the reprieve was exceptionally short-lived. Within one week of the positive prognosis, Luke’s blood counts began to fluctuate, necessitating further diagnostic testing. The results confirmed a rapid and aggressive relapse, with the cancer spreading more extensively than during the initial presentation.
The post-relapse phase was marked by an escalation of physiological distress. Luke’s body, already weakened by months of high-dose chemotherapy, struggled to cope with the combined effects of the disease and continued medical intervention. He developed a myriad of complications, including graft-versus-host disease (GVHD)—a condition where donor immune cells attack the recipient’s body—and secondary diabetes induced by the prolonged use of prednisone, a corticosteroid used to manage inflammation and immune responses. By September 2019, the medical consensus reached a somber conclusion: the disease had become refractory to treatment. Luke was transitioned to hospice care and passed away on October 20, 2019, at the age of 14.
Understanding the FLT3-ITD Mutation in Pediatric AML
The specific genetic profile of Luke’s cancer, AML with FLT3-ITD, is a focal point for researchers seeking to improve survival rates. The FLT3 (Fms-like tyrosine kinase 3) gene plays a critical role in the proliferation and survival of hematopoietic stem cells. When an internal tandem duplication (ITD) occurs, the FLT3 receptor is constitutively activated, meaning it remains "switched on," signaling the cells to divide uncontrollably.
Statistically, while AML accounts for only about 20% of pediatric leukemias, it is responsible for a disproportionate number of leukemia-related deaths in children. The FLT3-ITD mutation is found in approximately 15% to 20% of pediatric AML cases. Patients with this mutation historically face a poorer prognosis compared to those with "wild-type" FLT3, as the mutation is often linked to a higher white blood cell count at diagnosis and a significantly higher rate of relapse following initial remission.
The challenge for oncologists lies in the balance between efficacy and toxicity. Current protocols for FLT3-ITD AML often involve the most intensive chemotherapy regimens available, followed by hematopoietic stem cell transplants. As seen in Luke’s case, the "collateral damage" of these treatments can be devastating, leading to long-term organ damage and acute secondary infections that can be as lethal as the primary malignancy.
The Socio-Institutional Response: "Luke’s Lego-cy"
In the wake of Luke’s passing, his memory has been institutionalized through both charitable efforts and physical tributes. Luke was known for his affinity for complex Lego models and his interest in culinary arts, having hosted "Chopped" style competitions in his home. These personal interests have been memorialized at the Children’s Wonder Tower at VCU Health. The "Luke’s Lego-cy" display serves as a permanent installation within the facility, providing a sense of community and normalcy for other children undergoing treatment.
Institutional responses to pediatric cancer deaths often focus on "legacy building," which helps families process grief while raising awareness for the specific needs of pediatric wards. For VCU Health, the inclusion of Luke’s models in the Wonder Tower represents a commitment to patient-centered care, acknowledging the personhood of the child beyond their clinical diagnosis.
The Research Frontier: Dr. Elvin Wagenblast and Mount Sinai
Addressing the limitations of current AML treatments requires a fundamental shift in how the disease is understood at the molecular level. A significant portion of current advocacy, including the fundraising efforts associated with Childhood Cancer Awareness Month, is directed toward the work of "Young Investigators" like Dr. Elvin Wagenblast at the Icahn School of Medicine at Mount Sinai.
Dr. Wagenblast’s research is focused on the "how" and "why" of leukemia development. By utilizing advanced CRISPR-Cas9 gene-editing technology and human stem cell models, his team aims to pinpoint the exact moment a healthy blood cell transforms into a leukemic one. Understanding these earliest evolutionary steps of the disease is crucial for developing "precision medicine" tools. If researchers can identify the specific pathways that allow FLT3-ITD cells to survive chemotherapy, they can develop targeted inhibitors that neutralize the cancer without damaging the heart, lungs, or immune system.
The goal of this research is to move away from the "sledgehammer" approach of traditional chemotherapy toward a "scalpel" approach. For children like Luke, who suffer from the extreme toxicity of medications designed for adults, these innovations are not merely academic—they are the difference between survival and terminal organ failure.
Data and Disparities in Pediatric Cancer Funding
Despite the severity of pediatric cancer, there remains a significant disparity in research funding. Data from the National Cancer Institute (NCI) and various pediatric cancer foundations suggest that only a small fraction of federal cancer research funding—estimated at approximately 4% to 8%—is dedicated specifically to childhood cancers. The majority of funding is directed toward adult cancers, such as breast, lung, and prostate cancer.
This funding gap has led to a reliance on philanthropic organizations to bridge the divide. Campaigns aiming to raise $25,000 or more for specific researchers are common because the cost of genomic sequencing, lab equipment, and clinical trials is immense. Furthermore, because pediatric cancers are biologically distinct from adult cancers, medications developed for adults cannot simply be "scaled down" for children. Children’s bodies are still developing, making them more susceptible to the long-term "late effects" of toxic treatments, including secondary cancers, infertility, and cognitive impairments.
Broader Implications and the Path Forward
The narrative of Luke Getchell is a poignant reminder of the current limitations of pediatric oncology. While overall survival rates for childhood cancers have improved significantly since the 1970s, those gains have not been distributed equally across all cancer types. Aggressive subtypes like FLT3-ITD AML continue to have survival rates that lag behind more common forms of leukemia, such as Acute Lymphoblastic Leukemia (ALL).
The implications of Luke’s story extend to healthcare policy and the pharmaceutical industry. There is an urgent need for the "RACE for Children Act" (Research to Accelerate Cure and Equity) and similar legislation to be fully leveraged to compel pharmaceutical companies to study adult oncology drugs in pediatric populations when the molecular targets are similar.
Furthermore, the emphasis on Childhood Cancer Awareness Month serves as a critical period for public education. It highlights the reality that "curing" cancer is not the only goal; the goal must be to cure cancer with a high quality of life. The horrors described by the Getchell family—the bleeding, the loss of vision, and the inability to walk—are the direct results of a medical system that, for many decades, has had to prioritize survival at any cost.
As the medical community continues to honor the memory of patients like Luke, the focus remains steadfast on the next generation of therapies. Through the support of researchers like Dr. Wagenblast and the continued advocacy of families and foundations, the objective is clear: to ensure that the "prognosis" for future children diagnosed with AML is defined by hope and healing rather than toxicity and loss. The legacy of Luke Getchell lives on not only in the Lego displays of Richmond but in the pursuit of a future where no child has to endure the devastating side effects of the very medicine meant to save them.